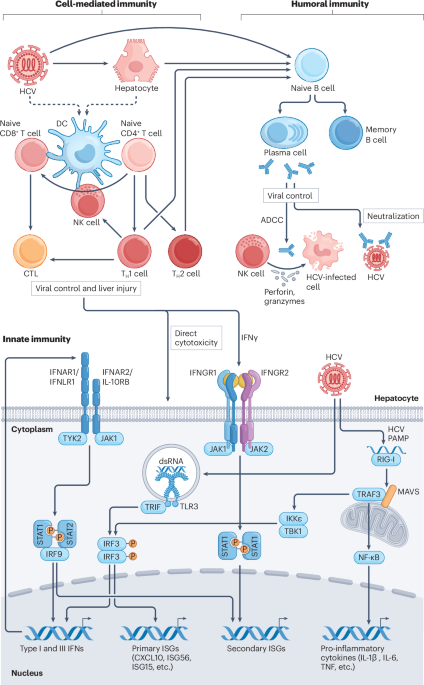
World Health Organization. Global Hepatitis Report 2024 (WHO, 2024).
Martinello, M. et al. Hepatitis C. Lancet 402, 1085–1096 (2023).
Trickey, A. et al. The contribution of injection drug use to hepatitis C virus transmission globally, regionally, and at country level: a modelling study. Lancet Gastroenterol. Hepatol. 4, 435–444 (2019).
Ly, K. N. et al. Deaths associated with hepatitis C virus infection among residents in 50 states and the District of Columbia, 2016-2017. Clin. Infect. Dis. 71, 1149–1160 (2020).
Hensel, N. et al. Memory-like HCV-specific CD8+ T cells retain a molecular scar after cure of chronic HCV infection. Nat. Immunol. 22, 229–239 (2021).
Tonnerre, P. et al. Differentiation of exhausted CD8+ T cells after termination of chronic antigen stimulation stops short of achieving functional T cell memory. Nat. Immunol. 22, 1030–1041 (2021).
Yates, K. B. et al. Epigenetic scars of CD8+ T cell exhaustion persist after cure of chronic infection in humans. Nat. Immunol. 22, 1020–1029 (2021).
Kim, S. Y. & Shin, E. C. Reply to: “Regulatory T cell dynamics during and after chronic hepatitis C treatment”. J. Hepatol. 81, e250 (2024).
Vo-Quang, E. & Pawlotsky, J. M. ‘Unusual’ HCV genotype subtypes: origin, distribution, sensitivity to direct-acting antiviral drugs and behaviour on antiviral treatment and retreatment. Gut 73, 1570–1582 (2024).
Farci, P. New insights into the HCV quasispecies and compartmentalization. Semin. Liver Dis. 31, 356–374 (2011).
Tanaka, Y. et al. Genome-wide association of IL28B with response to pegylated interferon-alpha and ribavirin therapy for chronic hepatitis C. Nat. Genet. 41, 1105–1109 (2009).
Rauch, A. et al. Genetic variation in IL28B is associated with chronic hepatitis C and treatment failure: a genome-wide association study. Gastroenterology 138, 1338–1345 (2010).
Prokunina-Olsson, L. et al. A variant upstream of IFNL3 (IL28B) creating a new interferon gene IFNL4 is associated with impaired clearance of hepatitis C virus. Nat. Genet. 45, 164–171 (2013). This work identifies the novel IFNL4 gene, created by a variant upstream of IFNL3, which advances our understanding of genetic factors affecting hepatitis C virus clearance and therapeutic outcomes.
Eslam, M. et al. IFN-λ3, not IFN-λ4, likely mediates IFNL3–IFNL4 haplotype-dependent hepatic inflammation and fibrosis. Nat. Genet. 49, 795–800 (2017).
Rehermann, B. Pathogenesis of chronic viral hepatitis: differential roles of T cells and NK cells. Nat. Med. 19, 859–868 (2013).
Hajarizadeh, B., Grebely, J. & Dore, G. J. Epidemiology and natural history of HCV infection. Nat. Rev. Gastroenterol. Hepatol. 10, 553–562 (2013).
Schulze zur Wiesch, J. et al. Broad repertoire of the CD4+ Th cell response in spontaneously controlled hepatitis C virus infection includes dominant and highly promiscuous epitopes. J. Immunol. 175, 3603–3613 (2005).
Pestka, J. M. et al. Rapid induction of virus-neutralizing antibodies and viral clearance in a single-source outbreak of hepatitis C. Proc. Natl Acad. Sci. USA 104, 6025–6030 (2007). This work shows that rapid induction of neutralizing antibodies in acute HCV infection correlates with viral clearance, providing key insights for vaccine strategies.
Khakoo, S. I. et al. HLA and NK cell inhibitory receptor genes in resolving hepatitis C virus infection. Science 305, 872–874 (2004).
Lindenbach, B. D. & Rice, C. M. The ins and outs of hepatitis C virus entry and assembly. Nat. Rev. Microbiol. 11, 688–700 (2013).
Kong, L. et al. Structural flexibility at a major conserved antibody target on hepatitis C virus E2 antigen. Proc. Natl Acad. Sci. USA 113, 12768–12773 (2016). This work shows that structural flexibility of HCV E2 at the conserved CD81-binding site challenges vaccine design, emphasizing the need for stabilization to elicit broadly neutralizing antibodies.
Dowd, K. A. et al. Selection pressure from neutralizing antibodies drives sequence evolution during acute infection with hepatitis C virus. Gastroenterology 136, 2377–2386 (2009). This work shows that neutralizing antibodies drive viral evolution during acute HCV infection, shaping outcomes and underscoring their significance for vaccine development.
El-Diwany, R. et al. Extra-epitopic hepatitis C virus polymorphisms confer resistance to broadly neutralizing antibodies by modulating binding to scavenger receptor B1. PLoS Pathog. 13, e1006235 (2017).
Frumento, N., Flyak, A. I. & Bailey, J. R. Mechanisms of HCV resistance to broadly neutralizing antibodies. Curr. Opin. Virol. 50, 23–29 (2021).
Nishio, A. et al. Serum neutralization activity declines but memory B cells persist after cure of chronic hepatitis C. Nat. Commun. 13, 5446 (2022).
Raghuraman, S. et al. Spontaneous clearance of chronic hepatitis C virus infection is associated with appearance of neutralizing antibodies and reversal of T-cell exhaustion. J. Infect. Dis. 205, 763–771 (2012).
Meuleman, P. et al. In vivo evaluation of the cross-genotype neutralizing activity of polyclonal antibodies against hepatitis C virus. Hepatology 53, 755–762 (2011).
Bukh, J. et al. Immunoglobulin with high-titer in vitro cross-neutralizing hepatitis C virus antibodies passively protects chimpanzees from homologous, but not heterologous, challenge. J. Virol. 89, 9128–9132 (2015).
Farci, P. et al. Prevention of hepatitis C virus infection in chimpanzees by hyperimmune serum against the hypervariable region 1 of the envelope 2 protein. Proc. Natl Acad. Sci. USA 93, 15394–15399 (1996).
Vieyres, G., Dubuisson, J. & Patel, A. H. Characterization of antibody-mediated neutralization directed against the hypervariable region 1 of hepatitis C virus E2 glycoprotein. J. Gen. Virol. 92, 494–506 (2011).
Bankwitz, D. et al. Hepatitis C virus hypervariable region 1 modulates receptor interactions, conceals the CD81 binding site, and protects conserved neutralizing epitopes. J. Virol. 84, 5751–5763 (2010).
Prentoe, J. et al. Hypervariable region 1 shielding of hepatitis C virus is a main contributor to genotypic differences in neutralization sensitivity. Hepatology 64, 1881–1892 (2016).
Goffard, A. & Dubuisson, J. Glycosylation of hepatitis C virus envelope proteins. Biochimie 85, 295–301 (2003).
Kong, L. et al. Hepatitis C virus E2 envelope glycoprotein core structure. Science 342, 1090–1094 (2013). This paper shows that the first partial structural resolution of HCV envelope proteins provides crucial insight into HCV infection, followed by other publications with additional structural details.
Alzua, G. P. et al. Identification of novel neutralizing determinants for protection against HCV. Hepatology 77, 982–996 (2023).
Yan, Y. et al. A nanoparticle-based hepatitis C virus vaccine with enhanced potency. J. Infect. Dis. 221, 1304–1314 (2020).
Broering, T. J. et al. Identification and characterization of broadly neutralizing human monoclonal antibodies directed against the E2 envelope glycoprotein of hepatitis C virus. J. Virol. 83, 12473–12482 (2009).
Morin, T. J. et al. Human monoclonal antibody HCV1 effectively prevents and treats HCV infection in chimpanzees. PLoS Pathog. 8, e1002895 (2012).
Keck, Z. et al. Cooperativity in virus neutralization by human monoclonal antibodies to two adjacent regions located at the amino terminus of hepatitis C virus E2 glycoprotein. J. Virol. 87, 37–51 (2013).
Stroh, L. J., Nagarathinam, K. & Krey, T. Conformational flexibility in the CD81-binding site of the hepatitis C virus glycoprotein E2. Front. Immunol. 9, 1396 (2018).
Tzarum, N., Wilson, I. A. & Law, M. The neutralizing face of hepatitis C virus E2 envelope glycoprotein. Front. Immunol. 9, 1315 (2018).
Flint, M. et al. Characterization of hepatitis C virus E2 glycoprotein interaction with a putative cellular receptor, CD81. J. Virol. 73, 6235–6244 (1999).
Ray, R. et al. Characterization of antibodies induced by vaccination with hepatitis C virus envelope glycoproteins. J. Infect. Dis. 202, 862–866 (2010).
Sabo, M. C. et al. Neutralizing monoclonal antibodies against hepatitis C virus E2 protein bind discontinuous epitopes and inhibit infection at a postattachment step. J. Virol. 85, 7005–7019 (2011).
Pantua, H. et al. Glycan shifting on hepatitis C virus (HCV) E2 glycoprotein is a mechanism for escape from broadly neutralizing antibodies. J. Mol. Biol. 425, 1899–1914 (2013).
Alhammad, Y. et al. Monoclonal antibodies directed toward the hepatitis C virus glycoprotein E2 detect antigenic differences modulated by the N-terminal hypervariable region 1 (HVR1), HVR2, and intergenotypic variable region. J. Virol. 89, 12245–12261 (2015).
Tzarum, N. et al. Genetic and structural insights into broad neutralization of hepatitis C virus by human VH1-69 antibodies. Sci. Adv. 5, eaav1882 (2019).
Keck, Z. Y. et al. Mutations in hepatitis C virus E2 located outside the CD81 binding sites lead to escape from broadly neutralizing antibodies but compromise virus infectivity. J. Virol. 83, 6149–6160 (2009).
Meola, A. et al. Structural flexibility of a conserved antigenic region in hepatitis C virus glycoprotein E2 recognized by broadly neutralizing antibodies. J. Virol. 89, 2170–2181 (2015).
Duan, H. et al. Amino acid residue-specific neutralization and nonneutralization of hepatitis C virus by monoclonal antibodies to the E2 protein. J. Virol. 86, 12686–12694 (2012).
Keck, Z. Y. et al. Human monoclonal antibodies to a novel cluster of conformational epitopes on HCV E2 with resistance to neutralization escape in a genotype 2a isolate. PLoS Pathog. 8, e1002653 (2012).
Law, M. et al. Broadly neutralizing antibodies protect against hepatitis C virus quasispecies challenge. Nat. Med. 14, 25–27 (2008).
Gopal, R. et al. Probing the antigenicity of hepatitis C virus envelope glycoprotein complex by high-throughput mutagenesis. PLoS Pathog. 13, e1006735 (2017).
Johansson, D. X. et al. Human combinatorial libraries yield rare antibodies that broadly neutralize hepatitis C virus. Proc. Natl Acad. Sci. USA 104, 16269–16274 (2007).
Giang, E. et al. Human broadly neutralizing antibodies to the envelope glycoprotein complex of hepatitis C virus. Proc. Natl Acad. Sci. USA 109, 6205–6210 (2012). This paper shows that broadly neutralizing antibodies targeting conserved epitopes of the E1E2 glycoprotein complex of HCV provide critical insights for effective vaccine design.
Colbert, M. D. et al. Broadly neutralizing antibodies targeting new sites of vulnerability in hepatitis C virus E1E2. J. Virol. 93, e02070-18 (2019).
Meunier, J. C. et al. Isolation and characterization of broadly neutralizing human monoclonal antibodies to the E1 glycoprotein of hepatitis C virus. J. Virol. 82, 966–973 (2008).
Augestad, E. H. et al. Global and local envelope protein dynamics of hepatitis C virus determine broad antibody sensitivity. Sci. Adv. 6, eabb5938 (2020).
Keck, Z. Y. et al. Human monoclonal antibody to hepatitis C virus E1 glycoprotein that blocks virus attachment and viral infectivity. J. Virol. 78, 7257–7263 (2004).
Meertens, L. et al. The tight junction proteins claudin-1, -6, and -9 are entry cofactors for hepatitis C virus. J. Virol. 82, 3555–3560 (2008).
Hopcraft, S. E. & Evans, M. J. Selection of a hepatitis C virus with altered entry factor requirements reveals a genetic interaction between the E1 glycoprotein and claudins. Hepatology 62, 1059–1069 (2015).
Cheng, J. J. et al. CD36 is a co-receptor for hepatitis C virus E1 protein attachment. Sci. Rep. 6, 21808 (2016).
Bailey, J. R. et al. Naturally selected hepatitis C virus polymorphisms confer broad neutralizing antibody resistance. J. Clin. Invest. 125, 437–447 (2015).
Holz, L. & Rehermann, B. T cell responses in hepatitis C virus infection: historical overview and goals for future research. Antivir. Res. 114, 96–105 (2015). This paper shows that cell responses are pivotal in determining HCV infection outcomes, providing crucial insights for vaccine development and immune-based therapies.
Kasprowicz, V. et al. High level of PD-1 expression on hepatitis C virus (HCV)-specific CD8+ and CD4+ T cells during acute HCV infection, irrespective of clinical outcome. J. Virol. 82, 3154–3160 (2008).
Chen, D. Y. et al. Hepatitis C virus-specific CD4+ T cell phenotype and function in different infection outcomes. J. Clin. Invest. 130, 768–773 (2020).
Kurktschiev, P. D. et al. Dysfunctional CD8+ T cells in hepatitis B and C are characterized by a lack of antigen-specific T-bet induction. J. Exp. Med. 211, 2047–2059 (2014).
Lechner, F. et al. Analysis of successful immune responses in persons infected with hepatitis C virus. J. Exp. Med. 191, 1499–1512 (2000).
Luxenburger, H. et al. HCV-specific T cell responses during and after chronic HCV infection. Viruses 10, 645 (2018).
Schulze Zur Wiesch, J. et al. Broadly directed virus-specific CD4+ T cell responses are primed during acute hepatitis C infection, but rapidly disappear from human blood with viral persistence. J. Exp. Med. 209, 61–75 (2012).
Grakoui, A. et al. HCV persistence and immune evasion in the absence of memory T cell help. Science 302, 659–662 (2003). This research shows that the absence of T helper cell response results in HCV chronicity, demonstrating the critical importance of T cell responses in controlling HCV infection.
Alfei, F. et al. TOX reinforces the phenotype and longevity of exhausted T cells in chronic viral infection. Nature 571, 265–269 (2019).
Khan, O. et al. TOX transcriptionally and epigenetically programs CD8+ T cell exhaustion. Nature 571, 211–218 (2019).
Utzschneider, D. T. et al. T cell factor 1-expressing memory-like CD8+ T cells sustain the immune response to chronic viral infections. Immunity 45, 415–427 (2016).
Cox, A. L. et al. Comprehensive analyses of CD8+ T cell responses during longitudinal study of acute human hepatitis C. Hepatology 42, 104–112 (2005).
Dazert, E. et al. Loss of viral fitness and cross-recognition by CD8+ T cells limit HCV escape from a protective HLA-B27-restricted human immune response. J. Clin. Invest. 119, 376–386 (2009).
Raziorrouh, B. et al. Inhibitory molecules that regulate expansion and restoration of HCV-specific CD4+ T cells in patients with chronic infection. Gastroenterology 141, 1422–1431 (2011).
Lindenbach, B. D. & Rice, C. M. Unravelling hepatitis C virus replication from genome to function. Nature 436, 933–938 (2005).
Filskov, J. et al. HCV p7 as a novel vaccine-target inducing multifunctional CD4+ and CD8+ T-cells targeting liver cells expressing the viral antigen. Sci. Rep. 9, 14085 (2019).
Youn, J. W. et al. Evidence for protection against chronic hepatitis C virus infection in chimpanzees by immunization with replicating recombinant vaccinia virus. J. Virol. 82, 10896–10905 (2008).
Wada, T., Kohara, M. & Yasutomi, Y. DNA vaccine expressing the non-structural proteins of hepatitis C virus diminishes the expression of HCV proteins in a mouse model. Vaccine 31, 5968–5974 (2013).
Wertheimer, A. M. et al. Novel CD4+ and CD8+ T-cell determinants within the NS3 protein in subjects with spontaneously resolved HCV infection. Hepatology 37, 577–589 (2003). This work shows that persistent multi-specific CD4+ and CD8+ T cell responses targeting the NS3 protein of HCV in individuals with resolved infection underscore its potential as a key vaccine candidate.
Zeng, R. et al. A novel combined vaccine candidate containing epitopes of HCV NS3, core and E1 proteins induces multi-specific immune responses in BALB/c mice. Antivir. Res. 84, 23–30 (2009).
Lang Kuhs, K. A. et al. Hepatitis C virus NS3/NS4A DNA vaccine induces multiepitope T cell responses in rhesus macaques mimicking human immune responses [corrected]. Mol. Ther. 20, 669–678 (2012).
Tan, W. G. et al. Qualitative differences in cellular immunogenicity elicited by hepatitis C virus T-cell vaccines employing prime-boost regimens. PLoS ONE 12, e0181578 (2017).
Dawood, R. M. et al. A multiepitope peptide vaccine against HCV stimulates neutralizing humoral and persistent cellular responses in mice. BMC Infect. Dis. 19, 932 (2019).
Han, J. W. et al. IFNL3-adjuvanted HCV DNA vaccine reduces regulatory T cell frequency and increases virus-specific T cell responses. J. Hepatol. 73, 72–83 (2020).
Holmstrom, F. et al. A synthetic codon-optimized hepatitis C virus nonstructural 5A DNA vaccine primes polyfunctional CD8+ T cell responses in wild-type and NS5A-transgenic mice. J. Immunol. 190, 1113–1124 (2013).
Polyak, S. J. et al. Induction of intrahepatic HCV NS4B, NS5A and NS5B-specific cellular immune responses following peripheral immunization. PLoS ONE 7, e52165 (2012).
Mekonnen, Z. A. et al. Single-dose vaccination with a hepatotropic adeno-associated virus efficiently localizes T cell immunity in the liver with the potential to confer rapid protection against hepatitis C virus. J. Virol. 93, e00202-19 (2019).
Donnison, T. et al. A pan-genotype hepatitis C virus viral vector vaccine generates T cells and neutralizing antibodies in mice. Hepatology 76, 1190–1202 (2022).
Burke, K. P. et al. Immunogenicity and cross-reactivity of a representative ancestral sequence in hepatitis C virus infection. J. Immunol. 188, 5177–5188 (2012).
Meissner, E. G. et al. IFNL4-ΔG genotype is associated with slower viral clearance in hepatitis C, genotype-1 patients treated with sofosbuvir and ribavirin. J. Infect. Dis. 209, 1700–1704 (2014).
O’Brien, T. R. et al. Comparison of functional variants in IFNL4 and IFNL3 for association with HCV clearance. J. Hepatol. 63, 1103–1110 (2015).
Terczynska-Dyla, E. et al. Reduced IFNλ4 activity is associated with improved HCV clearance and reduced expression of interferon-stimulated genes. Nat. Commun. 5, 5699 (2014).
Hamming, O. J. et al. Interferon lambda 4 signals via the IFNλ receptor to regulate antiviral activity against HCV and coronaviruses. EMBO J. 32, 3055–3065 (2013).
Hong, M. et al. Interferon lambda 4 expression is suppressed by the host during viral infection. J. Exp. Med. 213, 2539–2552 (2016).
Grubbe, W. S. et al. Structural studies of the IFNλ4 receptor complex using cryoEM enabled by protein engineering. Nat. Commun. 16, 818 (2025).
Sung, P. S. et al. IFN-λ4 potently blocks IFN-α signalling by ISG15 and USP18 in hepatitis C virus infection. Sci. Rep. 7, 3821 (2017).
Chen, Q. et al. Interferon lambda 4 impairs hepatitis C viral antigen presentation and attenuates T cell responses. Nat. Commun. 12, 4882 (2021).
McFarland, A. P. et al. The favorable IFNL3 genotype escapes mRNA decay mediated by AU-rich elements and hepatitis C virus-induced microRNAs. Nat. Immunol. 15, 72–79 (2014).
Neumann-Haefelin, C. et al. Protective effect of human leukocyte antigen B27 in hepatitis C virus infection requires the presence of a genotype-specific immunodominant CD8+ T-cell epitope. Hepatology 51, 54–62 (2010).
Neumann-Haefelin, C. et al. Human leukocyte antigen B27 selects for rare escape mutations that significantly impair hepatitis C virus replication and require compensatory mutations. Hepatology 54, 1157–1166 (2011).
Kim, A. Y. et al. Spontaneous control of HCV is associated with expression of HLA-B*57 and preservation of targeted epitopes. Gastroenterology 140, 686–696.e1 (2011).
Merani, S. et al. Effect of immune pressure on hepatitis C virus evolution: insights from a single-source outbreak. Hepatology 53, 396–405 (2011).
Kuniholm, M. H. et al. Specific human leukocyte antigen class I and II alleles associated with hepatitis C virus viremia. Hepatology 51, 1514–1522 (2010).
Valencia, A. et al. Trans-ancestral fine-mapping of MHC reveals key amino acids associated with spontaneous clearance of hepatitis C in HLA-DQβ1. Am. J. Hum. Genet. 109, 299–310 (2022).
Liang, T. J. Current progress in development of hepatitis C virus vaccines. Nat. Med. 19, 869–878 (2013).
Pihl, A. F. et al. Inactivated whole hepatitis C virus vaccine employing a licensed adjuvant elicits cross-genotype neutralizing antibodies in mice. J. Hepatol. 76, 1051–1061 (2022).
Alzua, G. P. et al. Inactivated genotype 1a, 2a and 3a HCV vaccine candidates induced broadly neutralising antibodies in mice. Gut 72, 560–572 (2023).
Akazawa, D. et al. Neutralizing antibodies induced by cell culture-derived hepatitis C virus protect against infection in mice. Gastroenterology 145, 447–455 (2013).
Yokokawa, H. et al. Induction of humoural and cellular immunity by immunisation with HCV particle vaccine in a non-human primate model. Gut 67, 372–379 (2018).
Bankwitz, D. et al. Maturation of secreted HCV particles by incorporation of secreted ApoE protects from antibodies by enhancing infectivity. J. Hepatol. 67, 480–489 (2017).
Sepulveda-Crespo, D., Resino, S. & Martinez, I. Hepatitis C virus vaccine design: focus on the humoral immune response. J. Biomed. Sci. 27, 78 (2020).
Mosa, A. I. et al. Polyvalent immunization elicits a synergistic broadly neutralizing immune response to hypervariable region 1 variants of hepatitis C virus. Proc. Natl Acad. Sci. USA 120, e2220294120 (2023).
Johnson, J. et al. A recombinant hepatitis C virus genotype 1a E1/E2 envelope glycoprotein vaccine elicits antibodies that differentially neutralize closely related 2a strains through interactions of the N-terminal hypervariable region 1 of E2 with scavenger receptor B1. J. Virol. 93, e00810–e00819 (2019).
Khan, A. G. et al. Structure of the core ectodomain of the hepatitis C virus envelope glycoprotein 2. Nature 509, 381–384 (2014).
Nagarathinam, K. et al. Epitope-focused immunogens targeting the hepatitis C virus glycoproteins induce broadly neutralizing antibodies. Sci. Adv. 10, eado2600 (2024). This work shows that epitope-focused immunogens targeting HCV glycoproteins elicit broadly neutralizing antibodies, representing a promising strategy for effective vaccine design.
Vietheer, P. T. et al. The core domain of hepatitis C virus glycoprotein E2 generates potent cross‐neutralizing antibodies in guinea pigs. Hepatology 65, 1117–1131 (2017).
Center, R. J. et al. Enhancing the antigenicity and immunogenicity of monomeric forms of hepatitis C virus E2 for use as a preventive vaccine. J. Biol. Chem. 295, 7179–7192 (2020).
Li, D. et al. Altered glycosylation patterns increase immunogenicity of a subunit hepatitis C virus vaccine, inducing neutralizing antibodies which confer protection in mice. J. Virol. 90, 10486–10498 (2016).
Li, D. et al. Immunization with a subunit hepatitis C virus vaccine elicits pan-genotypic neutralizing antibodies and intrahepatic T-cell responses in nonhuman primates. J. Infect. Dis. 215, 1824–1831 (2017).
Wang, X. et al. A trivalent HCV vaccine elicits broad and synergistic polyclonal antibody response in mice and rhesus monkey. Gut 68, 140–149 (2019).
Tarr, A. W. et al. Immunization with a synthetic consensus hepatitis C virus E2 glycoprotein ectodomain elicits virus-neutralizing antibodies. Antivir. Res. 160, 25–37 (2018).
Meunier, J. C. et al. Vaccine-induced cross-genotype reactive neutralizing antibodies against hepatitis C virus. J. Infect. Dis. 204, 1186–1190 (2011).
Wang, R. et al. Induction of broadly neutralizing antibodies using a secreted form of the hepatitis C virus E1E2 heterodimer as a vaccine candidate. Proc. Natl Acad. Sci. USA 119, e2112008119 (2022).
Kundu, J. et al. Recombinant H77C gpE1/gpE2 heterodimer elicits superior HCV cross-neutralisation than H77C gpE2 alone. J. Hepatol. 81, 941–948 (2024).
Frey, S. E. et al. Safety and immunogenicity of HCV E1E2 vaccine adjuvanted with MF59 administered to healthy adults. Vaccine 28, 6367–6373 (2010).
Krapchev, V. B. et al. Recombinant Flag-tagged E1E2 glycoproteins from three hepatitis C virus genotypes are biologically functional and elicit cross-reactive neutralizing antibodies in mice. Virology 519, 33–41 (2018).
Lin, T. et al. Recombinant full-length hepatitis C virus E1E2 dimer elicits pangenotypic neutralizing antibodies. Front. Immunol. 13, 831285 (2022).
Barnes, E. et al. Novel adenovirus-based vaccines induce broad and sustained T cell responses to HCV in man. Sci. Transl. Med. 4, 115ra1 (2012).
Swadling, L. et al. A human vaccine strategy based on chimpanzee adenoviral and MVA vectors that primes, boosts, and sustains functional HCV-specific T cell memory. Sci. Transl. Med. 6, 261ra153 (2014).
Kelly, C. et al. Chronic hepatitis C viral infection subverts vaccine-induced T-cell immunity in humans. Hepatology 63, 1455–1470 (2016).
von Delft, A. et al. The generation of a simian adenoviral vectored HCV vaccine encoding genetically conserved gene segments to target multiple HCV genotypes. Vaccine 36, 313–321 (2018).
Page, K. et al. Randomized trial of a vaccine regimen to prevent chronic HCV infection. N. Engl. J. Med. 384, 541–549 (2021). This paper provides that heterologous prime-boost HCV vaccine regimen showed immunogenicity but failed to prevent chronic infection, underscoring challenges in vaccine efficacy.
Reyes-del Valle, J. et al. Broadly neutralizing immune responses against hepatitis C virus induced by vectored measles viruses and a recombinant envelope protein booster. J. Virol. 86, 11558–11566 (2012).
Baumert, T. F. et al. Hepatitis C virus structural proteins assemble into viruslike particles in insect cells. J. Virol. 72, 3827–3836 (1998).
Lechmann, M. et al. Hepatitis C virus-like particles induce virus-specific humoral and cellular immune responses in mice. Hepatology 34, 417–423 (2001).
Jeong, S. H. et al. Immunization with hepatitis C virus-like particles induces humoral and cellular immune responses in nonhuman primates. J. Virol. 78, 6995–7003 (2004).
Elmowalid, G. A. et al. Immunization with hepatitis C virus-like particles results in control of hepatitis C virus infection in chimpanzees. Proc. Natl Acad. Sci. USA 104, 8427–8432 (2007).
Earnest-Silveira, L. et al. Characterization of a hepatitis C virus-like particle vaccine produced in a human hepatocyte-derived cell line. J. Gen. Virol. 97, 1865–1876 (2016).
Christiansen, D. et al. Pre-clinical evaluation of a quadrivalent HCV VLP vaccine in pigs following microneedle delivery. Sci. Rep. 9, 9251 (2019).
Hourioux, C. et al. Core protein domains involved in hepatitis C virus-like particle assembly and budding at the endoplasmic reticulum membrane. Cell Microbiol. 9, 1014–1027 (2007).
Beaumont, E. et al. Chimeric hepatitis B virus/hepatitis C virus envelope proteins elicit broadly neutralizing antibodies and constitute a potential bivalent prophylactic vaccine. Hepatology 57, 1303–1313 (2013).
Beaumont, E. et al. Mixing particles from various HCV genotypes increases the HBV-HCV vaccine ability to elicit broadly cross-neutralizing antibodies. Liver Int. 40, 1865–1871 (2020).
Gomez-Escobar, E. et al. Incorporation of apolipoprotein E into HBV-HCV subviral envelope particles to improve the hepatitis vaccine strategy. Sci. Rep. 11, 21856 (2021).
Czarnota, A. et al. Specific antibodies induced by immunization with hepatitis B virus-like particles carrying hepatitis C virus envelope glycoprotein 2 epitopes show differential neutralization efficiency. Vaccines 8, 294 (2020).
Garrone, P. et al. A prime-boost strategy using virus-like particles pseudotyped for HCV proteins triggers broadly neutralizing antibodies in macaques. Sci. Transl. Med. 3, 94ra71 (2011).
Sliepen, K. et al. Induction of cross-neutralizing antibodies by a permuted hepatitis C virus glycoprotein nanoparticle vaccine candidate. Nat. Commun. 13, 7271 (2022). This work shows that permuted HCV E1E2 glycoprotein nanoparticles elicit broadly neutralizing antibodies, presenting a promising approach for effective cross-genotype HCV vaccine development.
He, L. et al. Proof of concept for rational design of hepatitis C virus E2 core nanoparticle vaccines. Sci. Adv. 6, eaaz6225 (2020).
Forns, X. et al. Vaccination of chimpanzees with plasmid DNA encoding the hepatitis C virus (HCV) envelope E2 protein modified the infection after challenge with homologous monoclonal HCV. Hepatology 32, 618–625 (2000).
Weiland, O. et al. Therapeutic DNA vaccination using in vivo electroporation followed by standard of care therapy in patients with genotype 1 chronic hepatitis C. Mol. Ther. 21, 1796–1805 (2013).
Vargas, J. E. et al. Retroviral vectors and transposons for stable gene therapy: advances, current challenges and perspectives. J. Transl. Med. 14, 288 (2016).
Haga, Y. et al. Hepatitis C virus modified sE2(F442NYT) as an antigen in candidate vaccine facilitates human immune cell activation. J. Virol. 98, e0180923 (2024).
Swetha, K. et al. Recent advances in the lipid nanoparticle-mediated delivery of mRNA vaccines. Vaccines 11, 658 (2023).
Freyn, A. W. et al. A multi-targeting, nucleoside-modified mRNA influenza virus vaccine provides broad protection in mice. Mol. Ther. 28, 1569–1584 (2020).
Hasso-Agopsowicz, M. et al. Identifying WHO global priority endemic pathogens for vaccine research and development (R&D) using multi-criteria decision analysis (MCDA): an objective of the Immunization Agenda 2030. EBioMedicine 110, 105424 (2024).
Kong, L. et al. Structure of hepatitis C virus envelope glycoprotein E1 antigenic site 314-324 in complex with antibody IGH526. J. Mol. Biol. 427, 2617–2628 (2015).
Torrents de la Peña, A. et al. Structure of the hepatitis C virus E1E2 glycoprotein complex. Science 378, 263–269 (2022).
Kong, L. et al. Structural basis of hepatitis C virus neutralization by broadly neutralizing antibody HCV1. Proc. Natl Acad. Sci. USA 109, 9499–9504 (2012).
Kong, L. et al. Structure of hepatitis C virus envelope glycoprotein E2 antigenic site 412 to 423 in complex with antibody AP33. J. Virol. 86, 13085–13088 (2012).
Potter, J. A. et al. Toward a hepatitis C virus vaccine: the structural basis of hepatitis C virus neutralization by AP33, a broadly neutralizing antibody. J. Virol. 86, 12923–12932 (2012).
Li, Y. et al. Structural basis for penetration of the glycan shield of hepatitis C virus E2 glycoprotein by a broadly neutralizing human antibody. J. Biol. Chem. 290, 10117–10125 (2015).
Keck, Z. Y. et al. Antibody response to hypervariable region 1 interferes with broadly neutralizing antibodies to hepatitis C virus. J. Virol. 90, 3112–3122 (2016).
Gu, J. et al. Escape of hepatitis C virus from epitope I neutralization increases sensitivity of other neutralization epitopes. J. Virol. 92, e02066-17 (2018).
Krey, T. et al. Structural basis of HCV neutralization by human monoclonal antibodies resistant to viral neutralization escape. PLoS Pathog. 9, e1003364 (2013).
Keck, Z. Y. et al. Affinity maturation of a broadly neutralizing human monoclonal antibody that prevents acute hepatitis C virus infection in mice. Hepatology 64, 1922–1933 (2016).
Deng, L. et al. Structural evidence for a bifurcated mode of action in the antibody-mediated neutralization of hepatitis C virus. Proc. Natl Acad. Sci. USA 110, 7418–7422 (2013).
Deng, L. et al. Discrete conformations of epitope II on the hepatitis C virus E2 protein for antibody-mediated neutralization and nonneutralization. Proc. Natl Acad. Sci. USA 111, 10690–10695 (2014).
Vasiliauskaite, I. et al. Conformational flexibility in the immunoglobulin-like domain of the hepatitis C virus glycoprotein E2. mBio 8, e00382–17 (2017).
Sandomenico, A. et al. Generation and characterization of monoclonal antibodies against a cyclic variant of hepatitis C virus E2 epitope 412-422. J. Virol. 90, 3745–3759 (2016).
Pierce, B. G. et al. Structure-based design of hepatitis C virus vaccines that elicit neutralizing antibody responses to a conserved epitope. J. Virol. 91, e01032-17 (2017).
Leroux-Roels, G. et al. Immunogenicity and tolerability of intradermal administration of an HCV E1-based vaccine candidate in healthy volunteers and patients with resolved or ongoing chronic HCV infection. Hum. Vaccin. 1, 61–65 (2005).
Luo, S. et al. Adenoviruses vectored hepatitis C virus vaccine cocktails induce broadly specific immune responses against multi-genotypic HCV in mice. Biomed. Pharmacother. 170, 115901 (2024).
Kord, E. et al. BacMam virus-based surface display for HCV E2 glycoprotein induces strong cross-neutralizing antibodies and cellular immune responses in vaccinated mice. Infect. Agents Cancer 16, 69 (2021).
Ahlen, G. et al. In vivo clearance of hepatitis C virus nonstructural 3/4A-expressing hepatocytes by DNA vaccine-primed cytotoxic T lymphocytes. J. Infect. Dis. 192, 2112–2116 (2005).
Ahlen, G. et al. Long-term functional duration of immune responses to HCV NS3/4A induced by DNA vaccination. Gene Ther. 21, 739–750 (2014).
Ratnoglik, S. L. et al. Induction of cell-mediated immune responses in mice by DNA vaccines that express hepatitis C virus NS3 mutants lacking serine protease and NTPase/RNA helicase activities. PLoS ONE 9, e98877 (2014).
Beaumont, E. et al. Hepatitis C virus E1 and E2 proteins used as separate immunogens induce neutralizing antibodies with additive properties. PLoS ONE 11, e0151626 (2016).
Czarnota, A. et al. Immunogenicity of Leishmania-derived hepatitis B small surface antigen particles exposing highly conserved E2 epitope of hepatitis C virus. Microb. Cell Fact. 15, 62 (2016).
Christiansen, D. et al. Immunological responses following administration of a genotype 1a/1b/2/3a quadrivalent HCV VLP vaccine. Sci. Rep. 8, 6483 (2018).
Vijayamahantesh, V. et al. Modified E2 glycoprotein of hepatitis C virus enhances proinflammatory cytokines and protective immune response. J. Virol. 96, e0052322 (2022).
Patra, T. et al. Hepatitis C virus E1 and modified E2 delivered from an mRNA vaccine induces protective immunity. npj Vaccines 8, 42 (2023).
Koziel, M. J. Cellular immune responses against hepatitis C virus. Clin. Infect. Dis. 41, S25–S31 (2005).
Bankwitz, D. et al. Hepatitis C reference viruses highlight potent antibody responses and diverse viral functional interactions with neutralising antibodies. Gut 70, 1734–1745 (2021).
Salas, J. H. et al. An antigenically diverse, representative panel of envelope glycoproteins for hepatitis C virus vaccine development. Gastroenterology 162, 562–574 (2022).
Hamming, R. W. Error detecting and error correcting codes. Bell Syst. Tech. J. 29, 147–160 (1950).
Christie, J. M. et al. Immune selection and genetic sequence variation in core and envelope regions of hepatitis C virus. Hepatology 30, 1037–1044 (1999).
Lamonaca, V. et al. Conserved hepatitis C virus sequences are highly immunogenic for CD4+ T cells: implications for vaccine development. Hepatology 30, 1088–1098 (1999).
Gerlach, J. T. et al. Minimal T-cell-stimulatory sequences and spectrum of HLA restriction of immunodominant CD4+ T-cell epitopes within hepatitis C virus NS3 and NS4 proteins. J. Virol. 79, 12425–12433 (2005).
Klade, C. S. et al. Hepatitis C virus-specific T cell responses against conserved regions in recovered patients. Vaccine 27, 3099–3108 (2009).
Seigel, B. et al. Factors that determine the antiviral efficacy of HCV-specific CD8+ T cells ex vivo. Gastroenterology 144, 426–436 (2013).
Shoukry, N. H. et al. Conserved hierarchy of helper T cell responses in a chimpanzee during primary and secondary hepatitis C virus infections. J. Immunol. 172, 483–492 (2004).
Molero-Abraham, M. et al. Selection of conserved epitopes from hepatitis C virus for pan-populational stimulation of T-cell responses. Clin. Dev. Immunol. 2013, 601943 (2013).
Fitzmaurice, K. et al. Molecular footprints reveal the impact of the protective HLA-A*03 allele in hepatitis C virus infection. Gut 60, 1563–1571 (2011).
Shoukry, N. H., Cox, A. L. & Walker, C. M. Immunological monitoring in hepatitis C virus controlled human infection model. Clin. Infect. Dis. 77, S270–S275 (2023).
Castro, K. M. et al. Computational design of vaccine immunogens. Curr. Opin. Biotechnol. 78, 102821 (2022).
Rappuoli, R. et al. Reverse vaccinology 2.0: human immunology instructs vaccine antigen design. J. Exp. Med. 213, 469–481 (2016).
Byrne, P. O. & McLellan, J. S. Principles and practical applications of structure-based vaccine design. Curr. Opin. Immunol. 77, 102209 (2022).
Gaiha, G. D. et al. Structural topology defines protective CD8+ T cell epitopes in the HIV proteome. Science 364, 480–484 (2019).
Ramirez, S. & Bukh, J. Current status and future development of infectious cell-culture models for the major genotypes of hepatitis C virus: essential tools in testing of antivirals and emerging vaccine strategies. Antivir. Res. 158, 264–287 (2018).
Burm, R. et al. Animal models to study hepatitis C virus infection. Front. Immunol. 9, 1032 (2018).
Mercer, D. F. et al. Hepatitis C virus replication in mice with chimeric human livers. Nat. Med. 7, 927–933 (2001).
Ploss, A. & Kapoor, A. Animal models of hepatitis C virus infection. Cold Spring Harb. Perspect. Med. 10, a036970 (2020).
Liang, T. J. et al. Controlled human infection model — fast track to HCV vaccine? N. Engl. J. Med. 385, 1235–1240 (2021).