image:
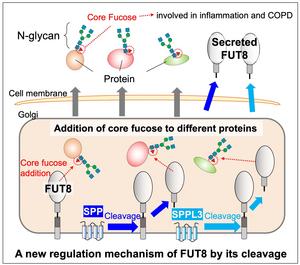
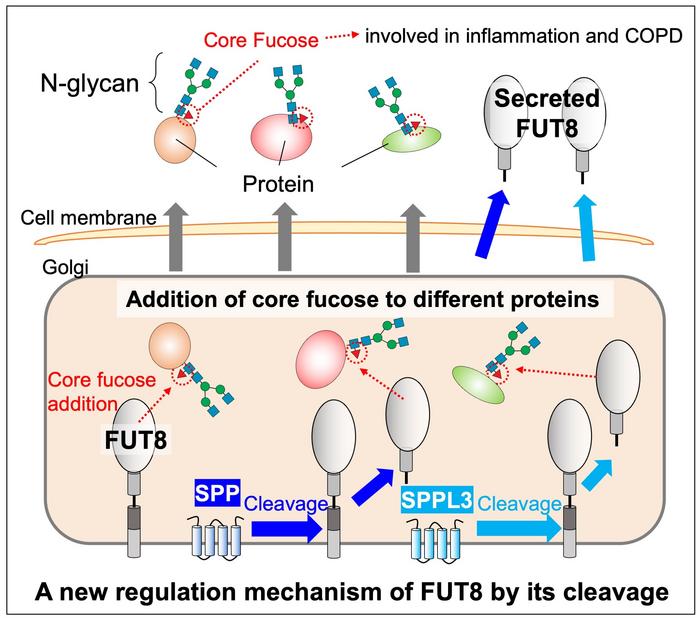
Secreted FUT8 proteins result from cleavages aided by the proteases SPP and SPPL3, and an unknown protease.
Credit: Yasuhiko Kizuka, Institute for Glyco-core Research (iGCORE), Gifu University
Deep in our cells, a wide range of processes are occurring constantly. These cellular processes rely on enzymes to act as catalysts and set off a series of molecular interactions. There are still many processes within the body that are not fully understood. Discovering exactly how these cellular pathways work can help researchers better understand how some diseases proliferate and develop new treatments that target part of these processes.
An important part of the body’s immune response is core fucosylation, which produces the building blocks of the molecules that make up the immune system. Core fucosylation relies on glycosylation, which is a reaction between glycans (carbohydrates) and a protein or lipid. Glycosylation relies on glycotransferases or glycosylation enzymes. The enzyme responsible for the glycosylation process is called FUT8, but the pathway regulating FUT8 has not been fully discovered and documented.
New research published in Journal of Biological Chemistry on January 28 reveals how FUT8 is regulated by the proteases SPP and SPPL3 for this important physiological process, while uncovering new mysteries in the process that are still to be solved. “The details and mechanisms governing how glycosyltransferase (glycosylation enzyme) is regulated in cells remains unclear. In our research, we focused on secretion of one of the physiologically important glycosyltransferases, called FUT8,” said Yasuhiko Kizuka, a researcher at the Institute for Glyco-core Research (iGCORE) at Gifu University in Gifu, Japan.
When FUT8 is deactivated in mice models, most of the mice do not grow properly, have impaired immune function, and behavioral problems. FUT8 has also been identified as a potential target for some types of cancer, including metastatic melanoma. FUT8 can lead to cancer cell growth by increasing the core fucosylation of cell receptors like EGFR that promote cell growth. Fully understanding FUT8 regulation is an important step toward exploring FUT8 as a potential treatment target.
Researchers started by looking at proteases, which are enzymes that help break down proteins. This is called cleavage. Proteases like SPP and SPPL3 are involved in the cleavage of other glycosyltransferases. When researchers intentionally blocked SPPL3, immune cell interactions were altered, which could point to an important interaction between SPPL3 and other proteases in its family and FUT8.
Researchers attempted to recreate the molecular mechanism of FUT8 secretion in the lab. They found that SPP and SPPL3 are involved in the first cleavage of FUT8, but another unknown protease is involved in the second cleavage. When SPP and SPPL3 were blocked, FUT8 substrate specificity was changed, pointing to the important role SPP and SPPL3 proteases play. “Substrate specificity, which is selectivity toward glycoproteins, is altered during cleavage. So, secretion of glycosyltransferase is not just throwing away junk or excess enzymes but a regulation process for controlling glycosylation,” said Kizuka.
Researchers are still unsure what this second protease is, but they do know some of its characteristics. The protease is most likely found in the late Golgi or post-Golgi compartments of the cell. Eventually, they hope to identify this protease, which would extend their understanding of this cellular process.
In addition to melanoma, FUT8 has been linked to cancer progression in some types of liver cancer, kidney cancer, non-small cell lung cancer, pancreatic cancer, colorectal cancer, stomach cancer, osteosarcoma, and cervical cancer. Understanding FUT8 regulation could be key to unlocking new treatments that target this cellular pathway.
Looking ahead, researchers hope to identify a new protease that cleaves glycosyltransferases. “Right now, SPPL3 is best characterized as a primary protease for glycosyltransferase cleavage, but not all glycosyltransferases are cleaved by SPPL3. I would like to look for other proteases and learn how secretion of glycosyltransferases is regulated,” said Kizuka.
Other researchers include Seita Tomida, Yuko Tokoro, and Takahiro Yamasaki of Gifu University; and Rebeca Kawahara and Kristina Mae Bienes of Nagoya University.
The Japan Science and Technology Agency, a Grant-in-Aid for Scientific Research, the Japan Society for the Promotion of Science, the Japan Agency for Medical Research and Development, the Ministry of Education, Culture, Sports, Science, and Technology, and a Grant-in-Aid for JSPS Fellows supported this research.
Journal
Journal of Biological Chemistry
Method of Research
Experimental study
Subject of Research
Cells
Article Title
Signal peptide peptidase– and SPP-like 3–dependent shedding of α1,6-fucosyltransferase differentially affects core fucosylation
Article Publication Date
28-Jan-2026
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of news releases posted to EurekAlert! by contributing institutions or for the use of any information through the EurekAlert system.