Ethical regulations
All research conformed to all pertinent ethical stipulations. The luciferin-expressing human GBM stem-like cells (GSCs) GBM#01 were derived from a GBM surgical specimen in the Department of Neurosurgery, Qilu Hospital, whose relevant studies were approved by the Research Ethics Committee of Shandong University and the Ethics Committee of Qilu Hospital (Shandong, China). The experiments complied with relevant guidelines and regulations. Consent forms were signed by the patients after they had been informed (permit nos. SDULCLL2021-1-17). All in vivo experiments with Animals in this study were carried out in strict accordance with the ARRIVE guidelines 2.0 and the Guidelines for the Care and Use of Laboratory Animals Ethics Committee of Nanjing University of Chinese Medicine (NO.202310A053 and NO.202411A063). The maximal tumor burden was strictly limited to 1.2 cm in diameter for mice, with a total tumor weight not exceeding 10% of the mice body weight. No animals exceeded these limits during the study.
Materials
Anhui Guoping Pharmaceutical Co. Ltd. synthesized the D4F peptide (Ac-FAEKFKEAVKDYFAKFWD, a biomimetic peptide of apolipoprotein A-I), the α-Mel peptide (DWFKAFYDKVAEKFKEAF-GSG-GIGAVLKVLTTGLPALISWIKRKRQQ-NH2), the α-Mel-OVA peptide (DWFKAFYDKVAEKFKEAF-GSG-GIGAVLKVLTTGLPALISWIKRKRQQ-GSG-SIINFEKL-NH2), the α-Mel-M25 peptide (DWFKAFYDKVAEKFKEAF-GSG-GIGAVLKVLTTGLPALISWIKRKRQQ-GSG-KDYTAAGFSSFQKLRLDLTSMQIITTD-NH2), and the α-Mel-mEpb4 (DWFKAFYDKVAEKFKEAF-GSG-GIGAVLKVLTTGLPALISWIKRKRQQ-GSG-ELEQFESTIGFKLPNLRAAKRLWK-NH2) with a purity of 90%. Sigma-Aldrich provided manganese dichloride (MnCl2·4H2O) and NaOH. Yuanye Bio-Technology Co. Ltd (Shanghai) sold BSA and ginsenoside Rh2. Gibco Co., Ltd. (USA) provided PBS (pH 7.4), FBS, high-glucose DMEM, and RPMI-1640. Beyotime Biotechnology (Shanghai, China) provided FITC, DMSO, L-glutamine, DiD, Hoechst33342, anti-fluorescence quencher (including DAPI), and BCA Protein Assay Kit. ELISA Kits were acquired from Elabscience Biotechnology Co, Ltd. Anti-CD11c-FITC (E-AB-F0991C), Anti-mouse CD86-APC (E-AB-F0994E), Anti-CD80-PE/Cyanine7 (E-AB-F0992H), Anti-CD40-APC (E-AB-F1028E), Anti-CD3-PE (E-AB-F1013D), Anti-CD4-PE/Cyanine7 (E-AB-F1097H), Anti-CD8a-FITC (E-AB-F1104C), Anti-F4/80-APC (E-AB-F0995E), Anti-CD86-PE/Cyanine7 (E-AB-F0994H), Anti-CD206-FITC (E-AB-F1135C), Anti-IFN-γ-APC (E-AB-F1101E), Anti-CD3-APC (E-AB-F1013E), Anti-CD44-PE (E-AB-F1100D), Anti-CD62L-PE/Cyanine7 (E-AB-F1011H), Anti-Foxp3-PE (E-AB-F1238D), Anti-CD16/32 (E-AB-F0997A), and Anti-CD107a-PE/Cyanine7 (E-AB-F1254H) for mice, and Anti-CD45-FITC (E-AB-F1137C), Anti-CD3-PE (E-AB-F1001D), Anti-CD8a-PE/Cyanine7 (E-AB-F1110H), Anti-CD4-APC (E-AB-F1109E), and Anti-IFN-γ-APC (E-AB-F1196E) for humans were purchased from Elabscience. Anti-IL-4-APC (504105), Anti-CD63-APC (143905), Anti-CD81-PE (104905), and Anti-CD9-APC (124811) for mice were purchased from BioLegend.
Cell lines and primary cells
GL261-luc, GL261-OVA-luc, and LLC-OVA cell lines were acquired from Zhenjiang Vigen Biotech Company Limited. GL261, CT-2A, LLC, 4T1, BCEC, and PC12 cell lines were acquired from Shanghai Cell Bank. Cells were characterized before use by short tandem repeat DNA fingerprinting (STR)-PCR DNA profiling and were determined to be free of mycoplasma contamination. GL261, GL261-OVA-luc, GL261-luc, BCECs, PC12, and LLC cells were grown in DMEM medium (KeyGEN) supplemented with 10% FBS (Vazyme). 4T1 cells were grown in RPMI-1640 medium (KeyGEN) supplemented with 10% FBS (Vazyme). GL261-luc cells were chosen by applying 10 μg/mL puromycin (Gibco). BMDCs were cultured in RPMI-1640 containing 20% FBS, GM-CSF (MedChem Express, 10 ng mL-1), and IL-4 (MedChem Express, 10 ng mL-1). The luciferase-expressing human GBM stem-like cells (GSCs) GBM#01 were cultured in serum-free Neurobasal™ medium (Gibco, 21103049) supplemented with B-27™ Neuro Mix (Thermo Fisher, 17504044), epidermal growth factor (Thermo Fisher, PHG0314), and basic fibroblast growth factor (PeproTech, 100-18B). Tumorspheres were split using Accutase™ (Thermo Fisher, A1110501) to expand GSCs. All cells were cultured in a humidified atmosphere containing 5% CO2 at 37 °C.
Animal models
Female mice of C57BL/6 strain (18–22 g, 6–8 weeks) and Balb/c strain (18–22 g, 6–8 weeks), and Male SD rats (180–220 g, 6–8 weeks) were sourced from Qinglongshan Animal Breeding Field (Nanjing, China). Female NOG mice (18–22 g, 6–8 weeks) were obtained from Weitong Lihua Experimental Animal Center (China). The cerebral cortex was excised at the right cerebral hemisphere (−1.0 mm, 3.0 mm) of the rats (strain SD), and the drug-loaded hydrogel was injected intracavely. For brain tumor modeling, the cell suspension was injected into the right cerebral hemisphere of mice (strain C57BL/6) at a depth of 2 mm (−0.6 mm, 1.8 mm). For breast cancer modeling, the cell suspension was inoculated into the mammary fat pad of mice (strain Balb/c). Animals with their wound sterilized and seamed were carefully housed in a specific pathogen-free (SPF) environment. The temperature of the animal room lies between 20 and 26 °C, the warm humidity is within 40 to 70%, light and darkness alternate for 12 h, and all mice can freely obtain food and water.
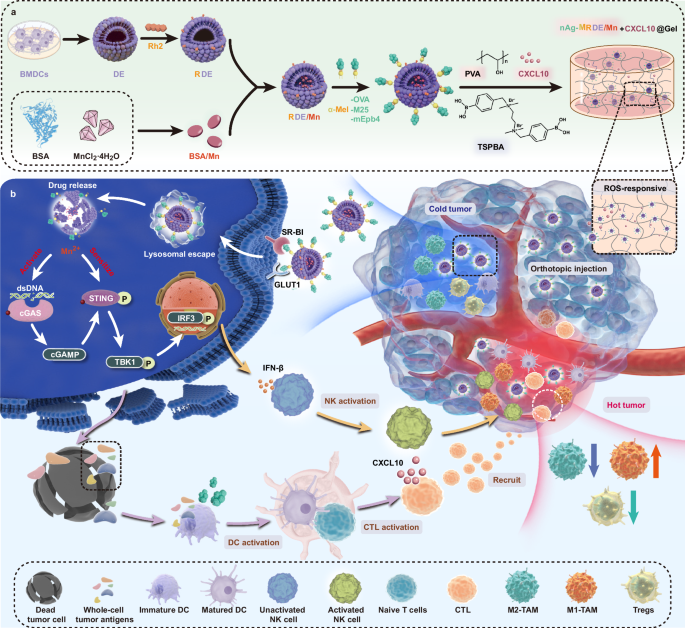
Preparation of nAg-MRDE/Mn
Serum-free medium for exosome isolation was obtained by centrifugation, and DEs were isolated from medium using ultracentrifugation techniques. BSA (10 mg mL−1) and MnCl2 (15 mg mL−1) were mixed and stirred, followed by adding aqueous solutions of NaOH (40 mg mL−1) to a pH of 11. After incubation at 37 °C for 2 h, free Mn2+ was removed by 30 kDa cutoff centrifugal filter device (Amicon). BSA was mixed with different concentrations of Mn2+ and the fluorescence spectra were scanned in the range of 290-450 nm using an excitation wavelength of 280 nm, and Gen5 (v2.0) was used for data collection.
Hybrid peptides were monitored by LC-ESI-MS/MS analysis, and Xcalibur (v4.0) and Masshunter Workstation Data Acquisition software (vB.08.00) were used for data collection. The Rh2 (1.5 mg) dissolved in DMSO was added to the DEs (400 μg mL−1). It was then incubated at 37 °C for 1 h, RDE was prepared. Following electroporation, BSA/Mn solution (1 mL) was added dropwise and incubated for an additional h, RDE/Mn was prepared. Subsequently, the α-Mel-nAg PBS solution (1 mg mL−1, 0.5 mL) was added dropwise and stirred magnetically for 1 h. Afterwards, the resultant blend was centrifuged employing an 30 kDa ultrafiltration tube to eliminate impurities. Then, PBS was added to achieve a final volume of 1 mL, nAg-MRDE/Mn was prepared.
Characterization of O-MRDE/Mn
The OVA-Mel-D4F was labeled with FITC and O-MRDE/Mn was prepared. Ultrafiltration was used to separate the unbound proteins and other impurities, the fluorescence intensity was measured using a microplate reader (PerkinElmer) and the loading efficiency of the hybridized peptides was calculated. The loading rate of Rh2 was determined by HPLC (Thermo) after O-MRDE/Mn was treated with methanol. The chromatographic conditions were as follows: mobile phase: acetonitrile: water = 60:40, column: BDS HYPERSIL C18 (250 mm × 4.6 mm, 5 μm), detection wavelength: 203 nm, column temperature: 30 °C. The encapsulation rate of Mn was determined by ICP-MS (PerkinElmer).
The typical morphology and structure of BSA/Mn, DEs, and O-MRDE/Mn were observed using the Hitachi HT7800 transmission electron microscope (Hitachi, Japan). The expression levels of CD81, CD63, and CD9 in DEs and O-MRDE/Mn were analyzed by flow cytometry. The Malvern Zetasizer Nano-ZS90 device (Malvern Zetasizer) was utilized to measure the particle size and zeta potential, and Malvern zetasizer (v7.13) was used for data collection. In order to examine the placement stability of O-MRDE/Mn, the aforementioned preparations were shielded from light and kept at a temperature of 4 °C. Particle size, zeta potential and PDI were conducted daily.
O-MRDE/Mn and MnCl2 were placed in a dialysis bag and agitated in phosphate buffer (pH 7.4) at 37 °C. Samples were taken and replenished with equal amounts of freshly released media at different time points, detected using an ICP-MS instrument, and the cumulative release rate calculated. Syngistix (v3.3) was used for data collection.
For the hemolysis assays, various concentrations of Mel, α-Mel, α-Mel-OVA, and O-MRDE/Mn were prepared. Plasma was eliminated from mice blood through centrifugation, followed by the adjustment of blood cell concentration to 2%. Subsequently, the blood was incubated with the drug for a duration of 3 h at a temperature of 37 °C. The absorbance of the supernatant after centrifugation at 540 nm was measured.
Preparation of O-MRDE/Mn + CXCL10@Gel
A clear solution was obtained by mixing PVA (72 kDa; 98% hydrolyzed; 5 g) and deionized water (100 ml), and stirring them at 90 °C. O-MRDE/Mn and CXCL10 were introduced into the PVA water-based solution. Hydrogel was formed by mixing TSPBA (5 wt% in H2O, 2 ml) and PVA (5 wt% in H2O, 2 ml) together.
Characterization of O-MRDE/Mn + CXCL10@Gel
1H NMR (Varian Unity Inova 500, Palo Alto) was used to examine TSPBA, and MestReNova Mnova (v15.0) was used for data collection. Teneo SEM (Thermo Fisher) was used to acquire SEM images. Rheology experiments were performed using the Anton Paar MCR 302 rheometer (Anton Paar), and Rheocompass (v1.22) was used for data collection. Take 6 data points of each order of magnitude for analysis during amplitude sweeping. The strain of the shear recovery test was selected as 1% and 500% and maintained for 15 s and 10 s, respectively. The cycle was repeated twice, and one data point was taken every second.
ROS responsiveness of O-MRDE/Mn + CXCL10@Gel
To detect ROS-responsive degradation of hydrogels in vitro, two gels of identical size were subjected to PBS and PBS + 1 mM H2O2. The visual appearance of the gels was recorded daily for 9 days, and the weight loss was measured to calculate the degradation rate. The release of CXCL10 and Mn from O-MRDE/Mn+CXCL10@Gel was studied using a dialysis bag at room temperature in PBS and PBS + 1 mM H2O2. 200Μl of hydrogel was taken for the experiment, 0.5 mL of release PBS was taken daily for eight days, and fresh PBS was added. The particle size of O-MRDE/Mn before and after hydrogel degradation was measured with NS300 (NanoSight), and NTA software (v3.4.4) was used for data collection. DIR dye (MCE) was used to label the DEs, and DIR-O-MRDE/Mn+CXCL10@Gel was prepared. It was implanted into the brains of healthy mice and post-tumor-surgery mice, respectively, and the fluorescence signal intensity of the hydrogel in the mouse brain was observed by using a small-animal in vivo imaging system, and quantitative analyses were carried out to examine the responsive degradation properties of the hydrogel. To detect ROS-responsive degradation of hydrogels in vivo, part of the cortex on the right side of SD rats was removed under the microscope to mimic the surgical wound. O-MRDE/Mn+CXCL10@Gel (120 μL) was injected. Every two days, three rats were euthanized. The hydrogel was weighed, and the degradation rate was calculated, then gently homogenized. ELISA (Elabscience) was used to determine the release of CXCL10, while ICP-MS (PerkinElmer) was employed to determine the presence of Mn.
Recruitment of CD8+ T cells in vitro
T cells were placed in the upper compartment of a Transwell, while the lower compartment received different concentrations of the drug. Following a 24 h incubation, the cells from the lower chamber were collected, and the chemotaxis index was calculated. After incubating of O-MRDE + CXCL10 (5 ng mL-1) for 6, 12, 24, and 36 h, the cells from the lower chamber were collected, and the chemotaxis index was calculated.
Cell uptake studies
Different preparations were prepared using FITC-labeled BSA/Mn (BSA/MnFITC). The administration groups were categorized as DE/MnFITC, D4F-DE/MnFITC, MDE/MnFITC, MDE(-)/MnFITC, Rh2-DE/MnFITC, Rh2-DE(-)/MnFITC, and O-MRDE/MnFITC. Where MDE(-)/MnFITC and Rh2-DE(-)/MnFITC denote the pre-treatment of GL261 cells with D4F and WZB 117 (MedChemExpress), respectively. After 2 h of treatment with the different preparations, the nuclei were visualized by Hoechst staining, and the images were captured using a fluorescence microimaging system (Keyence), and BZ-X800 Wide Image Viewer (v1.0) was used for data collection. In addition, DMSO was introduced to lyse the cells and the intracellular fluorescence intensity was measured using a microplate reader. (Fitc: λEx/λEm = 488/525 nm)
Confocal imaging analyses on the endo-lysosomal escape
Dye D4F-DE/Mn, MDE/Mn, and O-MRDE/Mn with DiD. The GL261 cells were cultured on cell climbing slices in 12-well plates. The GL261 cells underwent treatment with D4F-DE/Mn, MDE/Mn, and O-MRDE/Mn for 0.5, 2, and 8 h respectively. Staining with 75 nM LysoTracker Green (Meilun Biotechnology) and DAPI (Beyotime) was performed. The Leica TCS-SP8 (Leica Microsystems) was utilized for the examination, and Leica Application Suite (v3.6) was used for data collection.
Evaluation of in vitro safety
Cell viability was evaluated with the CCK8 assay Kit. In short, GL261, BCEC, BMDCs, and PC12 cells were placed in 96-well plates. The medium was then substituted with either serum-free medium, serum-free medium containing gel, or O-MRDE/Mn+CXCL10@Gel. The absorbance was recorded at 450 nm.
Detection of ICD biomarkers
GL261 cells were treated with different formulations, namely BSA/Mn, DE/Mn, MDE/Mn, and O-MRDE/Mn. The cells were labeled with anti-mouse CRT antibody (Bioss) at 4 °C for 30 min and Alexa Fluor 568 Donkey Anti-Rabbit IgG secondary antibody (Proteintech) at 4 °C for 30 min. The HMGB1 and ATP Chemiluminescence Assay Kit (Elabscience) were used to analyze the content of HMGB1 and ATP released by GL261 cells after different treatments.
Mechanisms of specific immune activation
To assess antigen presentation, O-MRDE/Mn and physical mixtures (pure physical mixing of each component) were co-incubated with BMDCs separately, and the cells were fixed, closed and labeled by addition of APC anti-mouse H-2Kb bound to SIINFEKL Antibody. Nuclei were visualized by Hoechst staining and images were captured using a fluorescence microimaging system (Keyence).
GL261 cells, in 24-well plates, were treated with various formulations, namely BSA/Mn, DE/Mn, MDE/Mn, and O-MRDE/Mn. Afterwards, cancer cells and supernatant were gathered and co-cultured with BMDCs in 24-well dishes. Cells were subjected to flow cytometer analysis after being incubated with anti-CD11c, anti-CD40, anti-CD80, anti-CD86, and H-2Kb-restricted SIINFEKL. After a 36 h incubation, BMDCs can be gathered and introduced into the 24-well plate of T cells (1: 10). The cells were stained with anti-CD3, anti-CD4, and anti-CD8, and H-2Kb OVA Tetramer-SIINFEKL. The supernatant obtained was utilized for ELISA experiments to measure TNF-α, IL-6, IL-12, IFN-γ, and IL-10 (Elabscience).
Activation of the innate immune system mechanisms
GL261 cells, in 24-well plates, were treated with various formulations, and The medium was gathered to evaluate IFN-β using ELISA Kit (Elabscience). Simultaneously, cells were harvested to measure cGAMP levels using the cGAMP ELISA Kit (Elabscience).
Antitumor study in orthotopic GBM model
3 × 105 GL261-luc cells and GL261-OVA-luc cells were injected into the right striatum of C57BL/6 mice. The pre-treatment was given O-MRDE/Mn on day 5, while the other group was dosed on day 10. Both groups were orthotopically injected at the surgical cavity of mice (0.4 μL g-1). Randomly selected brains from each group were removed after administration for H&E staining, and Slide Viewer (v2.6) was used for data collection. The weight of mice was measured, and GBM growth was monitored by IVIS imaging system (PerkinElmer), and Living Image (v4.4) used for data collection. Mouse survival was tracked to obtain Kaplan-Meier survival curves. The biochemical assays were conducted using the serum collected on day 11 post-administration. Major organs were harvested for H&E staining. Serum levels of cytokines and antibodies were determined according to ELISA Kit instructions (Elabscience). 8 days after administration, samples were treated with anti-CD16/32, followed by staining with anti-CD3, anti-NK1.1, anti-CD11c, anti-MHC-I, anti-CD4, anti-CD8, anti-IFN-γ, and anti-GrB. Kaluza (v2.2.1) was used for data collection.
Activation of antigen-specific immunity and reprogramming of the TME in orthotopic GBM models
To examine antigen-specific responses, 3 × 105 GL261-OVA cells were administered into the right striatum of C57BL/6 mice. Each group was dosed on day 10. Mice were euthanized 8 days after administration. Flow samples were treated with anti-CD16/32, followed by staining with anti-CD11c, anti-CD40, anti-CD80, anti-CD86, H-2Kb-restricted SIINFEKL, anti-CD3, anti-CD4, anti-CD8, H-2Kb OVA Tetramer-SIINFEKL, anti-IFN-γ, anti-GrB, and anti-CD107a.
After orthotopic GBM models were euthanized, tumor tissues were collected and treated with anti-CD16/32, followed by staining with anti-F4/80, anti-CD86, and anti-CD206, anti-CD3, anti-CD8, anti-CD4, anti-IFN-γ, anti-IL-4, and anti-Foxp3. The brain was stained with Ki67, CD31, CD44, CD3, CD8, CD4, GATA-3, T-bet, and Foxp3 antibodies, respectively, and DAPI.
Antitumor study in postoperative GBM model and GBM rechallenge model
Ten days after inoculation, mice carrying in situ GL261-luc or GL261-OVA-luc cells were operated on and administered. According to the change in bioluminescence intensity, whether the tumor is effectively removed can be judged. Subsequently, the mice were treated with saline, blank gel, BSA/Mn@Gel, DE/Mn@Gel, MDE/Mn@Gel, O-MRDE/Mn@Gel, and O-MRDE/Mn+CXCL10@Gel. The weight of mice was measured, and GBM growth was monitored by the IVIS imaging system. Mouse survival was tracked to obtain Kaplan-Meier survival curves. Mice were sacrificed 10 days after administration. Serum levels of cytokines and antibodies were determined according to the ELISA Kit (Elabscience) instructions. Immunofluorescence staining of Ki67, CD8, and Foxp3 was performed on sections of the brain. Flow samples were treated with anti-CD16/32, followed by staining with anti-CD3, anti-NK1.1, anti-CD11c, anti-CD80, anti-CD86, anti-F4/80, anti-CD206, anti-CD3, anti-CD8, anti-CD4, anti-CD44, and anti-CD62L. After 60 days of administration, the surviving mice of the O-MRDE/Mn+CXCL10@Gel were injected with 3 × 105 GL261 or GL261-OVA cells at the left striatum. Blank mice of the same week age were injected with GL261 (3 × 105) at the right and left striatum. Mouse survival was tracked to obtain Kaplan-Meier survival curves. Ten days after establishing the GBM rechallenge model, sections were labeled with H&E and Ki67. Flow cytometry was used for CD8+ T cells in the spleen and tumor.
Antitumor study in lung cancer brain metastasis model
1 × 106 LLC cells were administered into the right striatum of C57BL/6 mice. Mouse survival was tracked to obtain Kaplan-Meier survival curves. After 8 days of administration, blood was collected by removing the eyeball after mice were anesthetized with isoflurane. Serum levels of cytokines and antibodies were determined according to the ELISA Kit instructions (Elabscience). Treated with anti-CD16/32, flow samples of tumors were followed by staining with anti-CD11c, anti-CD80, anti-CD86, H-2Kb-restricted SIINFEKL, anti-CD3, anti-CD4, and anti-CD8. Flow samples of spleens were followed by staining with anti-CD3 and anti-CD8. Flow samples of peripheral blood were followed by staining with anti-CD3, anti-CD8, H-2Kb OVA Tetramer-SIINFEKL. Brain tissues were removed 8 days after administration for H&E and immunofluorescence staining of CD4, CD8, and Tregs.
Antitumor study of M25-MRDE/Mn + CXCL10@Gel in combination with αPD-1 in the orthotopic 4T1 models
5 × 105 4T1 cells were administered to the right mammary fat pad of BALB/c mice. αPD-1 injection: 20 mg kg-1 intraperitoneally. Tumor size and weight were measured. Mouse survival was tracked to obtain Kaplan-Meier survival curves. Tumor samples were obtained 20 days post inoculation with 4T1 cells. The tumor cells were labeled with anti-mouse CRT antibody (Proteintech) at 4 °C for 30 min and Alexa Fluor 568 Donkey Anti-Rabbit IgG secondary antibody (Proteintech) at 4 °C for 30 min. Flow samples were first processed with anti-CD16/32, and then labeled with anti-CD3, anti-CD49b, anti-CD80, anti-CD86, anti-CD8, anti-IFNγ, anti-GrB, anti-CD4, anti-Foxp3. Then, the dyed cells were quantified using a flow cytometer (Beckman). The lungs were immobilized with Bouin solution (G-CLONE) for 4 h, then white metastatic nodules were counted, and finally H&E staining was conducted.
Anti-tumor studies in CT-2A and PDX model
To further evaluate the translational feasibility, 3 × 105 CT-2A cells were administered into the right striatum of C57BL/6 mice. Flow samples were treated with anti-CD16/32, followed by stained with anti-CD3, anti-NK1.1, anti-CD11c, anti-CD80, anti-CD86, anti-CD8, anti-PD-1, anti-IFN-γ, anti-GrB, anti-F4/80, anti-CD86, and anti-CD206, anti-CD4, and anti-Foxp3.
To further evaluate the antitumor efficacy against PDX model mice, we established an orthotopic humanized mouse model by intracranial injection of luciferase-expressing human GSCs GBM#01 into NOG mice. In brief, 0.2 mL (1 × 106 peripheral blood mononuclear cells (PBMC)) of cell suspension was administered via the tail vein in each mouse. Human Peripheral Blood CD14+ Cells were obtained from IPHASE Biotechnology (Suzhou). To obtain PBMV-derived DEs, PBMC were cultured in medium containing 100 ng mL-1 human GM-CSF (PeproTech) and 20 ng mL-1 human IL-4 (InvivoGen) for 48 h, and DCS were harvested by culturing in medium containing 10 ng mL-1 TNF-α (InvivoGen) for 3 days and further cultured to obtain DEs. 3 weeks later, GBM#01 cells were intracranially implanted. On day 4, mice injected with or without PBMC were euthanized. Successful construction of the PDX model was checked by immunofluorescence staining (CD45, Servicebio, GB300644-H) and flow cytometric analyses. The progression of the tumor was observed using IVIS imaging, and the survival was recorded. To evaluate the degree of activation in the immune system, spleens and tumors were isolated and processed. CD4+ T cells in spleen were staining with anti-CD45, anti-CD3, anti-CD4, and IFN-γ+ T cells in tumor were staining with anti-CD45, anti-CD3, anti-CD8, anti-IFN-γ. The Animal Ethics Committee of Nanjing University of Chinese Medicine granted approval for the experiments (permit NO.202411A063).
Statistics and reproducibility
We used a variety of tumor cell lines and mouse breeds for experimental design. FlowJo (v10.9.0) was used to analyze the obtained flow cytometry data. Graphpad Prism (v9.0.0) was used for statistical analysis throughout this study. One-way analysis of variance was used for comparison among multiple groups. P < 0.05 was considered statistically significant. The values and scatter in the bar graphs represent the mean ± SD and the number of individual replicates, respectively. Experiments performed in biological replicates are provided in the corresponding legends. No statistical method was used to predetermine sample size. No data were excluded from the analyses. The experiments were not randomized. The Investigators were not blinded to allocation during experiments and outcome assessment.
Figures and artwork
Graphic elements in Fig. 1a, b, Fig. 2a, f, Fig. 3a, n, Fig. 5f, and Fig. 7g were created using ChemDraw Professional (v16.0), 3ds Max (v2022) and Adobe Illustrator (v2021).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.