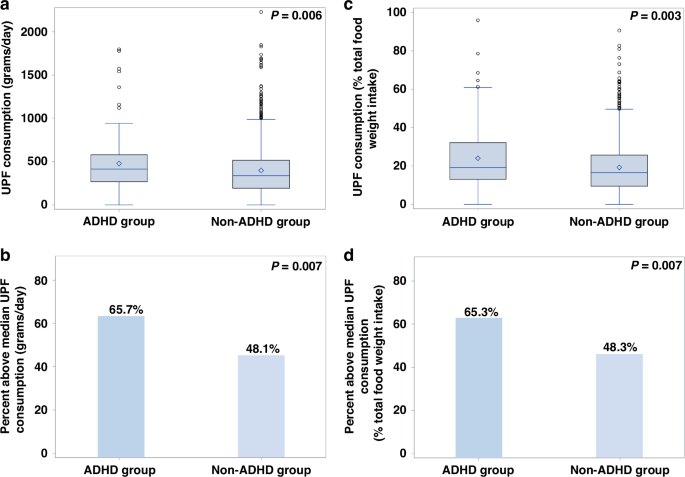
Main findings
In this nationally representative sample of Israeli children, ADHD was associated with higher UPF intake when measured both as daily weight (grams/day) and as the percentage of total daily food weight. The associations persisted in analyses using continuous UPF variables and were largely consistent in the sensitivity analysis, supporting the robustness of the findings. No differences in UPF consumption were observed by stimulant treatment status among children with ADHD. To the best of our knowledge, this is the first study to examine the association between physician-diagnosed ADHD and UPF consumption in children, a contribution of particular importance given the high prevalence and significant implications of both.
Notably, the magnitude of the association was modest. In fully adjusted models, ADHD was associated with roughly 1.6-fold higher odds of high UPF intake and an average increase of about 57 grams of UPF per day. However, even a modest increase in UPF consumption could be meaningful given the prolonged duration of exposure across the lifespan, and the broad range of detrimental health outcomes associated with these products25.
ADHD prevalence and group characteristics
ADHD prevalence in the current study (10.5%) is higher than global estimates. An umbrella review of meta-analyses by Ayano et al. reported a worldwide prevalence of 8.0% (95% CI: 6.0–10.0%)48. Nevertheless, Israeli studies have consistently shown higher rates across a range of ages. Cohen et al., based on medical records of children aged 6–13 in Israel’s second-largest health fund, reported a prevalence of 12.3%, which increased with age (3.7% at age 6; 16% at age 11; 17.1% at age 13)49. Pinhas-Hamiel et al. reported a high prevalence of ADHD among Israeli adolescents (20.1%), based on a nationwide analysis of medical records50, further suggesting elevated rates in Israel compared with global estimates.
Other characteristics of the ADHD group in this study were a higher proportion of boys, in line with the existing literature51, and no significant difference in SES compared with the non-ADHD group. This lack of difference may reflect a counterbalance between evidence linking ADHD to lower SES52 and, conversely, greater awareness and access to diagnosis in higher SES groups53. In addition, no differences were observed between the ADHD group and the comparison group in overall macronutrient intake. Nevertheless, children with ADHD demonstrated a lower dietary fiber intake, a finding that may indicate poorer dietary quality. Additionally, a higher intake of alpha-linolenic acid was observed in the ADHD group, which may reflect greater awareness of its documented association with symptom regulation54. Both groups’ macro- and micronutrient intakes were within the acceptable distribution ranges according to the Dietary Reference Intakes55.
To note, no differences were observed between the ADHD and comparison group in either total daily caloric intake from UPF or the percentage of UPF calories relative to overall energy intake. However, a significant difference was noted in UPF consumption when measured by food weight. This apparent discrepancy may be explained by the fact that not all UPFs are calorically dense; rather, they vary significantly in energy density. Some formulations contain ingredients with little or no caloric contribution, such as artificial colorants, preservatives, or non-nutritive sweeteners. In addition, many ultra-processed products are specifically manufactured to be low-calorie or marketed as “light” alternatives, including artificially sweetened beverages (which add substantial weight due to liquid content), sugar-free gelatin desserts, and low-fat snack products13,41. These items can increase the overall weight of UPF consumed without proportionally raising caloric intake.
At the same time, a considerable proportion of commonly consumed UPF, such as sweet baked goods, confectionery, fried snacks, and certain breakfast cereals, are highly energy-dense and contribute disproportionately to total caloric intake despite their relatively low physical weight. When such items coexist in the diet alongside energy-dilute UPF, population-level UPF intake may appear high when expressed as calories yet comparatively lower when expressed as grams (and vice versa). This heterogeneity in energy density provides a plausible explanation for the divergence between calorie-based and weight-based UPF measures observed in our study: it suggests that the greater weight of UPF consumed by the ADHD group may be primarily driven by a higher intake of low-energy-density products.
Dietary and nutritional aspects of ADHD
Prior literature linked ADHD to eating disorders and disordered eating. A meta-analysis of 12 studies (ADHD n = 4013; controls n = 29,404) by Nazar et al. found markedly higher odds of eating disorders among individuals with ADHD (pooled OR ≈ 3.8)4. A systematic review by Kaisari et al. concluded that there is moderate strength of evidence for a positive association between ADHD and disordered eating, particularly overeating5. A nationally representative school-based study in Israel (Namimi-Halevi et al.) found that adolescents with ADHD had significantly elevated odds of disordered eating, defined via a positive SCOFF screener: 50.2% versus 43.9% in non-ADHD peers (adjusted OR = 1.41; 95% CI: 1.17–1.70)56.
Evidence also indicates that ADHD is associated with variations in dietary intake and nutritional patterns, generally characterized as less healthy compared with peers without the diagnosis. Rojo Marticella et al. conducted a study across preschool and elementary populations (n = 734) and identified that the ADHD group was negatively associated with a healthy dietary pattern and positively associated with a Western-like diet6. A case–control study (n = 390) by Akin et al. found that children with ADHD consumed more processed meat products, milk-based desserts, and chocolate sweets than controls26. Wolff et al.; n = 1187) demonstrated that children and adolescents with ADHD reported more frequent and higher consumption of candy and fruit gums compared with the comparison group57. Findings from Salvat et al. also indicated that children with ADHD consumed more simple sugars and ready-made meals compared with their peers58.
Collectively, the evidence underscores distinct dietary behaviors among children with ADHD. Nevertheless, the present study addresses an existing knowledge gap concerning the association between ADHD and UPF consumption. To the best of our knowledge, only two studies have examined the association between UPF and ADHD. Both differed from the present study in two important aspects: they examined whether UPF intake predicts ADHD-related symptoms rather than investigating the reverse direction, and they focused on symptoms of ADHD rather than a clinical diagnosis of ADHD. The first study, a large Norwegian cohort study by Borge et al., investigated 77,768 mother–child pairs and reported that a one SD increase in maternal UPF intake during pregnancy was associated with a 3.0% higher ADHD symptom score at age eight (95% CI: 1.5–4.5%)59. The second, a longitudinal Brazilian study by Ferreira et al., followed children from infancy to adolescence (n = 173 at age 12–13 years) and found that higher UPF consumption at ages three to four predicted subsequent hyperactivity/inattention symptoms. Specifically, each 10-gram, 10-kcal, and 10% increase in UPF intake at preschool age conferred relative risks of ADHD symptoms of 1.01 (95% CI: 1.00–1.02), 1.02 (95% CI: 1.01–1.02), and 1.25 (95% CI: 1.04–1.51), respectively, for later hyperactivity/inattention60.
Health implications of UPF
UPF exposure in childhood has been linked to higher risks of obesity, metabolic disturbances, allergy outcomes, and adverse behavioral outcomes15,16,17,18,19, while in adults it has been associated with obesity, type 2 diabetes, cardiovascular disease, common mental disorders, auto-immune diseases, cancer, and mortality21,22,23,24,25,61,62. Although causal mechanisms remain under investigation, several pathways have been proposed: displacement of nutrient-dense foods; excess sugars and high glycemic load leading to dopaminergic dysregulation and metabolic strain; the impact of food additives (e.g., colorants, preservatives, emulsifiers) on neurobehavioral and inflammatory processes; exposure to packaging-derived contaminants such as phthalates and bisphenols; and modulation of the gut–brain axis through changes in microbiota composition and function19,63,64,65.
Nevertheless, it is important to emphasize that UPF represents a heterogeneous group, ranging from items that may carry some nutritional benefits to products that are consistently considered unhealthy, such as sugar-sweetened beverages, ready meals, and confectionery13,66.
Stimulant treatment
Stimulant medications for ADHD are known to improve executive functioning and may also suppress appetite, potentially reducing consumption of UPF67. However, the current study found no differences in UPF consumption between stimulant-treated and untreated children with ADHD. These findings should be interpreted cautiously, given the relatively small subsample. It is possible that children receiving stimulants represent those with more severe or definite ADHD, whereas the remaining children may have varying degrees of behavioral disturbance31, with treatment potentially restoring dietary patterns to a level comparable to those of peers with ADHD who are not treated with stimulants. In addition, while one might expect stimulant-treated children to consume less UPF, given that ADHD stimulants often suppress appetite during the day and may enhance inhibitory control over food selection67, many children experience “rebound effect” as medication effects wear off in the evening68, potentially leading to increased intake of convenient, ultra-processed snacks later in the day. Such compensatory overeating, individual variability in response, and family practices may further mask differences in net daily consumption68,69. Additional studies are required to further explore this issue.
ADHD and UPF consumption
Several explanations may underlie the observed association between ADHD and increased UPF consumption. On the one hand, ADHD symptoms themselves can drive dietary choices. Impulsivity, emotional dysregulation, and reward-processing difficulties bias individuals toward immediately gratifying foods, strongly flavored, and visually appealing, while devaluing minimally processed options that require planning, preparation, or delayed reward70. Evidence of dopaminergic and opioid system dysfunction in ADHD supports this explanation, as disturbances in the brain reward cascade foster heightened reward dependence and may increase preference for foods that rapidly stimulate dopamine release71. Parental feeding practices may contribute to higher UPF consumption in children with ADHD, as parents sometimes provide readily available UPF at mealtimes to avoid conflicts72. Moreover, behavior management strategies often rely on immediate edible rewards, frequently involving ultra-processed sugary foods, which may further reinforce preferences for rapidly gratifying options73. In addition, children with ADHD may exhibit atypical sensory processing and selective eating, which narrows dietary variety and promotes reliance on familiar packaged products74. Finally, distractions and difficulties in daily self-regulation may lead children with ADHD to postpone eating during the day, resulting in increased evening consumption of ready-to-eat and conveniently available UPF75.
UPF and ADHD symptoms
Mechanisms underlying the observed association may also operate in the reverse direction, whereby UPF contribute to ADHD-related symptoms. Interest in diet’s role in ADHD dates back to the 1970s when Dr. Benjamin Feingold hypothesized that eliminating certain additives (salicylates, artificial colors/flavors) could alleviate hyperactivity76. Early controlled studies provided mixed results; a National Institutes of Health panel in 1982 found only a “limited positive association” between Feingold-type elimination diets and reduced hyperactivity, with no consistent or widespread effect77. An early meta-analysis in 1983 similarly reported negligible overall benefits of dietary modification (reviewing 23 studies, it found no significant behavioral improvements)78. However, more recent evidence from rigorous trials has identified a small but significant link between artificial food additives and hyperactive behavior. A 2004 meta-analysis of 15 double-blind, placebo-controlled trials reported that artificial food colorings have an adverse effect on children’s behavior, with an overall effect size of ~0.21 (a modest increase in hyperactivity)79. Likewise, single-trial data confirm this pattern: in one placebo-controlled challenge, the removal of artificial colors and preservatives from the diet of preschoolers led to reduced hyperactivity, and these children showed significantly increased hyperactive behavior when given a drink containing those additives, an effect observed even in children without ADHD80. Another large trial in both preschool-age and school-age children found that consuming a mix of artificial food colorings and a sodium benzoate preservative resulted in increased hyperactivity in 3-year-olds and 8/9-year-olds from the general population81. Notably, the magnitude of these effects was small, but the findings have been robust across studies. This history, from Feingold’s initial diet hypothesis to modern trials, underscores that while diet is not a primary cause of ADHD, certain dietary components (particularly synthetic food additives) can subtly exacerbate hyperactive behaviors, warranting consideration in the overall context of ADHD symptoms.
Stevenson et al. showed a potential gene-diet interaction in behavioral responses to challenge mixtures containing food colorants and sodium benzoate, proposing non-IgE-dependent histamine release triggered by specific additives as a biologically plausible mechanism82. Central histamine H3 receptor activity has been implicated in inhibitory learning, increased hyperactivity in murine models, and enhanced dopamine release in the frontal cortex83, supporting the hypothesis that histamine may mediate the effects of food additives on ADHD-related symptoms. In particular, azo dyes have been reported to provoke urticaria in some individuals irrespective of aspirin sensitivity, providing clinical evidence consistent with additive-induced histamine release84,85,86. Building on this mechanistic rationale, individual susceptibility may be partly explained by polymorphisms in the histamine N-methyltransferase (HNMT) gene, which may impair histamine degradation and thereby moderate a child’s behavioral response to additive challenges82. The interplay between environmental triggers and genetic vulnerability could help explain the heterogeneity of responses observed in the general population.
While not all UPF contain high levels of sugar or additives, many products are characterized by formulations that combine refined carbohydrates, added sugars, artificial colorants, preservatives, or flavor enhancers13. Emerging evidence suggests that diets high in refined sugars may contribute to destabilization of circulating glucose levels and impair prefrontal inhibitory control and attention regulation87,88. Certain additives, including colorants and preservatives, may exacerbate hyperactivity in susceptible children89. UPF may also influence biological pathways relevant to ADHD through the gut. For instance, emulsifiers and some artificial sweeteners can increase intestinal permeability, allowing pro-inflammatory molecules to enter circulation and contribute to neuroinflammatory processes90,91. Similarly, UPF-driven alterations of the gut microbiota may disrupt the production of neurotransmitters and signaling along the gut–brain axis, mechanisms implicated in attentional control and emotional regulation65. At the same time, reliance on UPF displaces nutrient-dense foods and may lead to lower intake of omega-3 fatty acids, iron, and zinc—nutrients shown to support cognitive function and mitigate ADHD symptoms92.
Taken together, these bidirectional explanations suggest a potentially self-perpetuating vicious cycle, whereby ADHD symptoms increase susceptibility to UPF consumption, and high UPF exposure in turn may exacerbate symptom severity and functional impairment.
Public health implications
Given that prior research from Israel and other countries has consistently demonstrated associations between ADHD and unhealthy lifestyle behaviors across the lifespan (such as smoking and alcohol consumption)3,93,94, UPF consumption may likewise represent a lifestyle characteristic of children with ADHD. Reducing children’s UPF burden is reasonable as part of comprehensive ADHD care. In practice, clinicians, parents, and educators can integrate several complementary approaches. Brief screening for UPF-heavy dietary patterns during ADHD evaluations and follow-up visits may help identify children at risk. When such patterns are detected, professionals should encourage practical substitutions with minimally processed, ready-to-eat options and promote the establishment of family-based routines such as shared meals and structured snack times, which provide a more stable nutritional environment. Parents play a central role in shaping daily food availability and choices, while schools and other educational settings strongly influence children’s eating patterns through meal provision, peer modeling, and exposure to food marketing. Health education programs can further enhance awareness of the risks associated with UPF and motivate children and parents to adopt healthier choices in daily life. Finally, policy-level actions, including improving school food environments, setting procurement standards, and regulating marketing to children, can reinforce these efforts, creating a supportive ecosystem for sustained dietary change.
The implications of the current study extend beyond children with ADHD, highlighting a broader child public health relevance in the general population. The cumulative evidence from Schab and Trinh’s systematic review and subsequent double-blind, placebo-controlled challenge studies indicates that artificial food colorants and benzoate preservatives, commonly found in UPFs, are associated with increases in hyperactive behavior in children, irrespective of a prior ADHD diagnosis or baseline levels of hyperactivity79,80,81. Consistent with this interpretation, the current study found comparable effects among children with and without stimulant medication use, which is consistent with the possibility that components of UPF may have a broadly acting effect on child behavior. In children with pre-existing behavioral difficulties, such dietary exposures may exacerbate symptoms to a clinically meaningful extent. Accordingly, the regulation and management of artificial additives in the diet can be considered a population-level child public health concern, rather than an issue confined to clinical subgroups.
Limitations and strengths
The study had several limitations. Selection bias may have occurred, as ~10% of eligible candidates did not participate, and children with missing ADHD or nutrition data, as well as those with implausible energy intakes, were excluded. To mitigate non-response effects, survey weights were applied, and sociodemographic characteristics of included and excluded participants were compared, with no differences found.
Information bias was another concern. ADHD status was determined by a physician’s diagnosis as reported by parents, lacking independent clinical verification. Relying on parent-reported diagnoses could introduce misclassification of ADHD status. However, previous research suggests that a parent-reported provider diagnosis of ADHD is fairly reliable for epidemiological purposes95, and the observed prevalence was consistent with Israeli data from medical records49,50. Nonetheless, over-diagnosis cannot be ruled out, as the distinction between simple behavioral difficulties and clinically defined ADHD is often challenging to delineate, particularly amid broad diagnostic trends in recent years, including an increase in diagnoses among children with milder symptomatology96. Consequently, the inclusion of misclassified or borderline cases within the ADHD group may have diluted the true magnitude of the associations observed.
Dietary intake was self-reported as well, but the use of validated questionnaires and visual aids32,33, together with exclusion of children with extreme energy intake values, helped mitigate this bias. In addition, the use of a single 24-h recall may have limited the ability to capture habitual patterns and normal day-to-day variation in diet32,33, potentially misrepresenting usual intake. Nonetheless, using one recall per person is a common approach in surveys and can be sufficient for estimating mean dietary intakes at the population level (as the estimates of the group’s intake are statistically robust and less affected by day-to-day within-person variation)97, and the large sample size, year-round data collection, and careful scheduling of interviews across different days of the week increased representativeness.
It should be noted that the NOVA classification has recognized limitations. The UPF group encompasses diverse foods, some of which are not uniformly harmful66. However, it remains a widely used and accepted system for classifying UPF in contemporary nutritional epidemiology25. The results need to be interpreted with appropriate nuance, and the NOVA classification should be viewed as a general indicator of highly processed dietary patterns rather than as a blanket condemnation of all NOVA 4 items.
Interviewer bias was also a possible concern, but this was reduced through standardized protocols and specially trained interviewers. Social desirability bias was likely non-differential between exposure groups, thereby attenuating rather than exaggerating observed associations.
As a cross-sectional study, causal inference cannot be drawn. It remains unclear whether ADHD symptoms influenced food choices or vice versa. Accordingly, the findings were interpreted as associations, with a recognition that the directionality of the relationship remains unclear; both the possibility that ADHD may contribute to higher UPF intake, and that UPF consumption may exacerbate ADHD-related symptoms, warrant consideration. Longitudinal studies are needed to clarify the temporal and causal nature of this association. In addition, dietary changes following ADHD diagnosis, such as adopting healthier eating habits98, may also have affected associations, but this effect is likewise expected to attenuate the observed relation. Finally, the focus on Israeli school-aged children further limits generalizability to other age groups or cultural settings.
Nevertheless, the study has several notable strengths. It included a large, nationally representative sample with a high response rate. Internal validity was strengthened by the use of validated measurement tools, standardized protocols, and interviews conducted by trained personnel. Comprehensive dietary information was obtained from all participants, along with detailed information on pharmacological treatment. The consistency of the results across various statistical approaches supports the robustness of the observed associations. Moreover, the analyses were rigorously adjusted for a wide range of potential confounders, reducing the likelihood of residual confounding. Nonetheless, the risk of residual confounding and the existence of an unmeasured third factor that is linked to both ADHD and UPF consumption still remains.