Abstract
Background:
The precise volatile compounds accountable for the characteristic scent of the wheat aroma were presently unidentified. Given the prominent manifestation of “wheat aroma” in wheat cultivated in saline-alkali (SA) soil, five wheat varieties planted in saline-alkali soil and control soil (low saline-alkali soil) to create “rich-aroma” and “bland-aroma” samples, respectively.
Methods:
The volatile profiles of these two groups of samples were analyzed using headspace solid-phase microextraction coupled with gas chromatographyed with gas chromathes-SPME-GC-MS), and differential characteristic volatile compounds between “rich-aroma” and “bland-aroma” samples were identified by combining paired t-tests, relative odor activity values (ROAVs), and molecular docking analysis.
Results:
A total of 75 volatile compounds were identified using HS-SPME-GC-MS. Paired t-test result revealed that a significant increase (p < 0.05) in the peak areas and relative contents of limonene (>109.74% increase), β-pinene (> 474.09% increase) contrasted with reduced butanoic acid (>38.71% reduction) and (E,E)-3,5-octadien-2-one (>16.05% reduction) in saline-alkali soil-cultivated wheats. Molecular docking demonstrated high binding energies (<-4.25 kcal/mol) of limonene, β-pinene, and (E,E)-3,5-octadien-2-one to olfactory receptors, corroborated by relative odor activity values (ROAVs. > 1).
Conclusion:
Limonene, β-pinene and (E,E)-3,5-octadien-2-one are identified as possible saline-alkali soil-induced wheat aroma biomarkers, and offer theoretical insights for verificating wheat aroma. Identifying potential characteristic biomarkers in wheat under saline-alkali stress provides important theoretical insights for improving the flavor characteristics of wheat.
1 Introduction
Saline-alkali soil leads to soil compaction and nutrient imbalance, hampering wheat growth (1). However, fully exploiting the utilization pathways of saline-alkali soil can not only provide complementary grain production but also yield distinctive crops, thereby enhancing agricultural biodiversity and economic value while contributing to food security. China possesses substantial areas of utilizable saline-alkali soil. How to utilize these lands has become a focus of attention. Notably, sensory evaluations by Cangzhou residents suggest that wheat grown in SA soil exhibits a distinct and intense “wheat aroma” (2), thereby enhancing its market appeal and added value (3, 4). However, the specific chemical alterations responsible for the enhancement of its wheat aroma remain unclear.
The wheat aroma is composed of volatile compounds such as aldehydes, alcohols, esters, ketones and terpenes, and its formation involves complex biosynthesis. The formation of wheat aroma involves complex biosynthetic pathways. During growth, ripening and post-ripening stages of wheat grains, enzyme-mediated carbohydrate, fatty acid and protein metabolism generate diverse volatile organic compounds (VOCs), which collectively define wheat aroma profiles (5). Studies on wheat flour have consistently identified lipid-derived aldehydes such as hexanal, nonanal and (E)-2-non-enal, together with cereal-like volatiles including 2-pentylfuran and 1-octen-3-ol, as key contributors to green, fatty and grain-like notes in wheat grain and flour (6). In addition, benzaldehyde and 2-acetyl-1-pyrroline, although present at lower concentrations, exhibit high odor potency and contribute almond-like, roasted and popcorn-like nuances (7).
The biosynthesis of these VOCs is profoundly influenced by abiotic stress conditions. Such stresses alter the pools of volatile precursors such as amino acids (e.g., glutamic acid and proline) (8), unsaturated fatty acids (e.g., linoleic acid) (9), and soluble sugars (e.g., glucose, fructose and sucrose) (10) by triggering osmotic adjustment and oxidative stress responses. Concurrently, signaling pathways based on reactive oxygen species (11) and phytohormones (12) regulate the expression of key enzyme genes, thereby selectively promoting the formation of specific aldehydes, lactones, ketones and terpenoids like limonene (13), β-ocimene (14), and β-pinene (15). This metabolic reprograming fundamentally reshapes the aroma profile of the plant. For instance, mild drought stress has been shown to significantly increase the diversity of VOCs in tea leaves, elevating the concentrations of low-threshold VOCs associated with sweet, floral, and fruity aromas (16). Similarly, salt stress (e.g., seawater irrigation in grapes) enhances the accumulation of volatiles derived from fatty acid and isoprene pathways, including terpenes such as citronellol and myrcene (17). While the influence of abiotic stress on cereal aroma has predominantly focused on rice—where moderate salt stress enhances the characteristic aroma by modulating the biosynthesis of 2-acetyl-1-pyrroline through altered amino acid metabolism and lipid peroxidation (18)—far less attention has been paid to wheat. Our previous research found that the characteristics of wheat under saline-alkali stress are elevated 1-butanol and 1-octen-3-ol but reduced ethyl 3-methylbutyrate and 1-octen-3-one by gas chromatography-ionmobility spectrometry (GC-IMS) (19). However, GC-IMS exhibits a certain selectivity or limited detection range for compounds, meaning that the aforementioned four compounds may represent only a fraction of the complex aroma profile. To capture the complete VOC profile more comprehensively and without biasy modulating the biosynthesis of 2ehydes, lac Dsity and economic value while contrib-IMSpture the complete VOC profilgas chromatography-mass spectrometry (GC-MS) for sample analysis. GC-MS offers broader compound identification capabilities, higher sensitivity, and robust spectral library support (20, 21), facilitating the discovery of more potential aroma-active compounds. This study employs an intensity-graded sampling strategy, classifying five wheat varieties grown in both SA and control soils into “rich-aroma” and “bland-aroma” groups. Therefore, the varieties are repetitive, this study does not focus on the differences brought by different genotypes. Using paired HS-SPME-GC-MS profiling, molecular docking, and ROAV analysis, this study aims to (1) Identify differential VOCs in SA-stressed wheat and select potential signature biomarkers; and (2) elucidate their molecular interactions with olfactory receptors. This integrated approach advances understanding of abiotic stress-induced aroma formation, and provides essential theoretical insights for improving wheat flavor profiles.
2 Materials and methods2.1 Sample collection
In the year 2022, five distinct wheat cultivars—Cangmai 6002 (C6002), Cangmai 6005 (C6005), Jimai 22 (JM22), Shiluan 02-1 (SL02-1), and Malannuomai NO.1 (N01)—were collected from two locations in Cangzhou, Hebei Province, China, namely Xi Huayuan (XHY) and Zhong Jie village (ZJ), which are 50 km apart in a straight line, with basically the same climate conditions but different longitude and latitude and soil salinity and alkalinity (Table 1). Among these cultivars, C6002 and C6005 are medium gluten wheat varieties with moderate tolerance to salinity-alkalinity, whereas JM22, SL02-1 and N01 are categorized as medium-gluten, strong-gluten, and waxy wheat varieties, respectively, all of which are sensitive to salinity-alkalinity stress, and are the main cultivated varieties in the local area (Supplementary Table 1). The five wheat varieties were assigned randomly to plots in each location following a randomized complete block design, with each plot covering an area of 666.67 m2 (1 mu).
The cultivation sitelongitude and latitudeSoil horizon [cm]pH (mean ± s.d.)Total salt content (mean ± s.d.)[g/kg]AbbreviationXi HuayuanE:116°52′38.22″
N:38°22′42.38″0-407.76 ± 0.071.23 ± 0.03XHYZhong JieE:117°28′16.99″
N:38°24′1.75″0-408.34 ± 0.20**2.54 ± 0.35**ZJ
The pH and total salt content of soil in wheat cultivation sites.
The pH and total salt content for the sampling sites was provided by the Academy of Agriculture and Forestry Sciences, Hebei Key Laboratory of Drought–Alkali Tolerance in Wheat, Cangzhou, China.“**” represents a significant difference (p < 0.01).
After the wheat had fully matured, they were collected using an Austrian Wintersteiger classic combine harvester. From each sample, approximately 5 kilograms were randomly selected and labeled. Then, three separate 100-gram portions were drawn at random from each 5-kilogram sample. A total of 30 samples were obtained (2 fields × 5 genotypes × 3 replicates), and all were processed using a versatile grinder (model ZK-100B, manufactured by Zhongke Haoyu Technology Co., Ltd., China). After processing, the samples were placed in sealed bags and kept at -20°C for subsequent use.
2.2 Gas chromatography-mass spectrometry analysis
The extraction and analysis of VOCs was conducted according to the published experimental scheme, with some improvements (22). The headspace solid-phase microextraction (HS-SPME) technique was employed to extract VOCs. Wheat wholemeal samples (2 g) were placed in 20 mL vials, equilibrated at 60°C for 40 min, and extracted with HS-SPME for 80 min at 60°C. The extracted fiber was 50/30 μm divinylbenzene/carboxenTM/polydimethylsiloxane (DVB/CAR/PDMS) from Supelco (Bellefonte, PA, United States).
Analysis of the VOCs was performed using GC-MS (TRACE1300, ISQ9000, United States), employing a DB-Wax capillary column (30 m × 0.25 mm × 0.25 μm). Post-extraction, the fiber was desorbed into the GC injector port for 2 min at 250°C in splitless mode. Helium at a flow rate of 1 mL/min was used as the carrier gas. The oven temperature was initially set at 40°C for 5 min, then ramped at 5°C/min to 220°C for 10 min. The MS was operated in electron ionization mode (EI) at 70 eV, with a multiplier voltage of 1247 V. The ion source temperature was maintained at 200°C, and the interface temperature at 250°C. Full scan mode was employed to detect VOCs across a mass range from m/z 35 to 600. Each sample underwent triplicate analysis.
2.3 Identification of volatile organic compounds
Compounds identification relied on comparison of their mass spectra with those in the NIST 20 library. VOCs with Match Factor greater than 750. Additionally, retention index was calculated using Kovats retention index based on a series of straight-chain alkanes (C7-C27) under the aforementioned chromatographic conditions.
To confirm GC-MS identifications (Table 2), the standards (reference compounds) of 2-pentyl-furan (3777-69-3, ≥ 98%), 1-hexanol (111-27-3, ≥ 99%), 1-octen-3-ol (3391-86-4, ≥ 98.04%) and limonene (138-86-3, 1,000 μg/mL) were selected. These standards were diluted and mixed with high-quality chromatographic grade methanol (99.9%). A stock solution containing a concentration of 50 ppm for the mixed standards was prepared. The sources of 2-pentyl-furan, 1-hexanol, 1-octen-3-ol and methanol were Shanghai Macklin Biochemical Co., Ltd., China, while the source of Limonene was Tan-Mo Technology Co., Ltd., China.
No.ClassificationVolatile organic compoundsMolecular formulaCAS No.Retention index
(Exp)Retention index (Lit)1Alcohols(30.05%∼36.13%)1-Propanol, 2-methyl-C4H10O78-83-11,091-21-Butanol, 3-methyl-C5H12O70907-83-41,2131,20931-PentanolC5H12O71-41-01,2481,25041-HexanolC6H14O111-27-31,3531,35552-Hexen-1-ol, (Z)-C6H12O928-94-91,4031,40761-Octen-3-olC8H16O3391-86-41,4501,45071-HeptanolC7H16O111-70-61,4531,4538Linalyl oxideC10H18O260047-17-81,4661,46791-Hexanol, 2-ethyl-C8H18O104-76-71,4951,491102,3-ButanediolC4H10O2513-85-91,5461,549111-OctanolC8H18O111-87-51,5571,55912Terpinen-4-olC10H18O562-74-31,6061,60213Benzyl alcoholC7H8O100-51-61,8821,8871Alkanes
(22.59%∼26.55%)Pentane, 3,3-dimethyl-C7H16562-49-21,039-2Hexane,2,2,5,5-tetramethyl-C10H221071-81-41,159-3Heptane, 3-ethyl-5-methyl-C10H2252896-90-91,242-4Dodecane, 4-methyl-C13H286117-97-11,256-5Octane, 2,3,6-trimethyl-C11H2462016-33-51,279-6TridecaneC13H28629-50-51,3001,3007Dodecane, 4,6-dimethyl-C14H3061141-72-81,358-8TetradecaneC14H30629-59-41,4001,4009PentadecaneC15H32629-62-91,5001,50010HeptadecaneC17H36629-78-71,700-1Terpenes (0.81∼16.41%)β-pineneC10H16692-96-61,1071,1082LimoneneC10H16127-91-31,2001,1991Aromatic compounds(9.39%∼15.73%)Benzene, 1,3-dimethyl-C8H10108-38-31,1451,1432o-XyleneC8H1095-47-61,1881,1873StyreneC8H8100-42-51,2631,2614p-CymeneC10H1499-87-61,268-5Benzene,1-methyl-4-propyl-C10H141074-17-51,3101,3126NaphthaleneC10H891-20-31,7511,7457Benzene,1-(1,5-dimethyl-4-hexenyl)-4-methyl-C15H22644-30-41,7781,7778Naphthalene, 2-methyl-C11H1091-57-61,8631,8569Naphthalene, 1-methyl-C11H1090-12-01,8981,884101,8-dimethyl-NaphthaleneC12H12569-41-51,9731,989111,6-dimethyl-NaphthaleneC12H12575-43-92,0032,006122-Methoxy-4-vinylphenolC9H10O27786-61-02,2052,220132,4-Di-tert-butylphenolC14H22O96-76-42,3172,3301Acids (5.56∼9.08%)Acetic acidC2H4O264-19-71,4441,4492Butanoic acidC4H8O2107-92-61,627-3Butanoic acid, 3-methyl-C5H10O2503-74-21,6591,6624Hexanoic acidC6H12O2142-62-11,8511,8465Heptanoic acidC7H14O2111-14-81,9571,9506Octanoic acidC8H16O2124-07-22,0642,0607Non-anoic acidC9H18O2112-05-02,1702,1708Undecanoic acid, 2-methyl-C12H24O224323-25-92,218-1Esters (3.11∼3.79%)Octanoic acid, methyl esterC9H18O2111-11-51,394-2Non-anoic acid, methyl esterC10H20O21731-84-61,5031,4993gamma-ValerolactoneC10H18O22890-67-71,613-4ButyrolactoneC4H6O296-48-01,638-5Lavender lactoneC7H10O21073-11-61,6801,6826gamma-HexalactoneC6H10O2695-06-71,6881,6947Dodecanoic acid, methyl esterC13H26O2111-82-01,8021,8048gamma-Non-anolactoneC9H16O2104-61-02,0402,0401Aldehydes (2.26∼4.63%)Non-analC9H18O124-19-61,3981,40122,4-HexadienalC6H8O142-83-61,4091,41032-Octenal, (E)-C8H14O2548-87-01,4351,43942-Non-enal, (E)-C9H16O18829-56-61,5411,54552,6-Non-adienalC9H14O557-48-21,5981,59962-Decenal, (E)-C10H18O3913-81-31,6491,65372,4-Non-adienal, (E,E)-C9H14O5910-87-21,7081,71482-UndecenalC11H20O2463-77-61,7571,75592,4-Decadienal, (E,E)-C10H16O25152-84-51,8171,82310VanillinC8H8O3121-33-52,5852,5801Ketones(1.75∼2.79%)2-OctanoneC8H16O111-13-71,2891,28725-Hepten-2-one, 6-methyl-C8H14O110-93-01,3421,34533-Octen-2-oneC8H14O1669-44-91,4121,41145-Octen-4-one, 7-methyl-C9H16O32064-78-11,469–5(E,E)-3,5-Octadien-2-oneC8H12O30086-02-31,5761,57062-UndecanoneC11H22O112-12-91,6021,59875,9-Undecadien-2-one, 6,10- dimethyl-, (E)-C13H22O3796-70-11,8591,8591Olefins1-MethylcyclohepteneC8H1455308-20-81,703-2(0.12%∼0.26%)3-Heptene, 2- methyl-, (E)-C8H16692-96-61,761-1Other compounds (3.66∼9.47%)Furan, 2-pentyl-C9H14O3777-69-31,2361,2392BenzothiazoleC7H5NS95-16-91,9651,958
Volatile organic compounds identified by GC-MS.
Retention Index (Exp) indicates experimental linear retention index; Retention Index (Lit) indicates retention index reported for reported in the literature. “-” indicates that no appropriate reference value was retrieved under similar chromatographic conditions.
An aliquot of 1 mL from the above-prepared mixed standard sample stock solution was extracted into a headspace bottle with a volume of 20 mL. Then it was analyzed by HS-SPME-GC-MS using the same conditions as the previous tests. The reliability of VOCs identification results in this study can be considered satisfactory, if the retention times for the aforementioned reference compounds in both standard and test samples were essentially identical. The confirmation results are presented in Supplementary Figure 1.
2.4 Molecular docking
The method was adapted from to Jia et al. (23) with minor modifications. This study investigated interactions between sixteen significant human olfactory receptors (OR51E1, OR52D1, OR1D2, OR7C1, OR8B3, OR6F1, OR13C3, OR10A6, OR5P3, OR3A1, OR14A2, OR9G1, OR11H6, OR2W1, OR11H4, and OR4Q3) and impotent VOCs. Protein structures for the olfactory receptors were sourced from UniProt and AlphaFold. SDF files for each small molecule ligand were obtained from the PubChem database and converted to PDB format using PyMOL 2.3.0. Molecular docking was performed using AutoDockTools (ADT) v1.5.7 and AutoDock Vina v1.2.0, prioritizing receptor-ligand complexes prioritized based on low binding energy rankings. The results were further analyzed using PyMOL and LigPlot for interaction visualization.
2.5 Relative odor activity values of volatile compounds analysis
The ROAV was a very useful tool for assessing the contribution of each aroma compound to the overall flavor profile (24). The larger the ROAV, the greater the contribution to the overall flavor of the sample.
The ROAVs. of each component were calculated to analyze its contribution of each component to the overall wheat aroma. The formula was as follows:
ROAV = 100 × C%A/C%max × Tmax/TA
Where, C%A and TA were the relative percentage content and odor threshold concentration (Table 3) of the compound, respectively. C%max and Tmax were the relative percentage content and odor threshold concentration (Table 3) of the component that contributes the most to the overall flavor of the sample, respectively.
Volatile organic compoundsSensory threshold(μ g/kg)C6002C6005JM22N01SL02-1Odor descriptionXHYZJXHYZJXHYZJXHYZJXHYZJLimonene200100.00100.00100.00100.0021.20100.00100.00100.00100.00100.00Sweet, orangeβ-pinene1407.2123.281.6115.9929.3135.77-2.370.545.56Pine, resin, turpentine(E,E)-3,5-octadien-2-one10014.999.9924.8010.67100.0021.4547.1114.0233.9810.53Fatty
ROAVs calculation of key volatile organic compounds of wheat cultivated in XHY and ZJ.
“ – ” indicates that the system is not detected.
2.6 Data analysis
All experiments were conducted in triplicates and the data are presented as the mean ± standard deviation. The data treatment was carried out using the SPSS 23.0 package software (SPSS Inc., Chicago, IL, United States). T-tests were performed to analyze significant differences in VOCs of wheat varieties cultivated in various cultivation sites. The bar graph and heat map were created using Origin 2024 software (OriginLab Corporation, Northampton, MA). Principal component analysis (PCA) and linear discriminant analysis (LDA) were utilized for data pattern identification and analysis.
3 Results3.1 Volatile organic compounds analysis by gas chromatography-mass spectrometry
The composition and contents of VOCs in five wheat varieties cultivated in SA and control soils were compared by GC-MS. A total of 75 VOCs were identified and classified into 11 chemical classes namely: alcohols, hydrocarbons, terpenes, aromatic compounds, acids, ethers, esters, aldehydes, ketones and heterocyclic compounds. Calculate the proportion of the peak area of each type of substance in all substances in each sample (Table 2). Among these classes, alcohols, alkanes, terpenes, aromatic compounds, acids, esters, aldehydes and the remaining substances accounted for 30.05–36.13%, 22.59–26.55%, 0.81–16.41%, 9.39–15.73%, 5.56–9.08%, 3.11–3.79%, 2.26–4.63%, and 5.53–12.52% of the total VOCs in the wheat varieties, respectively.
Paired t-tests demonstrated significant alterations in the volatile profiles of saline-alkali (SA) soil-cultivated wheat versus control samples (Figure 1A). Under SA cultivation, seven compounds exhibited significantly elevated peak areas (Supplementary Table 2): β-pinene showing the most pronounced increase (525.91%, p < 0.05), followed by limonene (121.15%, p < 0.01), 1-(1,5-dimethyl-4-hexenyl)-4-methyl-benzene (35.28%, p < 0.05), lavender lactone (35.71%, p < 0.05), 1,6-dimethyl-naphthalene (38.82%, p < 0.01), 1,8-dimethyl-naphthalene (13.78%, p < 0.05), and tetradecane (4.72%, p < 0.05). In contrast, the levels of (E,E)-3,5-octadien-2-one and butanoic acid were significantly decreased (Supplementary Table 2) by 16.05% (p < 0.05) and 38.71% (p < 0.01), respectively. It was demonstrated SA stress induces metabolic reprograming that favors the biosynthesis of terpenoid and aromatic compounds.
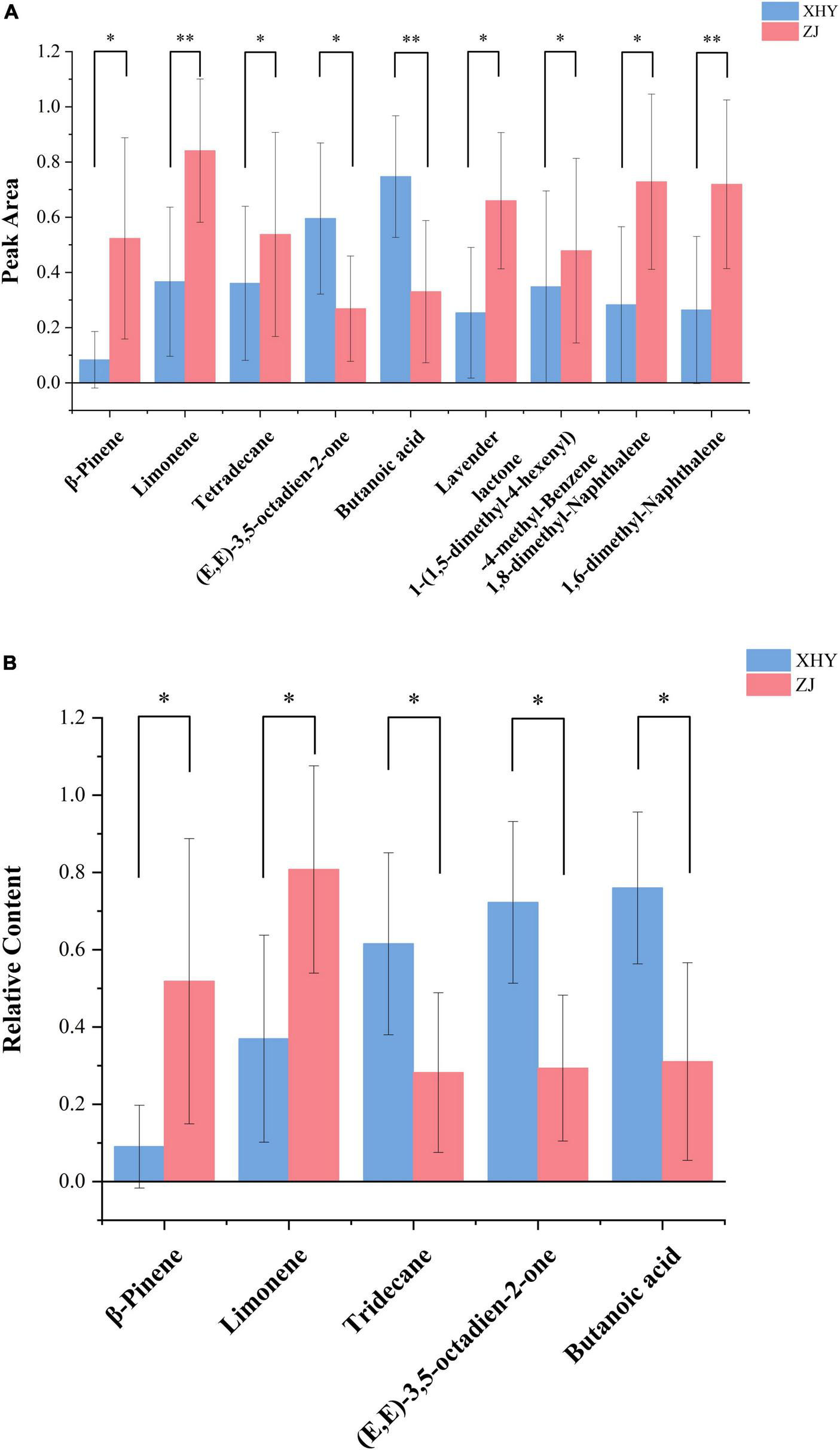
Bar chart of compounds with significantly different peak areas (A) and relative contents (B) in wheat cultivated at two sites (Xi Huayuan and Zhong Jie). * and ** represent the significant difference (p < 0.05) and (p < 0.01) respectively. All data were normalized using Min-Max.
To better characterize VOCs changes across five wheat varieties cultivated in soils with different SA levels, the relative contents of key VOCs were measured, and the results are shown in Figure 1Bβ-pinene and limonene exhibited marked increases (Supplementary Table 3) by 474.09% (p < 0.05) and 109.74% (p < 0.01), respectively, confirming their roles as SA stress-responsive biomarkers. Conversely, tridecane, (E,E)-3,5-octadien-2-one and butanoic acid exhibited significant reductions (Supplementary Table 3): 10.17% (p < 0.05), and 42.95% (p < 0.01), respectively.
Based on the pairing results of peak areas and relative contents, β-pinene, limonene, (E,E)-3,5-octadien-2-one and butanoic acid could be regarded as important indicator compounds for wheat cultivation in SA soil. Given that wheat cultivated on SA soil has a relatively strong aroma, these compounds might also be the ones that contribute to the wheat aroma.
3.2 Molecular docking analysis
To further investigate whether these four VOCs act as critical compounds for wheat cultivated in saline-alkali land, molecular docking was employed. In the docking simulations, a negative binding energy indicates spontaneous ligand-receptor binding, with its magnitude correlating directly to binding affinity. More negative values reflect stronger intermolecular interactions and higher probability of stable complex formation (25). Based on established criteria: binding energy < -4.25 kcal/mol suggests detectable binding activity, implying potential ligand-receptor recognition; binding energy < -5.0 kcal/mol signifies high binding affinity, likely corresponding to biologically relevant interactions (26). For this study, compounds meeting the threshold of binding energy < -4.25 kcal/mol were prioritized for further analysis. The binding energy heat map between the important compounds and the olfactory receptors (Figure 2) revealed that binding energy range of butanoic acid with all 16 olfactory receptors was -4.79 to -3.82 kcal/mol, and its affinity is relatively weak (> -5 kcal/mol), whereas limonene, β-pinene and (E,E)-3,5-octadien-2-one exhibited moderate to strong binding affinity, with binding energies of -6.02 to -4.64 kcal/mol, -6.19 to -4.39 kcal/mol, and -6.17 to -4.29 kcal/mol, respectively. These results align with prior evidence that such binding energies modulate receptor conformational dynamics and enhance agonist efficacy in flavor perception (27).
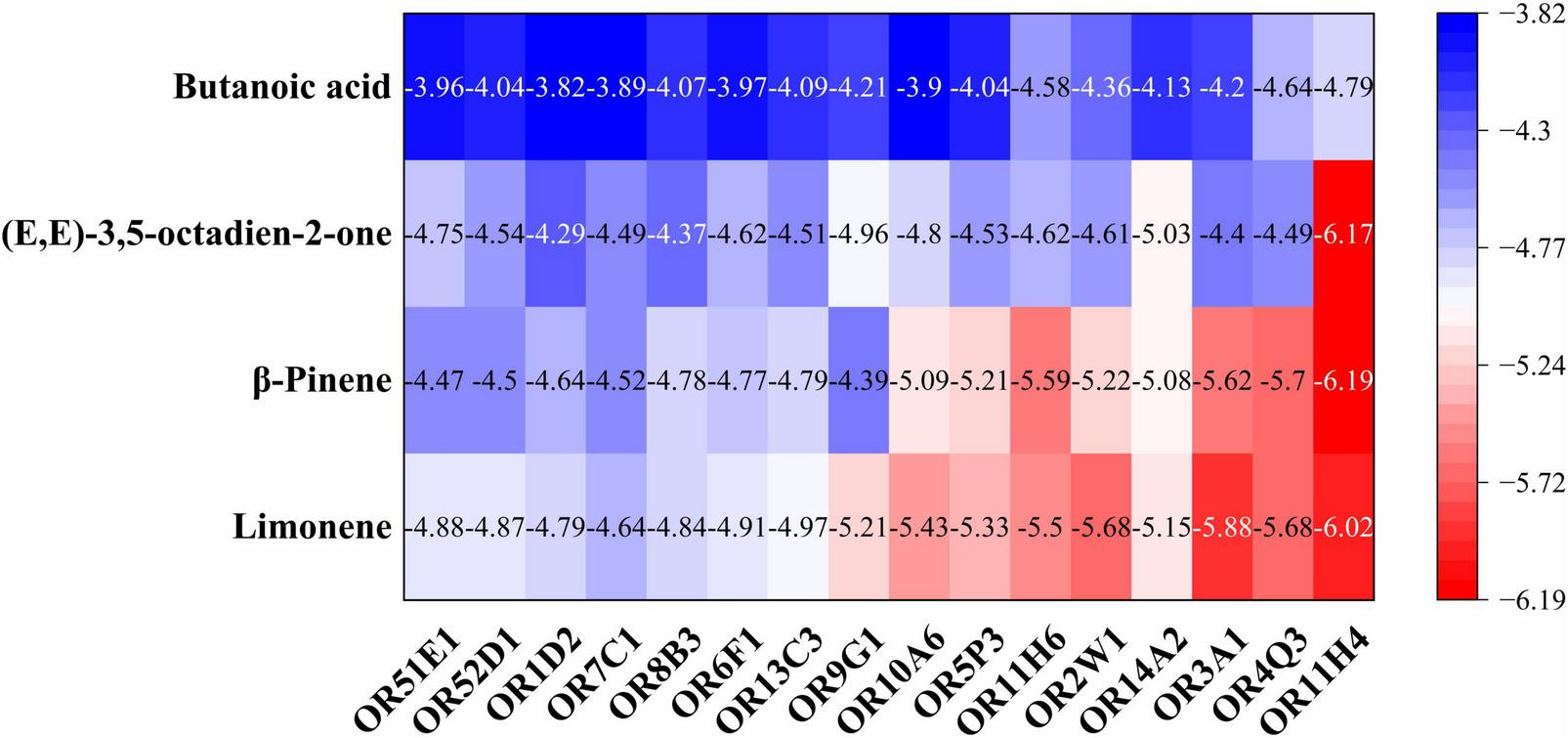
The binding energy heat map between the important compounds and the olfactory receptors.
Molecular docking studies have demonstrated that aroma compounds enhance food flavor by binding to potential active sites of ORs (28). Among the sixteen ORs examined, OR11H4 exhibited significantly stronger binding affinity (Figure 2), and was therefore selected to elucidate mutual binding interactions with three key compounds. As shown in Figure 3, fourteen interacting amino acid binding residues (Leu217, Ser213, Tyr269, Tyr83, Tyr166, Tyr288, Val265, Val216, Phe114, Phe115, Phe261, Gly118, Thr119, and Cys122) were identified in the OR11H4 binding porket. Notably, Leu217, Ser213, Tyr269, Phe115, and Thr119 served as common binding sites, suggesting their suggesting their critical role as key interaction residues (23). Furthermore, limonene formed hydrophobic interactions with Leu217, Ser213, Tyr269, Tyr83, Val265, Val216, Phe114, Phe115, Gly118, and Thr119; β-pinene bound via hydrophobic interactions to Leu217, Ser213, Tyr269, Tyr166, Tyr288, Val265, Phe115, Phe261, Gly118, and Thr119; while (E,E)-3,5-octadien-2-one established hydrogen bonds with Tyr269 and Tyr288, alongside hydrophobic interactions involving Leu217, Ser213, Tyr83, Tyr166, Val216, Phe114, Phe115, Thr119, and Cys122.
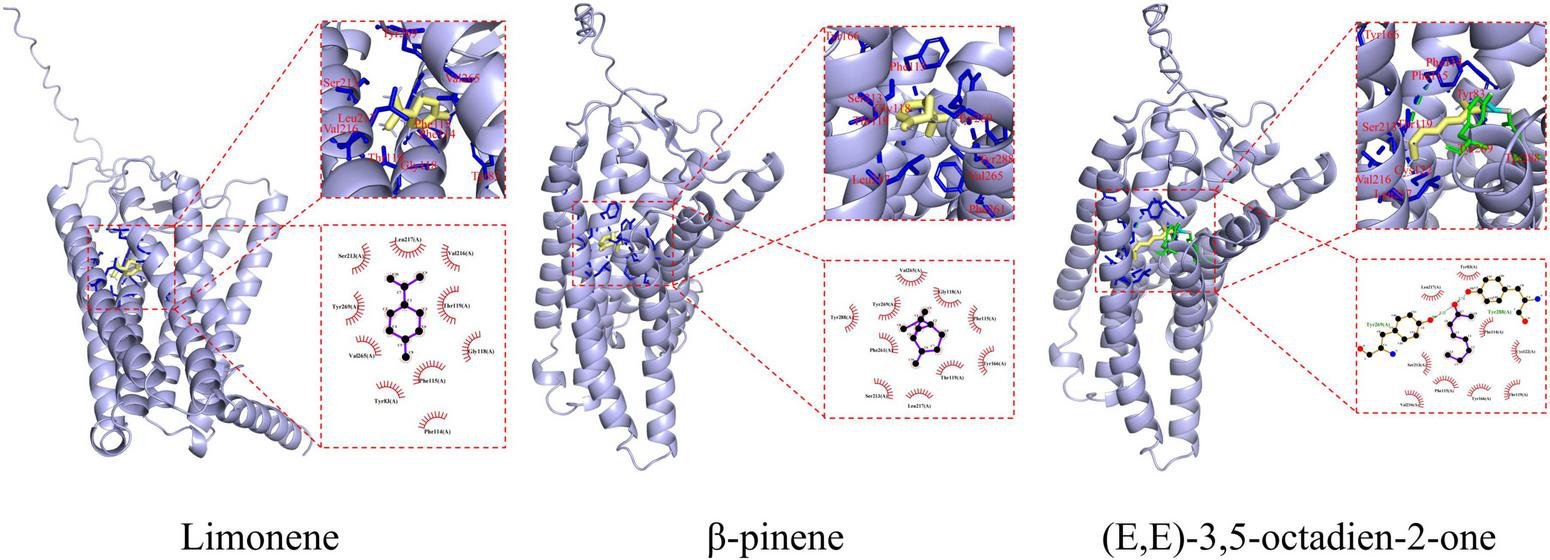
Binding sites and interactions for molecular docking between key volatile organic compounds (limonene, β-pinene, and (E,E)-3,5-octadien-2-one) and OR11H4.
In summary, Leu217, Ser213, Tyr269, Phe115, and Thr119 are critical binding residues, with hydrophobic interactions and hydrogen bonds acting as primary driving forces in ligand-receptor recognition.
3.3 Volatile organic compounds analysis by relative odor activity values
The characteristic “wheat aroma” is influenced not only by the concentration of key odorants but also by their sensory threshold. Relative odor activity values (ROAVs) could be utilized for assessing the contribution of individual components toward the overall flavor (24). Based on established criteria (29), VOCs with ROAVs. > 1 dominate the overall flavor, while those with ROAVs between 0.1 and 1 exert modifying effects; VOCs with ROAVs. < 0.1 contribute negligibly. To identify key aroma compounds in the SA soil-cultivated wheat, ROAVs were calculated for limonene, β-pinene, and (E,E)-3,5-octadien-2-one. Consistently, ROAVs exceeded 1 across five wheat varieties grown in soils with distinct salinity-alkalinity gradients (Table 3), indicating their significant contribution to the characteristic flavor of SA-cultivated wheat.
Based on this premise, the three VOCs were considered as key flavor compounds that potentially contribute significantly to the overall flavor perception of “wheat aroma.” The proportion range of each component can be determined by calculating the percentage of peak area for each key flavor compound among all key flavor compounds, as presented in Supplementary Table 4.
3.4 Principal component analysis and linear discriminant analysis
Principal component analysis (PCA), an unsupervised dimensionality reduction technique, extracts orthogonal components that maximize variance retention while approximating original variables. This method effectively identifies inter-group similarities and intra-group differences within complex datasets (30). PCA conducted on the three potential key aroma compounds revealed that the first three principal components (PC1-PC3) accounted for 67.45, 22.09, and 10.46% of total variance, respectively, with a cumulative variance contribution rate was 100% (Figure 4A). This confirms PC1-PC3 comprehensively capture the dominant variation patterns in the aroma profile. Loadings analysis further identified limonene, β-pinene and (E,E)-3,5-octadien-2-one could serve as reliable indicators for distinguishing XHY and ZJ cultivated wheat.
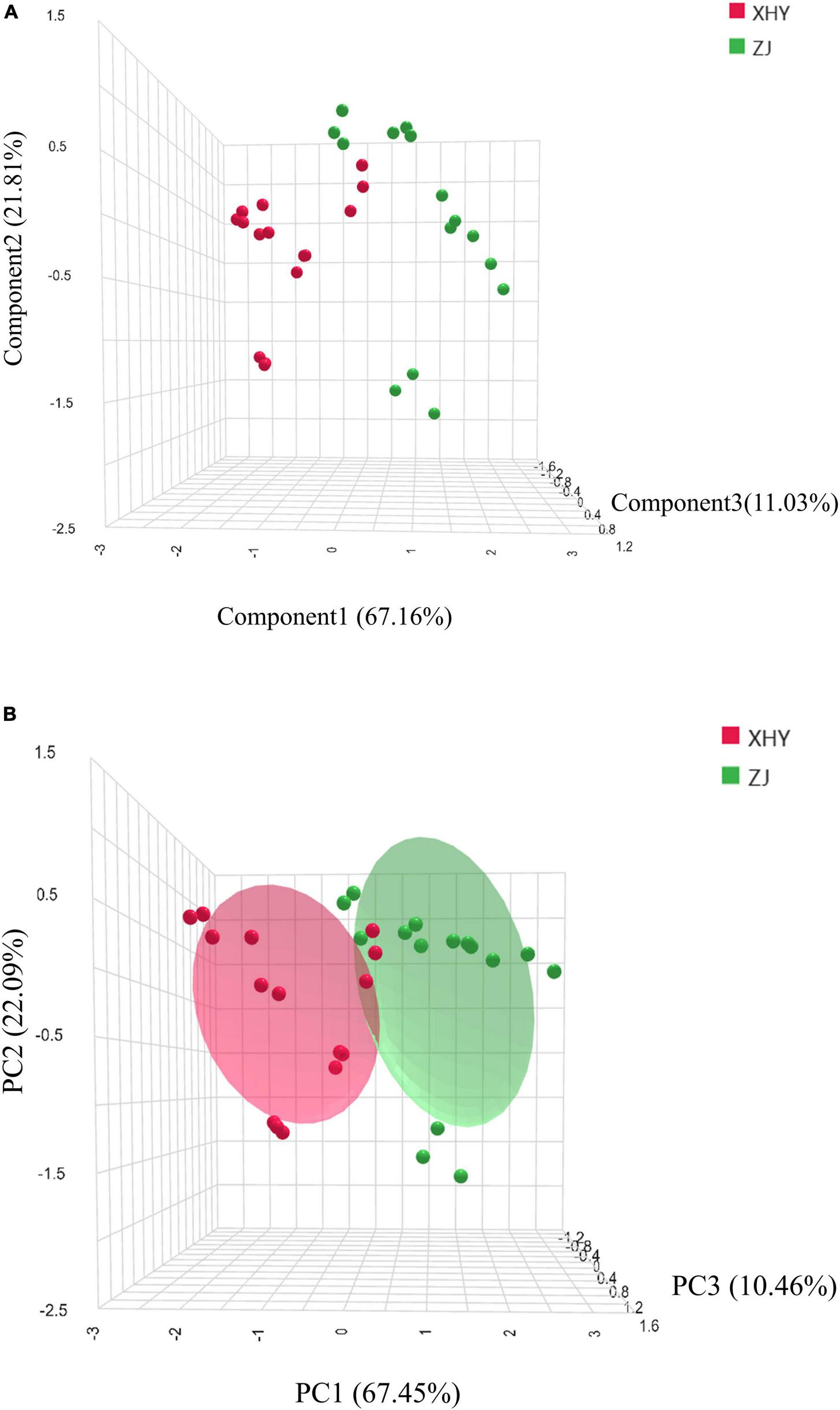
PCA (A) and LDA (B) of the selected 3 key volatile organic compounds in wheat cultivated in XHY and ZJ.
Complementarily, linear discriminant analysis (LDA)—a supervised technique optimizing inter-class separation while minimizing intra-class variance (31)—was employed to validate the classification efficacy of these three VOCs. Notably, LDA achieved 90.0% cross-validated accuracy in distinguishing wheat grown in distinct salinity-alkalinity soils. Projection plots (Figure 4B) demonstrated complete spatial segregation between XHY and ZJ samples along the first three discriminant functions (LD1-LD3), which collectively explained 100% of total variance. LD1 dominated class separation (67.16% variance), with limonene exhibiting the highest standardized coefficient (discriminant coefficient = 0.73), indicating its pronounced sensitivity to salinity gradients.
Synthesizing PCA and LDA outcomes, limonene, β-pinene, and (E,E)-3,5-octadien-2-one are established as signature flavor compounds defining the aroma signature of SA-cultivated wheat, and may also be the characteristic biomarkers of wheat aroma.
4 Discussion
The results show that limonene, β-pinene and (E,E)-3,5-octadien-2-one can serve as an reliable indicators for the identification of wheat cultivated in SA soil (Figure 1). This is in line with the more general notion that abiotic stress reshapes plant secondary metabolism (including volatile terpenoids and carbonyl compounds).
As shown in Table 3, β-pinene exhibited flavors of pine, resin, and turpentine, while limonene possessed sweet and orange notes (32). As monoterpenes, both compounds exhibit significant accumulation under SA stress (pH range: 8.14∼8.54; total salt content: 2.19∼2.89 g/kg) condition (Figure 1), aligning with established patterns of terpenoid enhancement in plants exposed to osmotic stressors. Plant monoterpenes were primarily synthesized through the 2-methyl-D-erythritol-4-phosphate pathway within plastids (33). Geranyl diphosphate served as a common precursor for all monoterpenes (34), which could be converted into β-pinene and limonene by various members of the terpene synthase/cyclase enzyme family (35). Although direct measurements of terpenoid volatile accumulation in wheat under saline-alkali stress are still limited, evidence suggest that wheat may cope with salt stress, at least in part, by modulating terpenoid biosynthetic pathways. Transcriptome analyses under salt stress have shown that pathways related to secondary metabolism, including terpenoid metabolism, are significantly enriched in the roots of salt-tolerant wheat genotypes (36). Similar stress-induced activation of terpenoid metabolic pathways has been reported in other plant species. According to report (14), an increased release of different terpenes in tomato plants with rising soil salt concentrations under salt stress conditions. It was found that there was an elevation in the content of certain terpenoids in salvia plants with increasing salt stress levels (37). Other studies also demonstrated that moderate drought could enhance plant terpenoids content (15). Stress treatment was carried out on Jinxuan tea tree varieties, and it was found that upregulation in gene expression related to the metabolic pathway involved in terpene carbon skeleton synthesis. This led to enhanced accumulation of precursors for terpenoid biosynthesis and increased volatile compound content (38). These findings may explain the elevated levels of these two monoterpenes under SA stress conditions.
Conversely, (E,E)-3,5-octadien-2-one content decreased markedly in SA-cultivated wheat, mirroring the suppression of 1-octen-3-one reported saline–alkali soil (19). This reduction likely stems from salt-triggered lipoxygenase (LOX) activation (39), which accelerates peroxidation of unsaturated fatty acids (e.g., linoleic/linolenic acid) (40) to generate hydroperoxide intermediates. These intermediates are preferentially metabolized to C6 aldehydes and alcohols (e.g., hexanal, 1-octen-3-ol) via hydroperoxide lyases (HPLs) and alcohol dehydrogenases (ADHs) (41), rather than to unsaturated ketones like (E,E)-3,5-octadien-2-one. Additionally, enzymatic reduction of (E,E)-3,5-octadien-2-one to its less volatile alcohol derivative may further deplete its concentration under oxidative stress. This metabolic rerouting reflects a broader reconfiguration of oxylipin networks under salinity, prioritizing compounds with lower flavor thresholds or stress-protective roles.
This study primarily focuses on the characteristics of VOCs in wheat kernels, while acknowledging that the VOCs of derived products, such as flour and cooked wheat-based foods, also warrant investigation. Future studies should (i) validate these biomarkers over many years and in multiple varieties to confirm their stability; (ii) analyze the effects of different saline-alkali stress environments and the spatial position of wheat grains on the characteristic VOCs of wheat aroma; and (ii) Conduct sensory-guided gas chromatography-olfactometry (GC-O) and recombination/omission experiments to directly confirm the contribution of the identified biomarkers to wheat aroma. These steps will help to further validate and refine the proposed biomarkers in future work.
5 Conclusion
In this study, the VOCs of five wheat varieties cultivated in soils with varying levels of salinity and alkalinity were analyzed by HS-SPME-GC-MS. Pairwise quantification revealed significant alterations (p < 0.05) in β-pinene, limonene, (E,E)-3,5-octadien-2-one, and butanoic acid, identifying them as diagnostic biomarkers for SA-cultivated wheat. Molecular docking further established β-pinene, limonene, and (E,E)-3,5-octadien-2-one as flavor determinants by exhibiting high-affinity binding (< -4.25 kcal/mol) to olfactory receptors, driven by hydrophobic interactions with residues Leu217, Ser213,Phe115, and Thr119 and hydrogen-bond networks involving Tyr269. Synergistically, ROAV analysis (> 1) identified these compounds as key biomarkers of “Wheat Aroma” in saline-alkali (SA) soil-cultivated wheat. These biomarkers serve as potential indicators, contributing to the understanding of the metabolic reprograming patterns in wheat under salinity-alkalinity stress.
StatementsData availability statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding authors.
Author contributions
JZ: Conceptualization, Writing – original draft, Formal analysis, Data curation. HW: Methodology, Writing – original draft, Data curation, Conceptualization. ZW: Investigation, Writing – original draft. BZ: Resources, Project administration, Funding acquisition, Writing – review & editing. HL: Supervision, Writing – review & editing, Investigation, QS: Writing – review & editing, Formal analysis, Writing – original draft, Methodology, Conceptualization.
Funding
The author(s) declared that financial support was received for this work and/or its publication. This work was supposed by the Cangzhou Drought-Alkali Wheat Special Project (no. 20241001005N), Key Project of Natural Science Foundation of Hebei Province (no. C2024110010), China Agriculture Research System of MOF and MARA (no. CARS-03), and Zhongyuan Research Center (no. ZYZX2023010108).
Conflict of interest
The author(s) declared that this work was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declared that generative AI was not used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
SA, Saline-alkali; VOCs, Volatile organic compounds; GC-IMS, Gas chromatography-ion mobility spectrometry; HS-SPME, Headspace solid phase microextraction; GC-MS, Gas chromatography-mass spectrometry; ROAV, Relative odor activity value; XHY, Xi HuayuanZJ Zhong Jie; PCA, Principal Components Analysis; LDA, Linear Discriminant Analysis
References
HasanuzzamanMNaharKAlamMMBhowmikPCHossainMARahmanMMet alPotential use of halophytes to remediate saline soils.Biomed Res Int. (2014) 2014:12. 10.1155/2014/589341
DongFZhaoLNQiJLiuYZ. Development status and countermeasures of drought alkali wheat industry in Cangzhou.Agricult Technol Commun. (2024) 2:40–5.
LiuYRenSWSunQQGuoBLZhangYQLiMet alGC-IMS determination of volatile organic compounds as potential indicators of wheat germination rate.Food Biosci. (2024) 57:10. 10.1016/j.fbio.2023.103535
LonginFBeckHGütlerHHeiligWKleinertMRappMet alAroma and quality of breads baked from old and modern wheat varieties and their prediction from genomic and flour-based metabolite profiles.Food Res. Int. (2020) 129:11. 10.1016/j.foodres.2019.108748
ChenXQuekSY. Free and glycosidically bound aroma compounds in fruit: biosynthesis, transformation, and practical control.Crit Rev Food Sci Nutr. (2023) 63:9052–73. 10.1080/10408398.2022.2064422
CzernyMSchieberleP. Important aroma compounds in freshly ground wholemeal and white wheat flour – Identifidation and quantitative changes during sourdough fermentation.J Agric Food Chem. (2002) 50:6835–40. 10.1021/jf020638p
QiangWLSunHJHuangXSongHLYingXWangXYet alAnalysis of aroma-active compounds in different wheat flour mill streams using dynamic headspace extraction and comprehensive two-dimensional gas chromatography-olfactometry-mass spectrometry.J Food Compos Anal. (2025) 140:10. 10.1016/j.jfca.2025.107241
YadavBJogawatARahmanMSNarayanOP. Secondary metabolites in the drought stress tolerance of crop plants: a review.Gene Rep. (2021) 23:14. 10.1016/j.genrep.2021.101040
BourgouSBettaiebISaidaniMMarzoukB. Fatty acids, essential oil, and phenolics modifications of black cumin fruit under NaCl stress conditions.J Agric Food Chem. (2010) 58:12399–406. 10.1021/jf103415q
HossainMSPersickeMElSayedAIKalinowskiJDietzKJ. Metabolite profiling at the cellular and subcellular level reveals metabolites associated with salinity tolerance in sugar beet.J Exp Bot. (2017) 68:5961–76. 10.1093/jxb/erx388
KnieperMViehhauserADietzKJ. Oxylipins and reactive carbonyls as regulators of the plant redox and reactive oxygen species network under stress.Antioxidants. (2023) 12:814. 10.3390/antiox12040814
LiuXFengYLiSLiDYuJZhaoZ. Jasmonate-induced MdMYC2 improves fruit aroma during storage of ‘Ruixue’ apple based on transcriptomic, metabolic and functional analyses.LWT-Food Sci Technol. (2023) 185:115168. 10.1016/j.lwt.2023.115168
ChrysargyrisAPapakyriakouEPetropoulosSATzortzakisN. The combined and single effect of salinity and copper stress on growth and quality of Mentha spicata plants.J Hazard Mater. (2019) 368:584–93. 10.1016/j.jhazmat.2019.01.058
TomescuDSumalanRCopoloviciLCopoloviciD. The influence of soil salinity on volatile organic compounds emission and photosynthetic parameters of Solanum lycopersicum L. varieties.Open Life Sci. (2017) 12:135–42. 10.1515/biol-2017-0016
TurtolaSManninenAMRikalaRKainulainenP. Drought stress alters the concentration of wood terpenoids in Scots pine and Norway spruce seedlings.J Chem Ecol. (2003) 29:1981–95. 10.1023/a:1025674116183
LiuXHDongFBLiYCLuFWangBTZhouTCet alImpact of mild field drought on the aroma profile and metabolic pathways of fresh tea (Camellia sinensis) leaves using HS-GC-IMS and HS-SPME-GC-MS.Foods. (2024) 13:23. 10.3390/foods13213412
LiuMLYuMYaoYXZhaiHTangMLGaoZet alEffect of seawater irrigation on the sugars, organic acids, and volatiles in ‘Reliance’, Grape.Horticulturae. (2022) 8:17. 10.3390/horticulturae8030248
GayFMaravalIRoquesSGunataZBoulangerRAudebertAet alEffect of salinity on yield and 2-acetyl-1-pyrroline content in the grains of three fragrant rice cultivars (Oryza sativa L.) in Camargue (France).Field Crop Res. (2010) 117:154–60. 10.1016/j.fcr.2010.02.008
SunQQZhangRLiuYNiuLYLiuHYRenPFet alInsights into “wheat aroma”: analysis of volatile components in wheat grains cultivated in saline-alkali soil.Food Res Int. (2024) 183:8. 10.1016/j.foodres.2024.114211
ChenJWangWWJinJHLiHChenFFeiYet alCharacterization of the flavor profile and dynamic changes in Chinese traditional fish sauce (Yu-lu) based on electronic nose, SPME-GC-MS and HS-GC-IMS.Food Res Int. (2024) 192:12. 10.1016/j.foodres.2024.114772
XiBNZhangJJXuXLiCShuYZhangYet alCharacterization and metabolism pathway of volatile compounds in walnut oil obtained from various ripening stages via HS-GC-IMS and HS-SPME-GC-MS.Food Chem. (2024) 435:11. 10.1016/j.foodchem.2023.137547
De FlaviisRSacchettiGMastrocolaD. Wheat classification according to its origin by an implemented volatile organic compounds analysis.Food Chem. (2021) 341:10. 10.1016/j.foodchem.2020.128217
JiaXWGaoYXiHCuiCYangXHeBJet alA flavor imitation method for Osmanthus aroma based on molecular docking screening and odor activity value analysis.LWT-Food Sci Technol. (2025) 223:9. 10.1016/j.lwt.2025.117697
BiJCLiYYangZLinZYChenFSLiuSXet alEffect of different cooking times on the fat flavor compounds of pork belly.J Food Biochem. (2022) 46:12. 10.1111/jfbc.14184
YuRXZhouHJXiongMChenJQWangSPXuWet alIntegrated UHPLC-Q-Exactive-MS/MS metabolomics and network pharmacology to explore the potential mechanism of obesity prevention in Pleurotus eryngii treated with different cooking procedures.J Future Foods. (2026) 6:117–30. 10.1016/j.jfutfo.2024.08.012
TianDHuangGHDengXJRenLYuJHuangYet alThe aroma compounds contributing to the characteristic flavour of ripe Pu-erh tea and their molecular mechanisms of interaction with olfactory receptors.LWT-Food Sci Technol. (2025) 224:14. 10.1016/j.lwt.2025.117808
WangZChangXYHaoWJWangYHuangMQSunBGet alStudy on interaction of aromatic substances and correlation between electroencephalogram correlates of odor perception in light flavor Baijiu.J Agric Food Chem. (2024) 72:16519–29. 10.1021/acs.jafc.4c02979
BaiBLiJZhangZZhangYBoTZhangJet alCharacterization of the flavor profile of Huangjiu brewed with Polygonatum sibiricum and Broomcorn millet using HS-SPME-GC*GC-TOF-MS, GC-IMS, intelligent sensory and molecular docking approaches.Food Chem. (2025) 492(Pt 1):145300. 10.1016/j.foodchem.2025.145300
ZengXFXiaWSJiangQXYangF. Chemical and microbial properties of Chinese traditional low-salt fermented whole fish product Suan yu.Food Control. (2013) 30:590–5. 10.1016/j.foodcont.2012.07.037
SebzalliYMWangXZ. Knowledge discovery from process operational data using PCA and fuzzy clustering.Eng Appl Artif Intell. (2001) 14:607–16. 10.1016/s0952-1976(01)00032-x
BirchANPetersenMAHansenÅ. Aroma of wheat bread crumb.Cereal Chem. (2014) 91:105–14. 10.1094/cchem-06-13-0121-rw
LiuXSLiuJBYangZMSongHLLiuYZouTT. Aroma-active compounds in jinhua ham produced with different fermentation periods.Molecules. (2014) 19:19097–113. 10.3390/molecules191119097
YuFNAUtsumiR. Diversity, regulation, and genetic manipulation of plant mono- and sesquiterpenoid biosynthesis.Cell Mol Life Sci. (2009) 66:3043–52. 10.1007/s00018-009-0066-7
OguraKKoyamaT. Enzymatic aspects of isoprenoid chain elongation.Chem Rev. (1998) 98:1263–76. 10.1021/cr9600464
CordobaESalmiMLeónP. Unravelling the regulatory mechanisms that modulate the MEP pathway in higher plants.J Exp Bot. (2009) 60:2933–43. 10.1093/jxb/erp190
AmirbakhtiarNIsmailiAGhaffariMRNazarian FirouzabadiFShobbarZS. Transcriptome response of roots to salt stress in a salinity-tolerant bread wheat cultivar.PLoS One. (2019) 14:e0213305. 10.1371/journal.pone.0213305
ValifardMMohsenzadehSKholdebarinBRowshanVNiaziAMoghadamA. Effect of salt stress on terpenoid biosynthesis in Salvia mirzayanii: from gene to metabolite.J Horticult Sci Biotechnol. (2019) 94:389–99. 10.1080/14620316.2018.1505443
ShiJMaCYQiDDLvHPYangTPengQHet alTranscriptional responses and flavor volatiles biosynthesis in methyl jasmonate-treated tea leaves.BMC Plant Biol. (2017) 15:233. 10.1186/s12870-017-1076-5
WuCAZhangMLiangYFZhangLDiaoXM. Salt stress responses in foxtail millet: Physiological and molecular regulation.Crop J. (2023) 11:1011–21. 10.1016/j.cj.2023.06.001
LiangXJWangYJLiYKAnWHeXRChenYZet alWidely-targeted metabolic profiling in Lycium barbarum fruits under salt-alkaline stress uncovers mechanism of salinity tolerance.Molecules. (2022) 27:18. 10.3390/molecules27051564
SuTFChenYNLiuHHGaoYQGuoJWLiYNet alThe biosynthesis of 1-octene-3-ol by a multifunctional fatty acid dioxygenase and hydroperoxide lyase in Agaricus bisporus.J Fungi. (2022) 8:11. 10.3390/jof8080827
Summary
Keywords
gas chromatography-mass spectrometry (GC-MS), molecular docking, relative odor activity values (ROAVs), saline-alkali (SA) soil, volatile organic compounds (VOCs), wheat aroma
Citation
Zhang J, Wang H, Wang Z, Zhang B, Li H and Sun Q (2026) Wheat aroma biomarkers induced by saline-alkali soil: based on HS-SPME-GC-MS and molecular docking. Front. Nutr. 13:1788319. doi: 10.3389/fnut.2026.1788319
Received
15 January 2026
Revised
03 February 2026
Accepted
16 February 2026
Published
04 March 2026
Volume
13 – 2026
Edited by
Nicoleta-Aurelia Chira, Polytechnic University of Bucharest, Romania
Reviewed by
Ufaq Fayaz, Sher-e-Kashmir University of Agricultural Sciences and Technology of Kashmir, India
Jinshui Wang, Henan University of Technology, China
Updates

Check for updates
Copyright
© 2026 Zhang, Wang, Wang, Zhang, Li and Sun.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Qianqian Sun, sunqianqian@caas.cnHuijing Li, huijingli2002@163.comBo Zhang, zjzb1978@126.com
†These authors have contributed equally to this work and share first authorship
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.







