Vitamin D Toxicity Market Summary
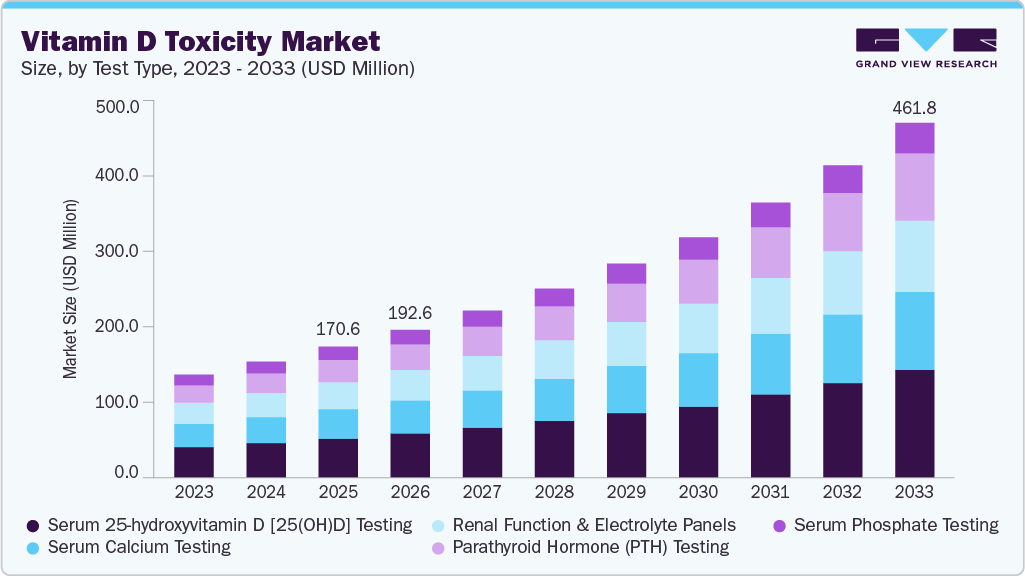
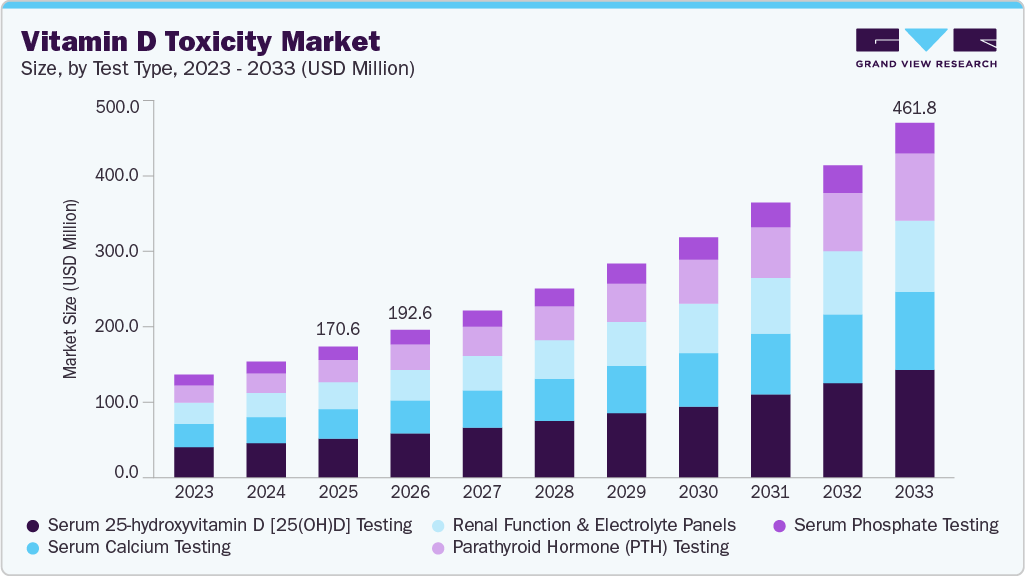
The global vitamin D toxicity market size was estimated at USD 170.58 million in 2025 and is expected to reach by USD 461.83 million by 2033, growing at a CAGR of 13.31% from 2026 to 2033. The increasing use of high-dose vitamin D supplements, rising awareness of vitamin D deficiency, and the growing adoption of preventive health practices drive the market growth.
Key Market Trends & Insights
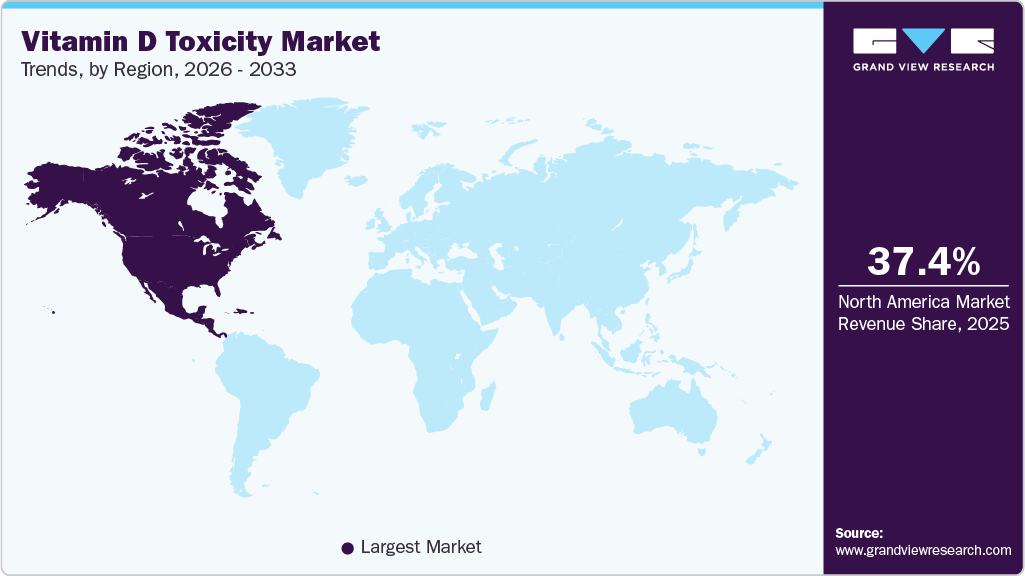
The North America vitamin D toxicity market accounted for the largest global revenue share of 37.43% in 2025.
The U.S. vitamin D toxicity industry led North America with the largest revenue share in 2025.
Based on test type, the serum 25-hydroxyvitamin D [25(OH)D] testing segment dominated the global market with the largest revenue share of 29.90% 2025.
Based on patient group, the adult population segment held the largest revenue share of 66.46% in 2025.
Based on end use, the independent diagnostic laboratories segment held the largest revenue share of 36.38% in 2025.
Market Size & Forecast
2025 Market Size: USD 170.58 Million
2033 Projected Market Size: USD 461.83 Million
CAGR (2026-2033): 13.31%
North America: Largest market in 2025
Excessive intake of vitamin D can lead to hypercalcemia, which may result in symptoms such as nausea, renal dysfunction, bone pain, and cardiovascular complications. As supplement consumption and therapeutic dosing continue to increase, the need for routine laboratory monitoring has become more critical to detect elevated vitamin D levels early and prevent associated metabolic and systemic complications.
The growth of the vitamin D toxicity diagnostics market is driven by the increasing consumption of high-dose vitamin D supplements and fortified nutritional products worldwide. Rising awareness of vitamin D deficiency, combined with preventive health trends, has led to widespread supplementation, often without medical supervision. Excessive intake can lead to hypervitaminosis D and hypercalcemia, thereby increasing the need for routine diagnostic monitoring. According to the National Institutes of Health, prolonged intake above recommended upper limits may cause adverse health effects, underscoring the importance of laboratory testing to monitor vitamin D levels and prevent toxicity-related complications.
The expanding adoption of preventive healthcare and routine wellness screening is further supporting market growth. Vitamin D testing is increasingly incorporated into annual health checkups, metabolic panels, and preventive screening programs, particularly in regions with high deficiency prevalence. Large diagnostic service providers have expanded wellness testing portfolios that include vitamin D screening as part of comprehensive health assessments, reflecting a shift toward proactive health management. This growing emphasis on early detection and preventive care is increasing testing volumes and supporting demand for toxicity monitoring.
Technological advancements in automated immunoassay analyzers and laboratory platforms are improving the accuracy, efficiency, and scalability of vitamin D testing. High-throughput systems enable precise measurement of serum 25-hydroxyvitamin D concentrations, which is essential for identifying toxic levels and guiding clinical intervention. For instance, Abbott developed automated vitamin D assays for its ARCHITECT and Alinity platforms to support high-volume clinical testing and improved laboratory workflow efficiency. These innovations are enhancing diagnostic reliability and enabling healthcare providers to monitor patients more effectively.
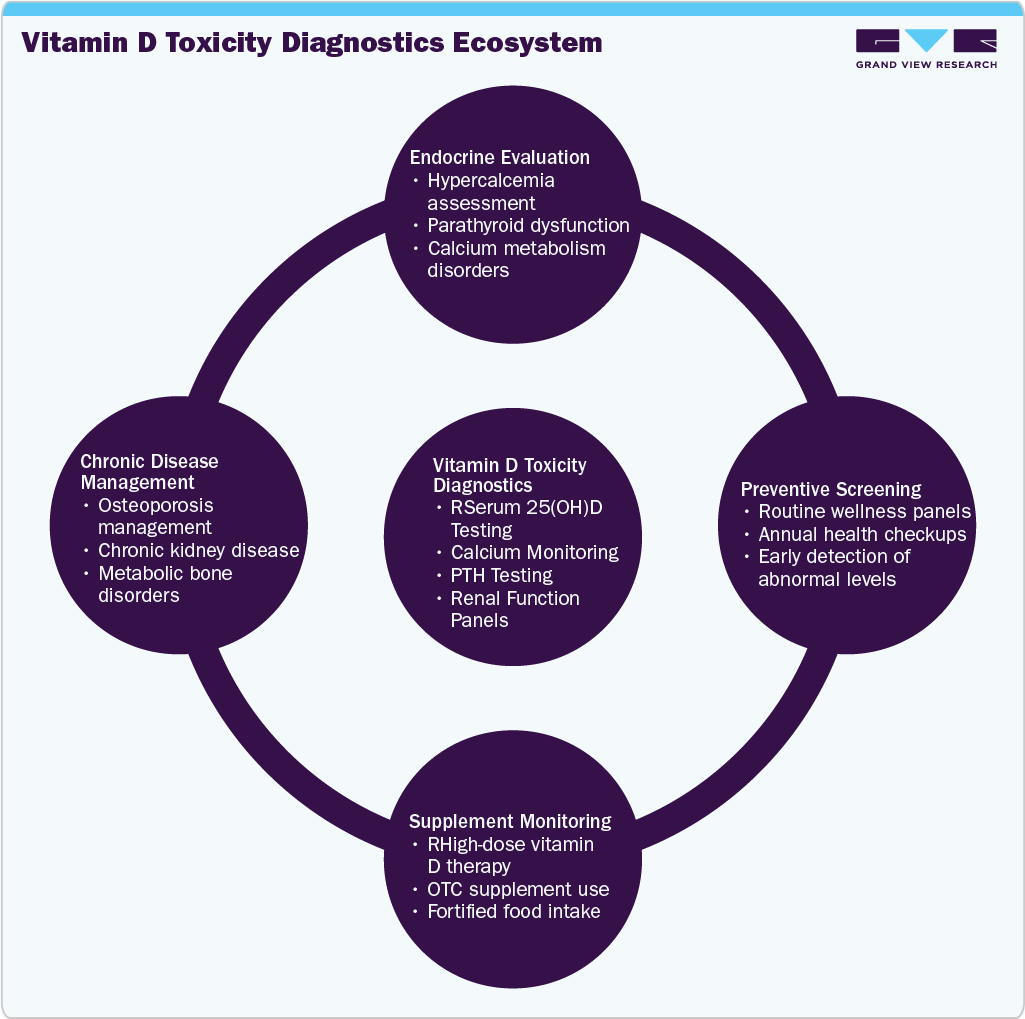
The increasing prevalence of osteoporosis, chronic kidney disease, and metabolic disorders is elevating the clinical importance of vitamin D monitoring. Patients receiving long-term supplementation or therapeutic high-dose vitamin D therapy require periodic testing to avoid complications such as hypercalcemia, nephrocalcinosis, and renal dysfunction. Aging populations and a growing focus on bone health management are further expanding the patient base requiring monitoring. Clinical laboratories are increasingly utilizing advanced immunoassay and mass spectrometry methods to support endocrine and metabolic testing, strengthening the role of vitamin D diagnostics in chronic disease management.
Despite growing demand, the industry’s expansion into vitamin D toxicity may be constrained by variability in testing standards, limited awareness of vitamin D toxicity risks, and cost considerations associated with routine monitoring. Differences in assay methodologies and reference ranges across laboratories can complicate the interpretation of results and clinical decision-making. In many regions, vitamin D testing is primarily performed to detect deficiency rather than toxicity, which may limit awareness of the risks of hypervitaminosis among both patients and healthcare providers. In addition, preventive testing may not be fully reimbursed in certain healthcare systems, restricting adoption in price-sensitive markets and slowing the uptake of routine toxicity monitoring.
Market Concentration & Characteristics
The degree of innovation in the vitamin D toxicity diagnostics market is moderate to high, driven by advancements in automated immunoassay platforms, assay standardization, and high-precision reference testing methods. Clinical laboratories are increasingly adopting chemiluminescent immunoassays and liquid chromatography–tandem mass spectrometry (LC-MS/MS) to improve the accuracy of measuring serum 25-hydroxyvitamin D levels. In 2024, efforts led by the Centers for Disease Control and Prevention Vitamin D Standardization-Certification Program continued to support assay harmonization and improve comparability of test results across laboratories. These innovations are strengthening diagnostic reliability and supporting the timely detection and management of vitamin D toxicity.
Mergers, acquisitions, and strategic partnerships in the vitamin D diagnostics space are moderate, reflecting efforts by diagnostic manufacturers to expand clinical chemistry portfolios and strengthen endocrine testing capabilities. Industry participants continue to invest in expanding automated testing platforms and assay menus to support the monitoring of metabolic and endocrine disorders. Strategic collaborations between diagnostic companies and laboratory networks are improving test accessibility and workflow efficiency, enabling broader adoption of vitamin D testing in hospital and reference laboratories.
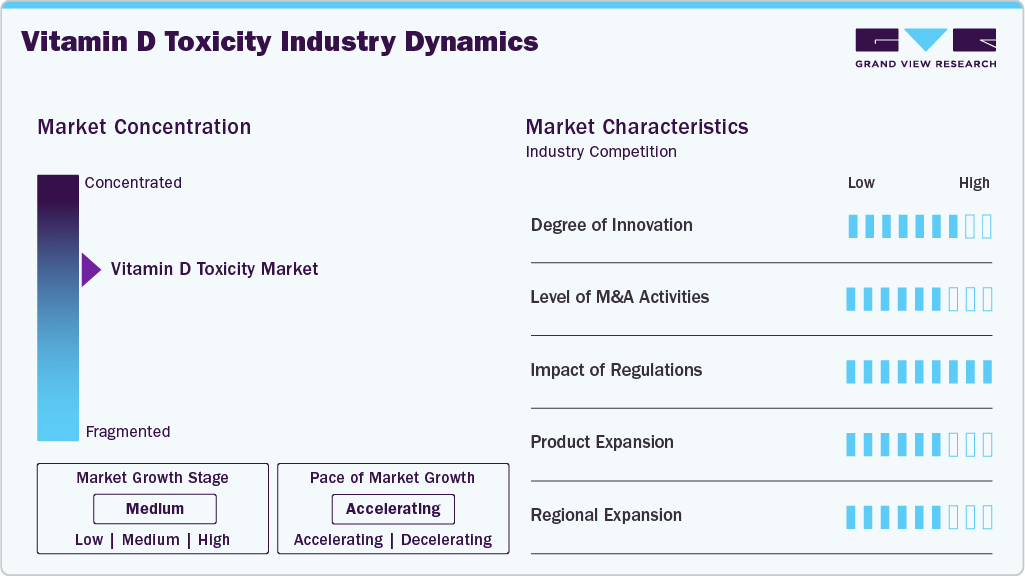
The regulatory impact on the vitamin D toxicity diagnostics market is high, as regulatory approvals and quality standards directly influence assay availability, performance validation, and laboratory adoption. Regulatory agencies and standardization bodies emphasize accuracy, traceability, and harmonization of vitamin D testing methods to ensure reliable clinical decision-making. Programs such as the CDC’s Vitamin D Standardization initiatives promote alignment with reference measurement procedures, strengthening confidence in test results and supporting wider clinical use.
Product expansion in the vitamin D toxicity market is moderate to high, with manufacturers enhancing assay menus and improving analyzer compatibility to support endocrine and metabolic testing. In 2024, Beckman Coulter expanded the availability of its Access 25(OH) Vitamin D Total assay across automated immunoassay systems, improving laboratory workflow efficiency and testing throughput. Expanding test menus to include complementary biomarkers, such as calcium, parathyroid hormone (PTH), and renal function panels, is enabling comprehensive assessment of vitamin D toxicity and related metabolic complications.
Regional expansion of vitamin D testing services is moderate, with strong adoption in North America and Europe and growing uptake in Asia Pacific and the Middle East. Increasing awareness of vitamin D deficiency, rising supplement consumption, and expanding preventive healthcare programs are driving global testing demand. In emerging markets, improvements in laboratory infrastructure and diagnostic capacity are enhancing access to endocrine testing services, although variability in reimbursement coverage and awareness levels continues to influence adoption rates.
Test Type Insights
The serum 25-hydroxyvitamin D [25(OH)D] testing segment dominated the vitamin D toxicity market, accounting for the largest revenue share of 29.90% in 2025. The segment’s dominance is primarily attributed to its status as the clinical gold standard for assessing vitamin D status and detecting toxic concentrations. Measurement of serum 25(OH)D provides the most reliable indicator of total vitamin D exposure from sunlight, diet, and supplementation, making it essential for diagnosing hypervitaminosis D and guiding treatment decisions. Increasing physician awareness of vitamin D over-supplementation risks, along with the growing inclusion of vitamin D testing in routine metabolic panels and wellness screening programs, continues to drive high testing volumes. In addition, automated immunoassay platforms and standardized testing protocols have improved accuracy, throughput, and laboratory efficiency, further supporting widespread adoption of 25(OH)D testing.
The Parathyroid Hormone (PTH) testing segment is projected to experience the fastest CAGR of 14.84% during the forecast period. Rising clinical recognition of the relationship between vitamin D toxicity, calcium imbalance, and parathyroid function is a key factor supporting growth in this segment. PTH testing plays a critical role in differentiating vitamin D-induced hypercalcemia from primary hyperparathyroidism and other metabolic disorders, enabling more precise diagnosis and patient management. Increasing use of comprehensive metabolic panels in endocrine and renal care, particularly among patients with chronic kidney disease and osteoporosis, is further driving demand. As clinicians adopt integrated diagnostic approaches to evaluate calcium metabolism and endocrine function, PTH testing is expected to grow rapidly.
Patient Group Insights
In 2025, the adult population segment led the vitamin D toxicity industry, accounting for the largest share of 66.46% and is anticipated to grow at the fastest CAGR over the forecast period. The segment’s dominance is attributed to the high prevalence of vitamin D supplementation among adults for bone health, immune support, and chronic disease management. Adults, particularly older individuals and postmenopausal women, are more likely to receive high-dose supplementation for osteoporosis prevention and metabolic health, increasing the risk of excessive intake and toxicity. In addition, routine wellness screenings and chronic disease monitoring programs often include vitamin D testing, which contributes to higher testing volumes. The growing burden of conditions such as osteoporosis, chronic kidney disease, and endocrine disorders further supports continued demand for diagnostic monitoring in the adult population.
The pediatric population segment is expected to grow at a lucrative CAGR over the forecast period. Growth is driven by increased awareness of the importance of vitamin D supplementation in infants and children, particularly for bone development and rickets prevention. While supplementation is widely recommended, improper dosing and unsupervised use of high-strength formulations have raised concerns regarding toxicity risks. Pediatricians are increasingly recommending laboratory monitoring in cases of prolonged supplementation or suspected overdose. Additionally, rising parental awareness and improved pediatric screening practices are expected to support increased testing and monitoring in this population segment.
End Use Insights
In 2025, the independent diagnostic laboratories segment led the vitamin D toxicity market, accounting for 36.38% of the revenue share, and is anticipated to grow at the fastest CAGR over the forecast period. The segment’s dominance is primarily driven by the high testing volumes handled by reference and independent laboratories, which serve hospitals, clinics, and preventive health programs. These laboratories are equipped with high-throughput automated analyzers and standardized assay platforms, enabling efficient processing of large sample volumes for serum 25-hydroxyvitamin D, calcium, and related metabolic markers. Increasing inclusion of vitamin D testing in routine wellness panels, corporate health screening programs, and preventive diagnostics packages continues to support demand. Additionally, partnerships between diagnostic chains and healthcare providers are improving accessibility and turnaround times, further strengthening the role of independent laboratories.
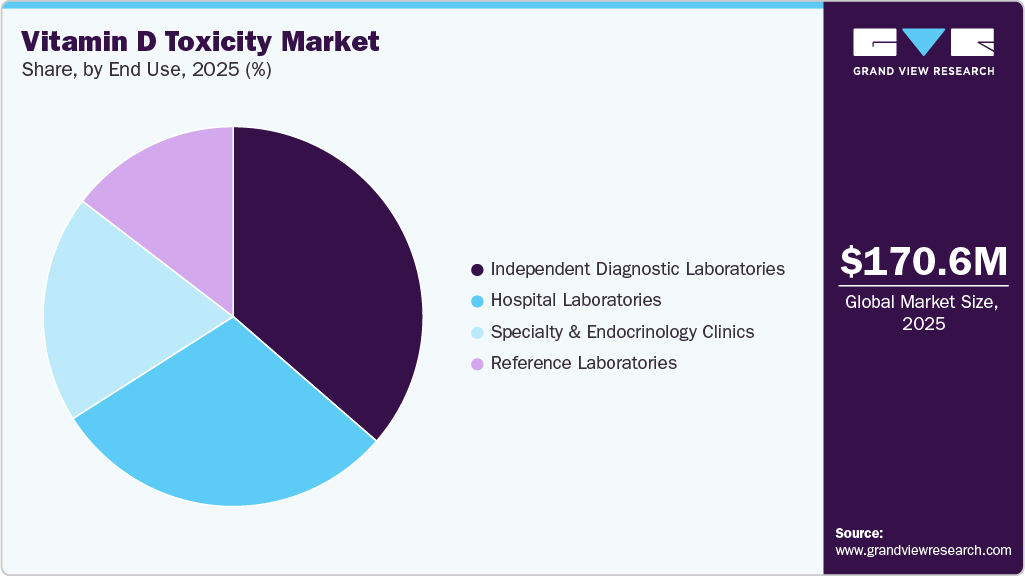
The specialty and endocrinology clinics segment is expected to grow lucratively over the forecast period. Growth of the segment is driven by the increasing clinical management of endocrine and metabolic disorders associated with vitamin D imbalance, including hypercalcemia, osteoporosis, and parathyroid dysfunction. Endocrinologists frequently rely on comprehensive biochemical assessments, including vitamin D, calcium, and parathyroid hormone testing, to guide diagnosis and treatment decisions. Rising referrals for metabolic bone disorders, chronic kidney disease–related mineral imbalance, and complex endocrine conditions are contributing to increased testing within specialty settings. As awareness of vitamin D toxicity and calcium metabolism disorders continues to expand, specialty and endocrinology clinics are expected to play an increasingly important role in diagnostic monitoring and patient management.
Regional Insights
North America Vitamin D Toxicity Market Trends
The North America vitamin D toxicity industry held the largest global revenue share of 37.44% in 2025. Growth in the region is supported by high consumption of dietary supplements, increasing awareness of vitamin D deficiency and the risks of over-supplementation, and strong adoption of preventive health screening. Well-established healthcare infrastructure, widespread access to laboratory testing services, and routine inclusion of vitamin D testing in wellness panels have accelerated diagnostic uptake. The presence of major diagnostic manufacturers and large reference laboratory networks further strengthens regional market growth.
U.S. Vitamin D Toxicity Market Trends
The U.S. vitamin D toxicity industry held the largest share of North America in 2025, driven by widespread supplement use, strong consumer awareness, and advanced diagnostic capabilities. Preventive healthcare practices and routine wellness testing programs often include vitamin D screening, which contributes to high testing volumes. In addition, continued research initiatives supported by the National Institutes of Health emphasize safe supplementation practices and metabolic health monitoring. The presence of leading diagnostic laboratories and automated testing platforms supports early detection and ongoing monitoring of abnormal vitamin D levels.
Europe Vitamin D Toxicity Market Trends
The Europe vitamin D toxicity diagnostics industry is experiencing significant growth driven by rising vitamin D supplementation, aging populations, and expanding preventive healthcare programs. Many European countries are strengthening initiatives to screen for metabolic and bone health to support early detection of deficiencies and prevent complications associated with excessive intake. Government-supported health screening programs and strong laboratory networks are improving access to vitamin D testing services across the region.
The UK vitamin D toxicity market is expanding as clinical focus on bone health, osteoporosis prevention, and safe vitamin D supplementation increases. The National Health Service (NHS) promotes vitamin D supplementation for at-risk populations while emphasizing appropriate monitoring when high doses are used. Growing awareness of metabolic health and preventive screening practices is encouraging routine vitamin D testing, particularly among elderly populations and individuals with chronic health conditions.
The vitamin D toxicity market in Germany is growing steadily, supported by advanced laboratory infrastructure and strong preventive healthcare practices. The country’s well-established diagnostic laboratory network enables efficient metabolic and endocrine testing. Increasing awareness of bone health, aging-related disorders, and mineral metabolism is contributing to higher testing rates and supporting market expansion.
Asia Pacific Vitamin D Toxicity Market Trends
Asia Pacific is projected to witness the fastest growth in the vitamin D toxicity diagnostics market. Growth is driven by greater awareness of vitamin D deficiency, expanding supplement use, and improved healthcare infrastructure. Rising middle-class populations, urban lifestyles with limited sunlight exposure, and growing preventive health awareness are encouraging routine vitamin D testing. Governments and healthcare providers in the region are strengthening diagnostic capacity and preventive screening programs.
China’s vitamin D toxicity market is expanding due to rapid healthcare modernization, increasing supplement consumption, and growing adoption of preventive health screening. Investments in diagnostic infrastructure and laboratory automation are improving access to metabolic testing. Increasing awareness of bone health and nutritional supplementation is supporting routine vitamin D monitoring across urban healthcare settings.
Latin America Vitamin D Toxicity Market Trends
The Latin America vitamin D toxicity diagnostics market is experiencing steady growth driven by increasing awareness of nutritional health, expanding preventive care programs, and improved access to diagnostic services. Countries such as Brazil and Argentina are strengthening healthcare infrastructure and promoting metabolic health screening, supporting broader adoption of vitamin D testing.
Middle East and Africa Vitamin D Toxicity Market Trends
The Middle East & Africa vitamin D toxicity industry is expanding due to rising healthcare investments and growing awareness of vitamin D deficiency and supplementation practices. The high prevalence of vitamin D deficiency in sun-rich regions, combined with increased supplementation, has underscored the importance of diagnostic monitoring. Improvements in laboratory infrastructure and preventive health initiatives are supporting the adoption of testing.
Saudi Arabia’s vitamin D toxicity market is expanding due to increased healthcare investment and ongoing modernization. The high prevalence of vitamin D deficiency, lifestyle factors limiting sun exposure, and increased supplementation have heightened the need for routine monitoring. Government initiatives promoting preventive healthcare and the expansion of diagnostic services are expected to support increased adoption of vitamin D testing.
Key Vitamin D Toxicity Company Insights
The competitive landscape of the vitamin D toxicity diagnostics market is characterized by strong participation from established in-vitro diagnostics manufacturers and clinical laboratory service providers focused on expanding endocrine and metabolic testing capabilities. Leading companies are investing in advanced immunoassay platforms, assay standardization, and high-throughput automation to support accurate measurement of serum 25-hydroxyvitamin D and related biomarkers. Market participants are also strengthening their positions through portfolio expansion, laboratory network partnerships, and workflow integration solutions that improve testing efficiency and clinical decision support. In addition, the growing emphasis on preventive healthcare and routine wellness screening has encouraged diagnostic laboratories to expand vitamin D testing services as part of comprehensive metabolic panels.
Key Vitamin D Toxicity Companies:
The following key companies have been profiled for this study on the vitamin D toxicity market.
F. Hoffmann-La Roche Ltd (Roche Diagnostics)
Abbott
Siemens Healthineers AG
Beckman Coulter, Inc.
Thermo Fisher Scientific, Inc.
DiaSorin S.p.A.
bioMérieux
QuidelOrtho Corporation
Randox Laboratories Ltd.
Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
Sysmex Corporation
Agilent Technologies, Inc.
Recent Developments
In April 2025, Roche Diagnostics announced expanded global adoption of its Elecsys® Vitamin D Total II assay across Cobas analyzer platforms, improving laboratory standardization and enabling high-throughput testing for metabolic and endocrine assessment. The assay supports accurate monitoring of vitamin D levels, helping clinicians detect deficiency and toxicity.
In September 2024, Siemens Healthineers enhanced its Atellica IM immunoassay portfolio with workflow and automation improvements designed to support high-volume endocrine testing, including vitamin D measurement. The enhancements aim to improve turnaround times and laboratory efficiency in routine metabolic testing.
In August 2024, Beckman Coulter expanded global availability of its Access 25(OH) Vitamin D Total assay on automated immunoassay systems, enabling laboratories to enhance throughput and support routine monitoring of vitamin D levels in endocrine and metabolic care.
Vitamin D Toxicity Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 192.60 million
Revenue forecast in 2033
USD 461.83 million
Growth rate
CAGR of 13.31% from 2026 to 2033
Actual data
2021 – 2024
Forecast period
2026 – 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test type, patient group, end use, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
F. Hoffmann-La Roche Ltd (Roche Diagnostics); Abbott; Siemens Healthineers AG; Beckman Coulter, Inc.; Thermo Fisher Scientific, Inc.; DiaSorin S.p.A.; bioMérieux; QuidelOrtho Corporation; Randox Laboratories Ltd.; Shenzhen Mindray Bio-Medical Electronics Co., Ltd.; Sysmex Corporation; Agilent Technologies, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Vitamin D Toxicity Market Report Segmentation
This report forecasts revenue growth at the global, regional & country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the vitamin D toxicity market report based on test type, patient group, end use, and region:
Test Type Outlook (Revenue, USD Million, 2021 – 2033)
Serum 25-hydroxyvitamin D [25(OH)D] Testing
Serum Calcium Testing
Serum Phosphate Testing
Parathyroid Hormone (PTH) Testing
Renal Function and Electrolyte Panels
Patient Group Outlook (Revenue, USD Million, 2021 – 2033)
Pediatric Population
Adult Population
Others
End Use Outlook (Revenue, USD Million, 2021 – 2033)
Regional Outlook (Revenue, USD Million, 2021 – 2033)
North America
Europe
UK
Germany
France
Italy
Spain
Norway
Denmark
Sweden
Denmark
Rest of Europe
Asia Pacific
Japan
China
India
Australia
South Korea
Thailand
Rest of Asia Pacific
Latin America
Brazil
Argentina
Rest of Latin America
Middle East & Africa
Frequently Asked Questions About This Report
b. North America dominated the vitamin D toxicity market with a share of 37.43% in 2025. This is attributable to high consumption of dietary supplements, increasing awareness of vitamin D deficiency and over-supplementation risks, and strong adoption of preventive health screening.
b. Some key players operating in the vitamin D toxicity market include F. Hoffmann-La Roche Ltd (Roche Diagnostics), Abbott, Siemens Healthineers AG, Beckman Coulter, Inc., Thermo Fisher Scientific, Inc., DiaSorin S.p.A., bioMérieux, QuidelOrtho Corporation, Randox Laboratories Ltd., Shenzhen Mindray Bio-Medical Electronics Co., Ltd., Sysmex Corporation, Agilent Technologies, Inc.
b. Key factors that are driving the market growth include increasing use of high-dose vitamin D supplements, rising awareness of vitamin D deficiency, and the growing adoption of preventive health practices.
b. The global vitamin D toxicity market size was estimated at USD 170.58 million in 2025 and is expected to reach USD 192.60 million in 2026.
b. The global vitamin D toxicity market is expected to grow at a compound annual growth rate of 13.31% from 2026 to 2033 to reach USD 461.83 million by 2033.