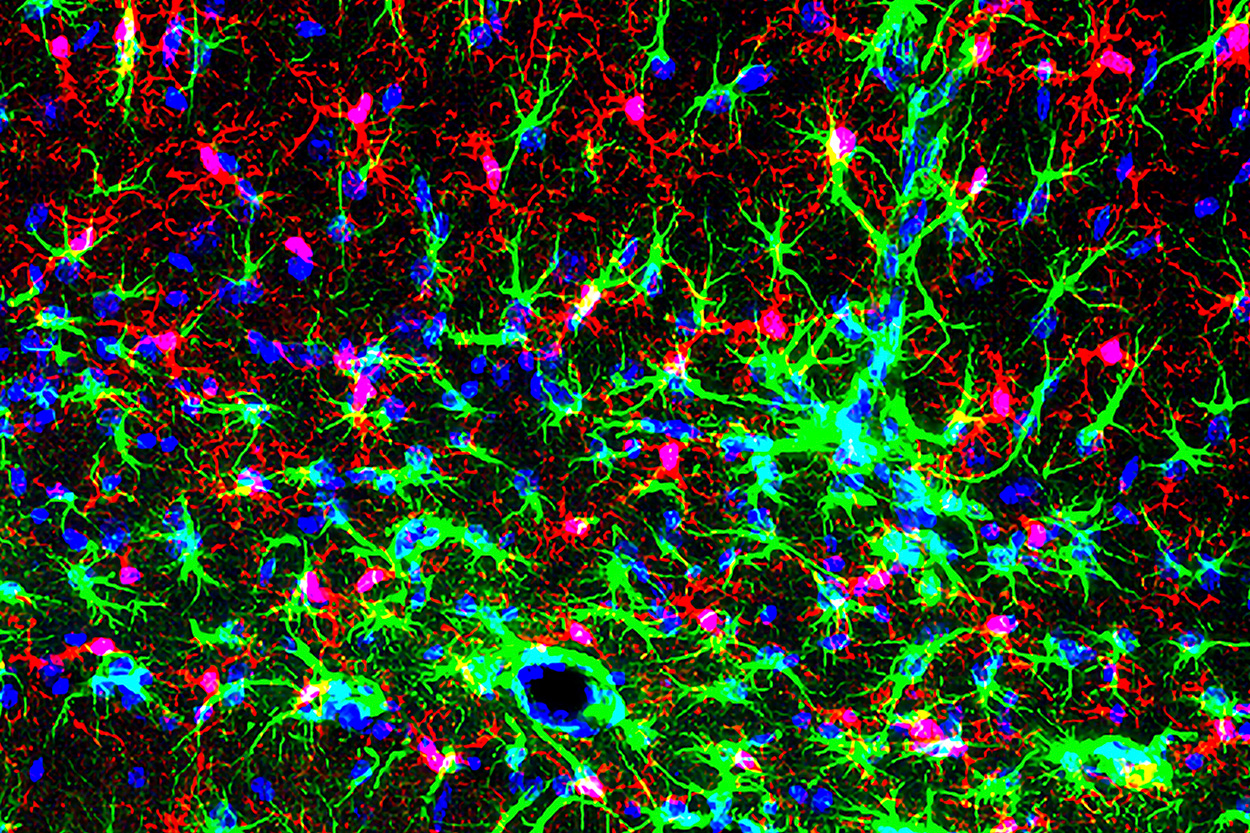
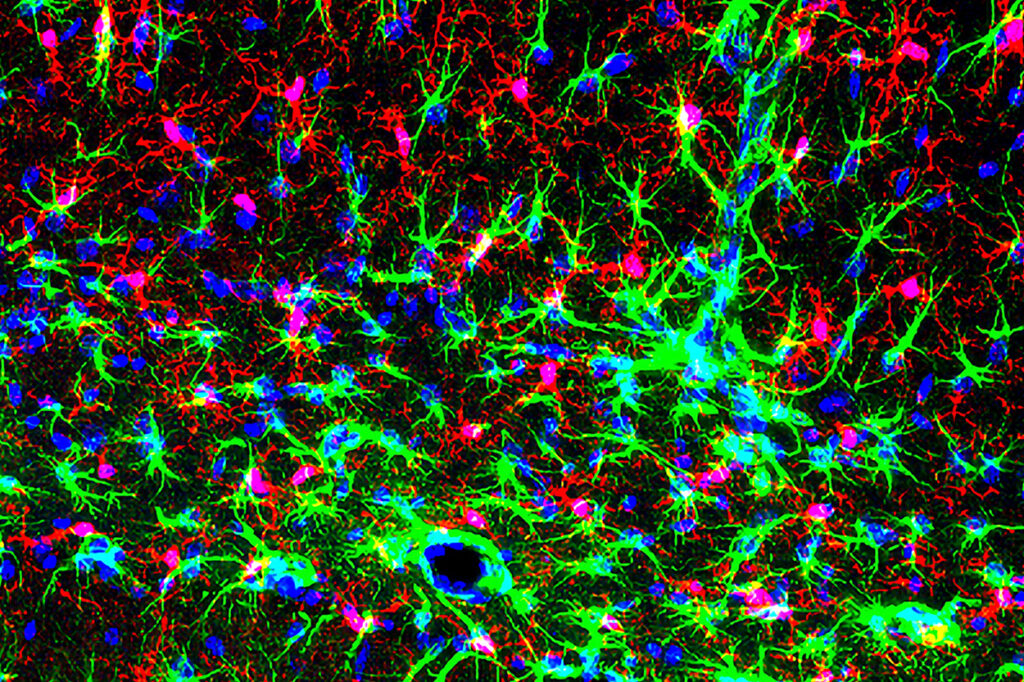
 The image shows inflammation-induced increases in markers for glial cells including microglia (red), astrocytes (green), as well as the nuclei of all cells (blue) in the mouse hippocampus. These inflammation-induced increases in inflammatory markers did not occur in mice lacking Orai1 in microglia. Courtesy of Murali Prakriya, PhD.
The image shows inflammation-induced increases in markers for glial cells including microglia (red), astrocytes (green), as well as the nuclei of all cells (blue) in the mouse hippocampus. These inflammation-induced increases in inflammatory markers did not occur in mice lacking Orai1 in microglia. Courtesy of Murali Prakriya, PhD.
Northwestern Medicine scientists have discovered how calcium signaling channels in microglia — the primary immune cells of the brain — regulate neuroinflammation and promote the development of behaviors associated with affective mood disorders, according to a recent study published in Science Signaling.
The findings suggest that inhibiting Orai1 calcium channels in microglia could modulate chronic neuroinflammation and may represent a potential therapeutic target for treating brain disorders, including depression and other neurodegenerative diseases, said Murali Prakriya, PhD, the Magerstadt Professor of Pharmacology and senior author of the study.
 Murali Prakriya, PhD, the Magerstadt Professor of Pharmacology, was senior author of the study published in Science Signaling.
Murali Prakriya, PhD, the Magerstadt Professor of Pharmacology, was senior author of the study published in Science Signaling.
“This could open up some interesting possibilities for therapeutic interventions for depression-like behaviors or maladaptive changes in motivational behaviors down the road where microglia are known to play a really important role,” said Prakriya, who is also a professor of Medicine in the Division of Allergy and Immunology and a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University.
Microglia are the brain’s primary immune cells that respond to injury and disease, transitioning between homeostatic and reactive cell states to regulate inflammation. However, the underlying mechanisms supporting microglia plasticity and driving these cell state transitions, which determine whether microglia promote or reduce inflammation throughout the central nervous system, have remained unclear.
In the current study, Prakriya’s team aimed to investigate the role of calcium (Ca2+) signaling in regulating microglia cell state transitions and neuroinflammatory signaling. Prakriya’s team had previously discovered that Orai1-mediated Ca2+ signaling plays a key role in regulating intracellular processes throughout the body, including gene expression, immune responses and phagocytosis, or the clearing of extracellular debris.
The scientists used a combination of transcriptomics, metabolomics and in vivo functional analysis techniques to study microglia in mouse models of central nervous system inflammation.
Using these techniques, they discovered that microglial cell state transitions are supported by Orai1 Ca2+ channels, and that these channels control the expression of proinflammatory genes linked to immunity, inflammation and cell metabolism.
In Orai1 knockout mice, the investigators found that Orai1-deficient microglia were unable to transition into a reactive, proinflammatory state. Instead, these microglia produced increased amounts of neuroprotective and anti-inflammatory mediators, including the BDNF protein, which supports neuronal survival and growth.
“It seems to be a pathway that is centrally plugged into the gene expression of inflammatory cytokines and happens through the regulation of gene transcription,” Prakriya said.
 Kaitlyn DeMeulenaere, a student in the Driskill Graduate Program in Life Sciences (DGP), was lead author of the study.
Kaitlyn DeMeulenaere, a student in the Driskill Graduate Program in Life Sciences (DGP), was lead author of the study.
Using genetic approaches, the scientists also found that deleting Orai1 in microglia in the mice weakened reactivity in microglia and astrocytes (a type of glial cell) and reduced the production of proinflammatory cytokines in the hippocampus, specifically IL-1 beta and IL-6.
In subsequent behavioral analyses of Orai1 knockout mice, the scientists found that the mice did not demonstrate changes in motivational behaviors, specifically showing impaired reward-seeking and avoidance behaviors.
“When we knockout the expression of Orai1, not only are inflammatory pathways blunted, but in fact, the cellular state of microglia is changed into an anti-inflammatory phenotype where they’re now secreting molecules and expressing genes that promote neural survival. That’s not something that we expected,” Prakriya said.
The findings establish Orai1 channels as key regulators of microglial cell state transitions and suggest that Orai1 may be a potential therapeutic target for brain disorders characterized by microglial activation and chronic neuroinflammation, such as depression, neurodegenerative diseases and chronic pain.
“We found that loss of Orai1 not only dampens the pro-inflammatory response but also promotes the production of neurotrophic factors and anti-inflammatory mediators. The concurrent shifts in inflammatory mediators and key metabolites further expand our understanding of Orai1’s role in regulating microglial responses to inflammation,” said Kaitlyn DeMeulenaere, a student in the Driskill Graduate Program in Life Sciences (DGP), who was lead author of the study.
This work was supported by National Institutes of Health grant R35NS132349, a Julius Kahn predoctoral fellowship, the Kimberly Querrey Fellowship in Data Science, the Schmidt Science Fellows program, and the Canadian Institutes of Health Research (FDN-154336 and PJT-191976).