A comprehensive review published in eGastroenterology highlights cluster of differentiation 47 (CD47) as a unifying immunoregulatory molecule that influences the pathogenesis, progression and therapeutic response of a wide spectrum of liver and gastrointestinal (GI) diseases. The article positions CD47 not merely as an immune “don’t-eat-me” signal, but as a multifunctional modulator of cellular crosstalk, metabolic stress responses, stromal interactions and tumour immunity.
A multifunctional immune checkpoint with broad influence
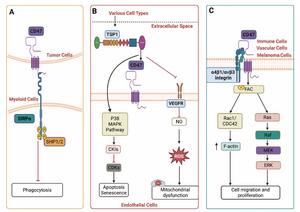
CD47 is a widely expressed transmembrane glycoprotein that interacts with SIRPα, TSP1 and multiple integrins to regulate phagocytosis, inflammation, cell migration, apoptosis and endothelial signalling. The review outlines its structural basis and signalling mechanisms, illustrated by detailed diagrams on pages 2–3 showing CD47–SIRPα ITIM signalling, the TSP1–CD47–NO axis, and CD47–integrin complexes that influence cytoskeletal dynamics and proliferation.
These interaction networks form the mechanistic foundation for CD47’s roles across digestive diseases, including steatotic liver disease, inflammatory bowel disease and gastrointestinal malignancies.
Steatotic liver disease and steatohepatitis: CD47 as a driver of inflammation and fibrosis
The review provides extensive insights into MASLD/MASH, conditions now recognised as major global drivers of chronic liver disease. Evidence from murine, human organoid and cell-based studies shows that CD47 orchestrates key pathological events:
Impaired clearance of necrotic hepatocytes: Upregulated CD47 on dying hepatocytes prevents efficient phagocytosis by Kupffer cells through SIRPα, sustaining inflammation and worsening MASH.
Activation of hepatic stellate cells: YAP/TEAD4-driven CD47 expression promotes HSC activation and extracellular matrix deposition, accelerating fibrosis.
Microvascular dysfunction via LSECs: TSP1–CD47 signalling triggers Rho-ROCK–myosin–mediated LSEC defenestration, impairing nutrient exchange and promoting fibrogenesis.
Conflicting effects on steatosis: While some studies show CD47 deficiency protects against diet-induced steatosis through enhanced lipolysis and mitochondrial efficiency, others report worsening steatosis under prolonged dietary stress—highlighting context specificity.
Collectively, these data underscore CD47 as a central regulator of metabolic inflammation and fibrosis across MASLD/MASH.
Hepatocellular carcinoma: a mediator of immune escape
In HCC, CD47 overexpression on tumour cells correlates with poor survival and metastatic potential. Mechanistically, CD47 blockade enhances macrophage phagocytosis, reactivates dendritic cell–NK cell axes, and disrupts IL-6–STAT3 feedback loops that fuel tumour expansion. Experimental models show robust tumour suppression with anti-CD47 therapy, and microwave ablation may heighten responsiveness by inducing CD47 expression via METTL3–IGF2BP1 signalling.
Inflammatory bowel disease: epithelial and immune dysregulation
The review summarises discoveries showing that CD47 is upregulated in inflamed intestinal epithelium and restricts epithelial renewal by suppressing stemness regulators including Oct4, Sox2 and KLF4. CD47 also enhances pathogenic SIRPα⁺CD103⁻ dendritic cell trafficking and promotes IL-17–mediated inflammation. Conversely, CD47 deficiency improves mucosal healing but may impair wound repair when blocked indiscriminately, underscoring the need for precise therapeutic targeting.
Drug-induced liver injury and liver transplantation
Innovative therapeutic platforms such as CD47-engineered exosomes and CD47-enhanced mesenchymal stem cells demonstrate hepatoprotection in acetaminophen toxicity models by evading macrophage clearance and promoting repair pathways. CD47 also emerges as a critical determinant in liver ischaemia–reperfusion injury, where its interaction with TSP1 exacerbates endothelial dysfunction and oxidative stress. Monoclonal antibody blockade has shown significant protection in mouse, rat and porcine models.
In transplantation immunology, CD47 expression on donor hepatocytes is essential for preventing macrophage-driven graft rejection, highlighting a dual, context-dependent role.
GI cancers beyond the liver: CRC and PDAC
Colorectal cancer (CRC): CD47 overexpression associates with poor differentiation, angiogenesis and worse overall survival. Combination approaches, including SHP2 inhibitors, PPARδ antagonists and cordycepin, synergise with CD47 blockade in preclinical models.
Pancreatic ductal adenocarcinoma (PDAC): CD47 is highly expressed in PDAC cells and CSCs, restricting phagocytosis and contributing to liver metastasis via necroptosis-induced upregulation. Anti-CD47 therapies, particularly when paired with necroptosis or CTLA-4 inhibitors, have shown potent anti-tumour effects.
Therapeutic landscape and clinical translation: A rapidly expanding pipeline of CD47-targeting agents—including monoclonal antibodies, bispecific antibodies and fusion proteins—is advancing through Phase I–II clinical trials across HCC, CRC, gastric cancer and PDAC. Early data from IMC-002 trials in HCC show encouraging disease control rates (80%) and a median progression-free survival of 8.3 months.
The review emphasises that future success will rely on strategies that balance efficacy with safety, given risks such as anaemia from red cell CD47 targeting. Emerging single-cell and spatial technologies are expected to refine cell-specific targeting and identify predictive biomarkers.
Conclusion
The review positions CD47 as a central immune and metabolic hub across liver and GI pathology. From steatohepatitis and fibrosis to cancer progression and transplant biology, CD47’s widespread influence makes it a compelling therapeutic target. As preclinical evidence accumulates and clinical trials progress, CD47-directed therapies may soon reshape treatment strategies in hepatology and gastroenterology.
See the article:
Otuagomah J, Newcomb A, Gwag T, et al. CD47: an immunoregulatory nexus in liver and gastrointestinal disorders. eGastroenterology 2025;3:e100242. doi:10.1136/egastro-2025-100242
About eGastroenterology
eGastroenterology, a BMJ journal partnered with Gut and launched by leading scientists in gastroenterology and hepatology, has been indexed in the Web of Science Core Collection (ESCI), PubMed, DOAJ, Scopus, CAS, ROAD, and many other major international databases within just two years of its launch. The journal is expecting to receive its first Impact Factor in June 2026.
For more information, please visit: egastroenterology.bmj.com and follow us on Twitter (@eGastro_BMJ).
Sign-up to Email Alerts for eGastroenterology: https://emails.bmj.com/k/Bmj/jausu/egastroenterology
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of news releases posted to EurekAlert! by contributing institutions or for the use of any information through the EurekAlert system.