Rausch, L. & Kallies, A. Molecular mechanisms governing CD8 T cell differentiation and checkpoint inhibitor response in cancer. Annu. Rev. Immunol. 43, 515–543 (2025).
McMichael, A. & Hanke, T. The quest for an AIDS vaccine: is the CD8+ T-cell approach feasible? Nat. Rev. Immunol. 2, 283–291 (2002).
Appay, V., Douek, D. C. & Price, D. A. CD8+ T cell efficacy in vaccination and disease. Nat. Med. 14, 623–628 (2008).
McMichael, A. J. HIV vaccines. Annu. Rev. Immunol. 24, 227–255 (2006).
Jones, R. B. & Walker, B. D. HIV-specific CD8+ T cells and HIV eradication. J. Clin. Invest. 126, 455–463 (2016).
Buchbinder, S. P. et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet 372, 1881–1893 (2008).
Hammer, S. M. et al. Efficacy trial of a DNA/rAd5 HIV-1 preventive vaccine. N. Engl. J. Med. 369, 2083–2092 (2013).
Li, S. S. et al. DNA priming increases frequency of T-cell responses to a vesicular stomatitis virus HIV vaccine with specific enhancement of CD8+ T-cell responses by interleukin-12 plasmid DNA. Clin. Vaccine Immunol. 24, e00263–17 (2017).
Rolland, M. et al. Genetic impact of vaccination on breakthrough HIV-1 sequences from the STEP trial. Nat. Med. 17, 366–371 (2011).
Fitzgerald, D. W. et al. An Ad5-vectored HIV-1 vaccine elicits cell-mediated immunity but does not affect disease progression in HIV-1-infected male subjects: results from a randomized placebo-controlled trial (the Step study). J. Infect. Dis. 203, 765–772 (2011).
Janes, H. et al. Vaccine-induced gag-specific T cells are associated with reduced viremia after HIV-1 infection. J. Infect. Dis. 208, 1231–1239 (2013).
Boppana, S. et al. HLA-I associated adaptation dampens CD8 T-cell responses in HIV Ad5-vectored vaccine recipients. J. Infect. Dis. 220, 1620–1628 (2019).
Haynes, B. F. et al. Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N. Engl. J. Med. 366, 1275–1286 (2012).
Yang, L. & Su, J. Type II grass carp reovirus infects leukocytes but not erythrocytes and thrombocytes in grass carp (Ctenopharyngodon idella). Viruses 13, 870 (2021).
Garcia-Beltran, W. F. et al. Innate immune reconstitution in humanized bone marrow–liver–thymus (HuBLT) mice governs adaptive cellular immune function and responses to HIV-1 infection. Front. Immunol. 12, 667393 (2021).
Chen, R. E. et al. Resistance of SARS-CoV-2 variants to neutralization by monoclonal and serum-derived polyclonal antibodies. Nat. Med. 27, 717–726 (2021).
Cele, S. et al. Omicron extensively but incompletely escapes Pfizer BNT162b2 neutralization. Nature 602, 654–656 (2022).
Moree, L. K., Faulkner, L. A. V. & Crowley, J. D. Heterometallic cages: synthesis and applications. Chem. Soc. Rev. 53, 25–46 (2024).
Cromer, D. et al. Prospects for durable immune control of SARS-CoV-2 and prevention of reinfection. Nat. Rev. Immunol. 21, 395–404 (2021).
Picker, L. J., Lifson, J. D., Gale, M. Jr, Hansen, S. G. & Fruh, K. Programming cytomegalovirus as an HIV vaccine. Trends Immunol. 44, 287–304 (2023).
Peluso, M. et al. Correlates of HIV-1 control after combination immunotherapy. Nature 650, 187–195 (2026). This proof-of-concept clinical study associates ART-free control of HIV following multimodal combination immunotherapy with enhanced CD8⁺ T cell responses.
Giles, J. R., Globig, A. M., Kaech, S. M. & Wherry, E. J. CD8+ T cells in the cancer-immunity cycle. Immunity 56, 2231–2253 (2023).
Galluzzi, L., Smith, K. N., Liston, A. & Garg, A. D. The diversity of CD8+ T cell dysfunction in cancer and viral infection. Nat. Rev. Immunol. 25, 662–679 (2025).
McMichael, A. J., Gotch, F. M., Noble, G. R. & Beare, P. A. Cytotoxic T-cell immunity to influenza. N. Engl. J. Med. 309, 13–17 (1983).
Sridhar, S. et al. Cellular immune correlates of protection against symptomatic pandemic influenza. Nat. Med. 19, 1305–1312 (2013).
Wang, Z. et al. Recovery from severe H7N9 disease is associated with diverse response mechanisms dominated by CD8+ T cells. Nat. Commun. 6, 6833 (2015).
Grant, E. J., Quinones-Parra, S. M., Clemens, E. B. & Kedzierska, K. Human influenza viruses and CD8+ T cell responses. Curr. Opin. Virol. 16, 132–142 (2016).
Kaech, S. M. & Cui, W. Transcriptional control of effector and memory CD8+ T cell differentiation. Nat. Rev. Immunol. 12, 749–761 (2012).
Rydyznski Moderbacher, C. et al. Antigen-specific adaptive immunity to SARS-CoV-2 in acute COVID-19 and associations with age and disease severity. Cell 183, 996–1012.e19 (2020).
Ramirez, S. I. et al. Early antiviral CD4+ and CD8+ T cells are associated with upper airway clearance of SARS-CoV-2. JCI Insight 9, e186078 (2024).
Tan, A. T. et al. Early induction of functional SARS-CoV-2-specific T cells associates with rapid viral clearance and mild disease in COVID-19 patients. Cell Rep. 34, 108728 (2021).
Wagner, K. I. et al. Recruitment of highly cytotoxic CD8+ T cell receptors in mild SARS-CoV-2 infection. Cell Rep. 38, 110214 (2022).
Zheng, H. Y. et al. Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients. Cell Mol. Immunol. 17, 541–543 (2020).
Gozzi-Silva, S. C. et al. Generation of cytotoxic T cells and dysfunctional CD8 T cells in severe COVID-19 patients. Cells 11, 3359 (2022).
Autaa, G. et al. Aging and inflammation limit the induction of SARS-CoV-2-specific CD8+ T cell responses in severe COVID-19. JCI Insight 10, e180867 (2025).
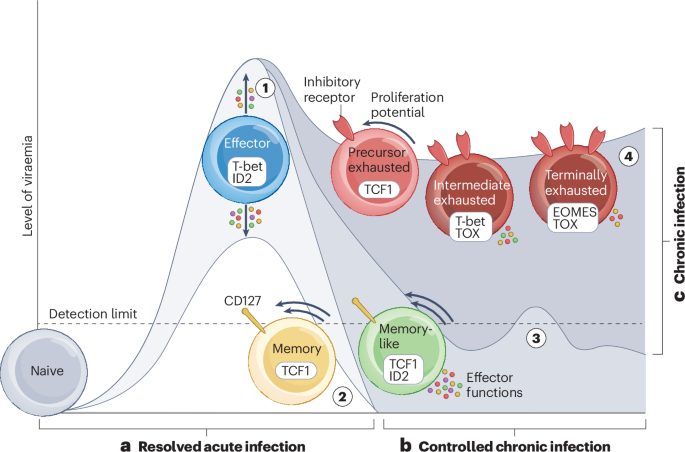
Zehn, D., Thimme, R., Lugli, E., de Almeida, G. P. & Oxenius, A. Stem-like precursors are the fount to sustain persistent CD8+ T cell responses. Nat. Immunol. 23, 836–847 (2022).
Zens, K. D., Chen, J. K. & Farber, D. L. Vaccine-generated lung tissue-resident memory T cells provide heterosubtypic protection to influenza infection. JCI Insight 1, e85832 (2016).
Pizzolla, A. et al. Influenza-specific lung-resident memory T cells are proliferative and polyfunctional and maintain diverse TCR profiles. J. Clin. Invest. 128, 721–733 (2018).
van de Wall, S. et al. Dynamic landscapes and protective immunity coordinated by influenza-specific lung-resident memory CD8+ T cells revealed by intravital imaging. Immunity 57, 1878–1892.e5 (2024).
Yang, G. et al. Initial COVID-19 severity influenced by SARS-CoV-2-specific T cells imprints T-cell memory and inversely affects reinfection. Signal Transduct. Target. Ther. 9, 141 (2024).
Painter, M. M. et al. Prior vaccination promotes early activation of memory T cells and enhances immune responses during SARS-CoV-2 breakthrough infection. Nat. Immunol. 24, 1711–1724 (2023). This study demonstrates that vaccine-induced cellular immunity has a key role in protecting vaccinating individuals from severe COVID.
Zhang, B. et al. Multimodal single-cell datasets characterize antigen-specific CD8+ T cells across SARS-CoV-2 vaccination and infection. Nat. Immunol. 24, 1725–1734 (2023).
Geers, D. et al. SARS-CoV-2 variants of concern partially escape humoral but not T-cell responses in COVID-19 convalescent donors and vaccinees. Sci. Immunol. 6, eabj1750 (2021).
Nogimori, T. et al. Functional changes in cytotoxic CD8+ T-cell cross-reactivity against the SARS-CoV-2 Omicron variant after mRNA vaccination. Front. Immunol. 13, 1081047 (2022).
Liu, J. et al. Vaccines elicit highly conserved cellular immunity to SARS-CoV-2 Omicron. Nature 603, 493–496 (2022).
Naranbhai, V. et al. T cell reactivity to the SARS-CoV-2 Omicron variant is preserved in most but not all individuals. Cell 185, 1041–1051.e6 (2022).
Collins, D. R., Gaiha, G. D. & Walker, B. D. CD8+ T cells in HIV control, cure and prevention. Nat. Rev. Immunol. 20, 471–482 (2020).
Lieberman, J., Shankar, P., Manjunath, N. & Andersson, J. Dressed to kill? A review of why antiviral CD8 T lymphocytes fail to prevent progressive immunodeficiency in HIV-1 infection. Blood 98, 1667–1677 (2001).
Lecuroux, C. et al. CD8 T-cells from most HIV-infected patients lack ex vivo HIV-suppressive capacity during acute and early infection. PLoS ONE 8, e59767 (2013).
Migueles, S. A. et al. Lytic granule loading of CD8+ T cells is required for HIV-infected cell elimination associated with immune control. Immunity 29, 1009–1021 (2008).
Passaes, C. et al. Optimal maturation of the SIV-specific CD8+ T cell response after primary infection is associated with natural control of SIV: ANRS SIC study. Cell Rep. 32, 108174 (2020).
Huang, S. H. et al. Latent HIV reservoirs exhibit inherent resistance to elimination by CD8+ T cells. J. Clin. Invest. 128, 876–889 (2018).
Day, C. L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006).
Petrovas, C. et al. PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J. Exp. Med. 203, 2281–2292 (2006).
Trautmann, L. et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat. Med. 12, 1198–1202 (2006).
Reuter, M. A. et al. HIV-specific CD8+ T cells exhibit reduced and differentially regulated cytolytic activity in lymphoid tissue. Cell Rep. 21, 3458–3470 (2017).
Rutishauser, R. L. & Trautmann, L. CD8+ T-cell responses in HIV controllers: potential implications for novel HIV remission strategies. Curr. Opin. HIV AIDS 17, 315–324 (2022).
Saez-Cirion, A. et al. HIV controllers exhibit potent CD8 T cell capacity to suppress HIV infection ex vivo and peculiar cytotoxic T lymphocyte activation phenotype. Proc. Natl Acad. Sci. USA 104, 6776–6781 (2007).
Rutishauser, R. L. et al. TCF-1 regulates HIV-specific CD8+ T cell expansion capacity. JCI Insight 6, e136648 (2021).
Migueles, S. A. et al. HIV-specific CD8+ T cell proliferation is coupled to perforin expression and is maintained in nonprogressors. Nat. Immunol. 3, 1061–1068 (2002).
Collins, D. R. et al. Cytolytic CD8+ T cells infiltrate germinal centers to limit ongoing HIV replication in spontaneous controller lymph nodes. Sci. Immunol. 8, eade5872 (2023).
Angin, M. et al. Metabolic plasticity of HIV-specific CD8+ T cells is associated with enhanced antiviral potential and natural control of HIV-1 infection. Nat. Metab. 1, 704–716 (2019).
Angin, M. et al. Preservation of lymphopoietic potential and virus suppressive capacity by CD8+ T cells in HIV-2-infected controllers. J. Immunol. 197, 2787–2795 (2016).
Leligdowicz, A. et al. Robust gag-specific T cell responses characterize viremia control in HIV-2 infection. J. Clin. Invest. 117, 3067–3074 (2007).
Strongin, Z. et al. Distinct SIV-specific CD8+ T cells in the lymph node exhibit simultaneous effector and stem-like profiles and are associated with limited SIV persistence. Nat. Immunol. 25, 1245–1256 (2024). This study identifies a stem-like, antigen-specific CD8+ T cell population associated with lower viraemia and a smaller viral reservoir, highlighting a promising target for curative immunotherapies.
Saez-Cirion, A. et al. Memory CD8+ T cells co-expressing TCF1 and ID2 combine effector and stem-like functions in persistent infections. Preprint at Res. Sq. https://doi.org/10.21203/rs.3.rs-8047338/v1 (2025).
Jiang, C. et al. Distinct viral reservoirs in individuals with spontaneous control of HIV-1. Nature 585, 261–267 (2020).
Salgado, M., Migueles, S. A., Yu, X. G. & Martinez-Picado, J. Exceptional, naturally occurring HIV-1 control: insight into a functional cure. Med 5, 1071–1082 (2024).
Harwood, O. E. et al. CD8+ cells and small viral reservoirs facilitate post-ART control of SIV replication in M3+ Mauritian cynomolgus macaques initiated on ART two weeks post-infection. PLoS Pathog. 19, e1011676 (2023).
Passaes, C. et al. Early antiretroviral therapy favors post-treatment SIV control associated with the expansion of enhanced memory CD8+ T-cells. Nat. Commun. 15, 178 (2024). This study shows that early ART promotes the development of stem-like memory CD8+ T cells with strong antiviral capacity that can expand after ART interruption and contribute to post-treatment control of SIV.
Migueles, S. A. et al. HIV vaccines induce CD8+ T cells with low antigen receptor sensitivity. Science 382, 1270–1276 (2023). This study shows that the limited efficacy of CD8⁺ T cell-based HIV vaccines is driven by suboptimal functional properties and TCR avidity, rather than the frequency of vaccine-induced cells.
Klenerman, P. & Oxenius, A. T cell responses to cytomegalovirus. Nat. Rev. Immunol. 16, 367–377 (2016).
Griffiths, P. & Reeves, M. Pathogenesis of human cytomegalovirus in the immunocompromised host. Nat. Rev. Microbiol. 19, 759–773 (2021).
Sylwester, A. W. et al. Broadly targeted human cytomegalovirus-specific CD4+ and CD8+ T cells dominate the memory compartments of exposed subjects. J. Exp. Med. 202, 673–685 (2005).
Huggins, M. A. & Hamilton, S. E. T cell economics: precursor cells predict inflation. Nat. Immunol. 21, 1482–1483 (2020).
Shoukry, N. H. et al. Memory CD8+ T cells are required for protection from persistent hepatitis C virus infection. J. Exp. Med. 197, 1645–1655 (2003).
Grakoui, A. et al. HCV persistence and immune evasion in the absence of memory T cell help. Science 302, 659–662 (2003).
Osburn, W. O. et al. Spontaneous control of primary hepatitis C virus infection and immunity against persistent reinfection. Gastroenterology 138, 315–324 (2010).
Kasprowicz, V. et al. High level of PD-1 expression on hepatitis C virus (HCV)-specific CD8+ and CD4+ T cells during acute HCV infection, irrespective of clinical outcome. J. Virol. 82, 3154–3160 (2008).
Shin, E. C., Sung, P. S. & Park, S. H. Immune responses and immunopathology in acute and chronic viral hepatitis. Nat. Rev. Immunol. 16, 509–523 (2016).
Iannacone, M. & Guidotti, L. G. Immunobiology and pathogenesis of hepatitis B virus infection. Nat. Rev. Immunol. 22, 19–32 (2022).
Heim, K. et al. Attenuated effector T cells are linked to control of chronic HBV infection. Nat. Immunol. 25, 1650–1662 (2024). This study identifies a subset of CD8+ T cells with traits of memory and attenuated effector cells in the absence of T cell exhaustion that is associated with natural HBV control during chronic infection.
Cheng, Y. et al. Multifactorial heterogeneity of virus-specific T cells and association with the progression of human chronic hepatitis B infection. Sci. Immunol. 4, eaau6905 (2019).
Almeida, J. R. et al. Antigen sensitivity is a major determinant of CD8+ T-cell polyfunctionality and HIV-suppressive activity. Blood 113, 6351–6360 (2009).
Tan, M. P. et al. T cell receptor binding affinity governs the functional profile of cancer-specific CD8+ T cells. Clin. Exp. Immunol. 180, 255–270 (2015).
Abdel-Hakeem, M. S., Boisvert, M., Bruneau, J., Soudeyns, H. & Shoukry, N. H. Selective expansion of high functional avidity memory CD8 T cell clonotypes during hepatitis C virus reinfection and clearance. PLoS Pathog. 13, e1006191 (2017).
Almeida, J. R. et al. Superior control of HIV-1 replication by CD8+ T cells is reflected by their avidity, polyfunctionality, and clonal turnover. J. Exp. Med. 204, 2473–2485 (2007).
Lissina, A. et al. The link between CD8+ T-cell antigen-sensitivity and HIV-suppressive capacity depends on HLA restriction, target epitope and viral isolate. AIDS 28, 477–486 (2014).
Foley, M. H. et al. High avidity CD8+ T cells efficiently eliminate motile HIV-infected targets and execute a locally focused program of anti-viral function. PLoS ONE 9, e87873 (2014).
Lima, N. S. et al. CTL clonotypes with higher TCR affinity have better ability to reduce the HIV latent reservoir. J. Immunol. 205, 699–707 (2020).
Schamel, W. W. et al. Coexistence of multivalent and monovalent TCRs explains high sensitivity and wide range of response. J. Exp. Med. 202, 493–503 (2005).
Price, D. A. et al. Public clonotype usage identifies protective Gag-specific CD8+ T cell responses in SIV infection. J. Exp. Med. 206, 923–936 (2009).
Ladell, K. et al. A molecular basis for the control of preimmune escape variants by HIV-specific CD8+ T cells. Immunity 38, 425–436 (2013).
Chen, L. & Flies, D. B. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat. Rev. Immunol. 13, 227–242 (2013).
Curtsinger, J. M. & Mescher, M. F. Inflammatory cytokines as a third signal for T cell activation. Curr. Opin. Immunol. 22, 333–340 (2010).
Cox, M. A., Kahan, S. M. & Zajac, A. J. Anti-viral CD8 T cells and the cytokines that they love. Virology 435, 157–169 (2013).
Achar, S. R. et al. Universal antigen encoding of T cell activation from high-dimensional cytokine dynamics. Science 376, 880–884 (2022). This study presents a universal, low-dimensional ‘antigen-encoding’ framework that provides a foundation for modelling and rationally optimizing vaccines and immunotherapies.
Blank, C. U. et al. Defining ‘T cell exhaustion’. Nat. Rev. Immunol. 19, 665–674 (2019).
Vigano, S. et al. Functional avidity: a measure to predict the efficacy of effector T cells? Clin. Dev. Immunol. 2012, 153863 (2012).
Yates, K. B. et al. Epigenetic scars of CD8+ T cell exhaustion persist after cure of chronic infection in humans. Nat. Immunol. 22, 1020–1029 (2021).
Tonnerre, P. et al. Differentiation of exhausted CD8+ T cells after termination of chronic antigen stimulation stops short of achieving functional T cell memory. Nat. Immunol. 22, 1030–1041 (2021).
Vieira, V. A. et al. An HLA-I signature favouring KIR-educated natural killer cells mediates immune control of HIV in children and contrasts with the HLA-B-restricted CD8+ T-cell-mediated immune control in adults. PLoS Pathog. 17, e1010090 (2021).
Hensel, N. et al. Memory-like HCV-specific CD8+ T cells retain a molecular scar after cure of chronic HCV infection. Nat. Immunol. 22, 229–239 (2021).
Utzschneider, D. T. et al. T cells maintain an exhausted phenotype after antigen withdrawal and population reexpansion. Nat. Immunol. 14, 603–610 (2013).
Thimme, R., Bertoletti, A. & Iannacone, M. Beyond exhaustion: the unique characteristics of CD8+ T cell dysfunction in chronic HBV infection. Nat. Rev. Immunol. 24, 775–776 (2024).
Collins, D. R. et al. Functional impairment of HIV-specific CD8+ T cells precedes aborted spontaneous control of viremia. Immunity 54, 2372–2384.e7 (2021). This study reveals that the loss of spontaneous HIV-1 control is preceded by changes in the transcriptional programme and functional impairment of HIV-specific CD8+ T cells, distinct from T cell exhaustion or senescence.
Sekine, T. et al. TOX is expressed by exhausted and polyfunctional human effector memory CD8+ T cells. Sci. Immunol. 5, eaba7918 (2020).
Gerlach, C. et al. One naive T cell, multiple fates in CD8+ T cell differentiation. J. Exp. Med. 207, 1235–1246 (2010).
Tubo, N. J. et al. Single naive CD4+ T cells from a diverse repertoire produce different effector cell types during infection. Cell 153, 785–796 (2013).
Stemberger, C. et al. Stem cell-like plasticity of naïve and distinct memory CD8+ T cell subsets. Semin. Immunol. 21, 62–68 (2009).
Quezada, L. K. et al. Early transcriptional and epigenetic divergence of CD8+ T cells responding to acute versus chronic infection. PLoS Biol. 21, e3001983 (2023).
Rudloff, M. W. et al. Hallmarks of CD8+ T cell dysfunction are established within hours of tumor antigen encounter before cell division. Nat. Immunol. 24, 1527–1539 (2023). This study reveals that divergent T cell fate decisions are programmed early by antigen exposure, thus redefining the temporal origins of T cell dysfunction.
Galletti, G. et al. Two subsets of stem-like CD8+ memory T cell progenitors with distinct fate commitments in humans. Nat. Immunol. 21, 1552–1562 (2020). This study identifies progenitor CD8+ T cells with alternative programme commitments.
Snell, L. M. et al. CD8+ T cell priming in established chronic viral infection preferentially directs differentiation of memory-like cells for sustained immunity. Immunity 49, 678–694.e5 (2018).
Bhandarkar, V., Dinter, T. & Spranger, S. Architects of immunity: how dendritic cells shape CD8+ T cell fate in cancer. Sci. Immunol. 10, eadf4726 (2025).
Williams, C. et al. CD28 and TCR differentially impact naïve and memory T cell responses. Discov. Immunol. 4, kyaf006 (2025).
Webb, G. M. et al. The human IL-15 superagonist ALT-803 directs SIV-specific CD8+ T cells into B-cell follicles. Blood Adv. 2, 76–84 (2018).
Miller, J. S. et al. Safety and virologic impact of the IL-15 superagonist N-803 in people living with HIV: a phase 1 trial. Nat. Med. 28, 392–400 (2022).
Cabral-Piccin, M. P. et al. Primary role of type I interferons for the induction of functionally optimal antigen-specific CD8+ T cells in HIV infection. eBioMedicine 91, 104557 (2023). This study demonstrates that HIV-2 elicits polyfunctional CD8⁺ T cell responses via cGAS–STING-dependent type I IFN signalling, providing a mechanistic blueprint for using STING agonists to boost CD8⁺ T cell-mediated immunity against HIV-1.
Martin-Gayo, E. et al. Potent cell-intrinsic immune responses in dendritic cells facilitate HIV-1-specific T cell immunity in HIV-1 elite controllers. PLoS Pathog. 11, e1004930 (2015).
Gutjahr, A. et al. The STING ligand cGAMP potentiates the efficacy of vaccine-induced CD8+ T cells. JCI Insight 4, e125107 (2019).
Chauveau, L. et al. Inclusion of cGAMP within virus-like particle vaccines enhances their immunogenicity. EMBO Rep. 22, e52447 (2021).
Odorizzi, P. M. & Wherry, E. J. Immunology. An interferon paradox. Science 340, 155–156 (2013).
Zhen, A. et al. Targeting type I interferon-mediated activation restores immune function in chronic HIV infection. J. Clin. Invest. 127, 260–268 (2017).
Chen, W. et al. Chronic type I interferon signaling promotes lipid-peroxidation-driven terminal CD8+ T cell exhaustion and curtails anti-PD-1 efficacy. Cell Rep. 41, 111647 (2022).
Reina-Campos, M., Scharping, N. E. & Goldrath, A. W. CD8+ T cell metabolism in infection and cancer. Nat. Rev. Immunol. 21, 718–738 (2021).
Wang, Y.-Y. et al. Elevated glutamate impedes anti-HIV-1 CD8 + T cell responses in HIV-1-infected individuals on antiretroviral therapy. Commun. Biol. 6, 696 (2023).
Tarancon-Diez, L. et al. Immunometabolism is a key factor for the persistent spontaneous elite control of HIV-1 infection. eBioMedicine 42, 86–96 (2019).
Schurich, A. et al. Distinct metabolic requirements of exhausted and functional virus-specific CD8 T cells in the same host. Cell Rep. 16, 1243–1252 (2016).
Aregay, A. et al. Elimination of hepatitis C virus has limited impact on the functional and mitochondrial impairment of HCV-specific CD8+ T cell responses. J. Hepatol. 71, 889–899 (2019).
Schuch, A. et al. Phenotypic and functional differences of HBV core-specific versus HBV polymerase-specific CD8+ T cells in chronically HBV-infected patients with low viral load. Gut 68, 905–915 (2019).
Nicoli, F. et al. Altered basal lipid metabolism underlies the functional impairment of naive CD8+ T cells in elderly humans. J. Immunol. 208, 562–570 (2022).
Li, S.-Y. et al. Altered lipid metabolites accelerate early dysfunction of T cells in HIV-infected rapid progressors by impairing mitochondrial function. Front. Immunol. 14, 1106881 (2023).
Tuchel, M.-E. & Oxenius, A. Lipids rewire T cell exhaustion. Nat. Immunol. 26, 984–985 (2025).
Ma, X. et al. Cholesterol induces CD8+ T cell exhaustion in the tumor microenvironment. Cell Metab. 30, 143–156.e5 (2019).
Raynor, J. L. & Chi, H. Nutrients: signal 4 in T cell immunity. J. Exp. Med. 221, e20221839 (2024).
McMichael, A. J. & Picker, L. J. Unusual antigen presentation offers new insight into HIV vaccine design. Curr. Opin. Immunol. 46, 75–81 (2017).
Gillespie, G. M., Quastel, M. N. & McMichael, A. J. HLA-E: immune receptor functional mechanisms revealed by structural studies. Immunol. Rev. 329, e13434 (2025).
Zanoni, M. et al. Innate, non-cytolytic CD8+ T cell-mediated suppression of HIV replication by MHC-independent inhibition of virus transcription. PLoS Pathog. 16, e1008821 (2020).
Mutascio, S. et al. CD8+ T cells promote HIV latency by remodeling CD4+ T cell metabolism to enhance their survival, quiescence, and stemness. Immunity 56, 1132–1147.e6 (2023).
Takata, H. et al. Long-term antiretroviral therapy initiated in acute HIV infection prevents residual dysfunction of HIV-specific CD8+ T cells. eBioMedicine 84, 104253 (2022).
White, E. et al. Clonal succession after prolonged antiretroviral therapy rejuvenates CD8+ T cell responses against HIV-1. Nat. Immunol. 25, 1555–1564 (2024). This study shows that long-term ART is linked to the replacement of exhausted HIV-1-specific clonotypes with stem-like CD8⁺ T cells, revealing unexpected immune resilience with potential therapeutic implications.
Cabral-Piccin, M. P. et al. CD8+ T-cell priming is quantitatively but not qualitatively impaired in people with HIV-1 on antiretroviral therapy. AIDS 38, 161–166 (2024).
Musunuri, S., Weidenbacher, P. A. B. & Kim, P. S. Bringing immunofocusing into focus. npj Vaccines 9, 11 (2024).
Gilfillan, C. B., Hebeisen, M., Rufer, N. & Speiser, D. E. Constant regulation for stable CD8 T-cell functional avidity and its possible implications for cancer immunotherapy. Eur. J. Immunol. 51, 1348–1360 (2021).
Schmidt, J. et al. Neoantigen-specific CD8 T cells with high structural avidity preferentially reside in and eliminate tumors. Nat. Commun. 14, 3188 (2023).
Huang, D. et al. TCR-mimicking STAR conveys superior sensitivity over CAR in targeting tumors with low-density neoantigens. Cell Rep. 43, 114949 (2024).
Muhlgrabner, V. et al. TCR/CD3-based synthetic antigen receptors (TCC) convey superior antigen sensitivity combined with high fidelity of activation. Sci. Adv. 10, eadj4632 (2024).
Carnevale, J. et al. RASA2 ablation in T cells boosts antigen sensitivity and long-term function. Nature 609, 174–182 (2022).
Mueller, A. L. et al. T-cell receptor/CD28-targeted immunotherapeutics selectively drive naive T-cell expansion to generate functional HIV-specific responses. J. Virol. 99, e00188255 (2025).
Smith, L. K. et al. Interleukin-10 directly inhibits CD8+ T cell function by enhancing N-glycan branching to decrease antigen sensitivity. Immunity 48, 299–312.e5 (2018).
Richer, M. J., Nolz, J. C. & Harty, J. T. Pathogen-specific inflammatory milieux tune the antigen sensitivity of CD8+ T cells by enhancing T cell receptor signaling. Immunity 38, 140–152 (2013).
Raue, H. P., Beadling, C., Haun, J. & Slifka, M. K. Cytokine-mediated programmed proliferation of virus-specific CD8+ memory T cells. Immunity 38, 131–139 (2013).
Rubinstein, M. P. et al. Interleukin-12 enhances the function and anti-tumor activity in murine and human CD8+ T cells. Cancer Immunol. Immunother. 64, 539–549 (2015).
Broomfield, B. J. et al. Transient inhibition of type I interferon enhances CD8+ T cell stemness and vaccine protection. J. Exp. Med. 222, e20241148 (2025).
Chatillon, J. F. et al. Direct Toll-like receptor 8 signaling increases the functional avidity of human CD8+ T lymphocytes generated for adoptive T cell therapy strategies. Immun. Inflamm. Dis. 3, 1–13 (2015).
Lissina, A. et al. Priming of qualitatively superior human effector CD8+ T cells using TLR8 ligand combined with FLT3 ligand. J. Immunol. 196, 256–263 (2016).
Nguema, L. et al. Subunit protein CD40.SARS.CoV2 vaccine induces SARS-CoV-2-specific stem cell-like memory CD8+ T cells. eBioMedicine 111, 105479 (2025).
Ninmer, E. K. et al. Multipeptide vaccines for melanoma in the adjuvant setting: long-term survival outcomes and post-hoc analysis of a randomized phase II trial. Nat. Commun. 15, 2570 (2024). This study shows that incorporating cognate melanoma-specific CD4⁺ T cell ‘helper’ peptides into a CD8⁺ T cell-targeted multipeptide vaccine durably enhances overall survival, uderscoring the clinical significance of targeting helper T cells.
Kundu, R. et al. Cross-reactive memory T cells associate with protection against SARS-CoV-2 infection in COVID-19 contacts. Nat. Commun. 13, 80 (2022).
Diniz, M. O. et al. Airway-resident T cells from unexposed individuals cross-recognize SARS-CoV-2. Nat. Immunol. 23, 1324–1329 (2022).
Lindeboom, R. G. H. et al. Human SARS-CoV-2 challenge uncovers local and systemic response dynamics. Nature 631, 189–198 (2024). This study maps the earliest local and systemic immune responses that differentiate abortive from sustained SARS-CoV-2 infection, providing a mechanistic blueprint for the development of improved vaccines and therapeutics.
Perreau, M. et al. Follicular helper T cells serve as the major CD4 T cell compartment for HIV-1 infection, replication, and production. J. Exp. Med. 210, 143–156 (2013).
Banga, R. et al. PD-1+ and follicular helper T cells are responsible for persistent HIV-1 transcription in treated aviremic individuals. Nat. Med. 22, 754–761 (2016).
Fukazawa, Y. et al. B cell follicle sanctuary permits persistent productive simian immunodeficiency virus infection in elite controllers. Nat. Med. 21, 132–139 (2015).
Buggert, M. et al. Identification and characterization of HIV-specific resident memory CD8+ T cells in human lymphoid tissue. Sci. Immunol. 3, eaar4526 (2018).
Lasrado, N. et al. An intramuscular prime and mucosal boost vaccine regimen protects against lethal clade 2.3.4.4b H5N1 challenge in cynomolgus macaques. Sci. Transl. Med. 17, eady2282 (2025). This study shows that an intramuscular prime followed by a mucosal boost in non-human primates elicits robust mucosal immunity, including T cell responses, supporting the potential of prime–boost mucosal vaccination strategies.
Corbett, K. S. et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 586, 567–571 (2020).
Lim, J. M. E. et al. A comparative characterization of SARS-CoV-2-specific T cells induced by mRNA or inactive virus COVID-19 vaccines. Cell Rep. Med. 3, 100793 (2022).
Reinscheid, M. et al. COVID-19 mRNA booster vaccine induces transient CD8+ T effector cell responses while conserving the memory pool for subsequent reactivation. Nat. Commun. 13, 4631 (2022).
Aoki, H. et al. CD8+ T cell memory induced by successive SARS-CoV-2 mRNA vaccinations is characterized by shifts in clonal dominance. Cell Rep. 43, 113887 (2024).
da Silva Antunes, R. et al. Evolution of SARS-CoV-2 T cell responses as a function of multiple COVID-19 boosters. Cell Rep. 44, 115907 (2025).
Arieta, C. M. et al. The T-cell-directed vaccine BNT162b4 encoding conserved non-spike antigens protects animals from severe SARS-CoV-2 infection. Cell 186, 2392–2409 (2023). This study demonstrates that an mRNA vaccine encoding conserved non-spike SARS-CoV-2 antigens elicits broad T cell immunity and protect animals from severe disease, supporting a next-generation COVID-19 vaccine strategy that is more resilient to viral variants.
Wang, C. Y. et al. Toward a pan-SARS-CoV-2 vaccine targeting conserved epitopes on spike and non-spike proteins for potent, broad and durable immune responses. PLoS Pathog. 19, e1010870 (2023).
Weber, J. S. et al. Individualised neoantigen therapy mRNA-4157 (V940) plus pembrolizumab versus pembrolizumab monotherapy in resected melanoma (KEYNOTE-942): a randomised, phase 2b study. Lancet 403, 632–644 (2024).
Grippin, A. J. et al. SARS-CoV-2 mRNA vaccines sensitize tumours to immune checkpoint blockade. Nature 647, 488–497 (2025).
Akahata, W. et al. Safety and immunogenicity of SARS-CoV-2 self-amplifying RNA vaccine expressing an anchored RBD: a randomized, observer-blind phase 1 study. Cell Rep. Med. 4, 101134 (2023).
Komori, M. et al. saRNA vaccine expressing membrane-anchored RBD elicits broad and durable immunity against SARS-CoV-2 variants of concern. Nat. Commun. 14, 2810 (2023).
Lu, H. H. et al. Enhanced durability of a Zika virus self-amplifying RNA vaccine through combinatorial OX40 and 4-1BB agonism. JCI Insight 10, e187405 (2025).
Aboshi, M. et al. Safety and immunogenicity of VLPCOV-02, a SARS-CoV-2 self-amplifying RNA vaccine with a modified base, 5-methylcytosine. iScience 27, 108964 (2024).
Tse, S. W. et al. mRNA-encoded, constitutively active STING(V155M) is a potent genetic adjuvant of antigen-specific CD8+ T cell response. Mol. Ther. 29, 2227–2238 (2021).
Aunins, E. A. et al. An Il12 mRNA-LNP adjuvant enhances mRNA vaccine-induced CD8 T cell responses. Sci. Immunol. 10, eads1328 (2025).
Han, X. et al. Adjuvant lipidoid-substituted lipid nanoparticles augment the immunogenicity of SARS-CoV-2 mRNA vaccines. Nat. Nanotechnol. 18, 1105–1114 (2023).
Misra, B. et al. TLR7-adjuvanted ionizable lipid nanoparticles for mRNA vaccine delivery. AAPS J. 27, 80 (2025).
Svensson, M. et al. mRNA-LNP vaccine strategies: effects of adjuvants on non-parenchymal liver cells and tolerance. Mol. Ther. Methods Clin. Dev. 33, 101427 (2025).
August, A. et al. A phase 1 trial of lipid-encapsulated mRNA encoding a monoclonal antibody with neutralizing activity against Chikungunya virus. Nat. Med. 27, 2224–2233 (2021).
Waldman, A. D., Fritz, J. M. & Lenardo, M. J. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat. Rev. Immunol. 20, 651–668 (2020).
Velu, V. et al. Enhancing SIV-specific immunity in vivo by PD-1 blockade. Nature 458, 206–210 (2009).
Barber, D. L. et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687 (2006).
Codarri Deak, L. et al. PD-1-cis IL-2R agonism yields better effectors from stem-like CD8+ T cells. Nature 610, 161–172 (2022).
Hashimoto, M. et al. PD-1 combination therapy with IL-2 modifies CD8+ T cell exhaustion program. Nature 610, 173–181 (2022).
Harper, J. et al. CTLA-4 and PD-1 dual blockade induces SIV reactivation without control of rebound after antiretroviral therapy interruption. Nat. Med. 26, 519–528 (2020).
Pereira Ribeiro, S. et al. Dual blockade of IL-10 and PD-1 leads to control of SIV viral rebound following analytical treatment interruption. Nat. Immunol. 25, 1900–1912 (2024). This study shows that dual blockade of IL-10 and PD1 in ART-treated, SIV-infected rhesus macaques can achieve durable post-treatment control, with lower viraemia after treatment interruption linked to higher frequencies of memory T cells expressing TCF1.
Pauken, K. E. et al. Epigenetic stability of exhausted T cells limits durability of reinvigoration by PD-1 blockade. Science 354, 1160–1165 (2016).
Song, J. et al. Targeting epigenetic regulators as a promising avenue to overcome cancer therapy resistance. Signal Transduct. Target. Ther. 10, 219 (2025).
Gaebler, C. et al. Prolonged viral suppression with anti-HIV-1 antibody therapy. Nature 606, 368–374 (2022).
Mendoza, P. et al. Combination therapy with anti-HIV-1 antibodies maintains viral suppression. Nature 561, 479–484 (2018).
Niessl, J. et al. Combination anti-HIV-1 antibody therapy is associated with increased virus-specific T cell immunity. Nat. Med. 26, 222–227 (2020).
Kiani, Z. et al. CD8+ T cell stemness precedes post-intervention control of HIV viremia. Nature 650, 196–204 (2026).
Michaud, H.-A. et al. A crucial role for infected-cell/antibody immune complexes in the enhancement of endogenous antiviral immunity by short passive immunotherapy. PLoS Pathog. 6, e1000948 (2010).
Nishimura, Y. et al. Immunotherapy during the acute SHIV infection of macaques confers long-term suppression of viremia. J. Exp. Med. 218, e20201214 (2020).
Rosás-Umbert, M. et al. Administration of broadly neutralizing anti-HIV-1 antibodies at ART initiation maintains long-term CD8+ T cell immunity. Nat. Commun. 13, 6473 (2022).
Gunst, J. D. et al. Early intervention with 3BNC117 and romidepsin at antiretroviral treatment initiation in people with HIV-1: a phase 1b/2a, randomized trial. Nat. Med. 28, 2424–2435 (2022).
Lim, S.-Y. et al. Induction of durable remission by dual immunotherapy in SHIV-infected ART-suppressed macaques. Science 383, 1104–1111 (2024).
Saez-Cirion, A. & Sereti, I. Immunometabolism and HIV-1 pathogenesis: food for thought. Nat. Rev. Immunol. 21, 5–19 (2021).
Alrubayyi, A. et al. Functional restoration of exhausted CD8 T cells in chronic HIV-1 infection by targeting mitochondrial dysfunction. Front. Immunol. 13, 908697 (2022).
Perdomo-Celis, F. et al. Reprogramming dysfunctional CD8+ T cells to promote properties associated with natural HIV control. J. Clin. Invest. 132, e157549 (2023). This proof-of-concept study demonstrates that dysfunctional CD8⁺ T cells from HIV-1 non-controllers can be reprogrammed to enhance stem-like, metabolic and antiviral features characteristic of natural HIV controllers.
Gramatica, A. et al. EZH2 inhibition mitigates HIV immune evasion, reduces reservoir formation, and promotes skewing of CD8+ T cells toward less-exhausted phenotypes. Cell Rep. 44, 115652 (2025).
Perdomo-Celis, F. et al. Impact of rosuvastatin on the memory potential and functionality of CD8+ T cells from people with HIV. eBioMedicine 114, 105672 (2025).
Cantini, L. et al. High-intensity statins are associated with improved clinical activity of PD-1 inhibitors in malignant pleural mesothelioma and advanced non-small cell lung cancer patients. Eur. J. Cancer 144, 41–48 (2021).
Henrich, T. J. et al. Sirolimus reduces T cell cycling, immune checkpoint marker expression, and HIV-1 DNA in people with HIV. Cell Rep. Med. 5, 101745 (2024).
Mannick, J. B. et al. mTOR inhibition improves immune function in the elderly. Sci. Transl. Med. 6, 268ra179 (2014).
Deeks, S. G. HIV: shock and kill. Nature 487, 439–440 (2012).
Nardo, D., Maddox, E. G. & Riley, J. L. Cell therapies for viral diseases: a new frontier. Semin. Immunopathol. 47, 5 (2025).
Kulpa, D. A., Paiardini, M. & Silvestri, G. Immune-mediated strategies to solving the HIV reservoir problem. Nat. Rev. Immunol. 25, 542–553 (2025).
Ward, A. R., Mota, T. M. & Jones, R. B. Immunological approaches to HIV cure. Semin. Immunol. 51, 101412 (2021).
Hataye, J. M. et al. Principles governing establishment versus collapse of HIV-1 cellular spread. Cell Host Microbe 26, 748–763.e20 (2019).
Gong, Q. et al. Multi-omic profiling reveals age-related immune dynamics in healthy adults. Nature 648, 696–706 (2025).
Briceno, O. et al. Reduced naive CD8+ T-cell priming efficacy in elderly adults. Aging Cell 15, 14–21 (2016).
Sturmlechner, I. et al. Antigen specificity shapes distinct aging trajectories of memory CD8+ T cells. Nat. Commun. 16, 6394 (2025).
Cevirgel, A. et al. Pre-vaccination immunotypes and immune entropy are indicators of multiple vaccine responsiveness. Aging Cell 24, e70151 (2025).
Fulop, T., Larbi, A., Hirokawa, K., Cohen, A. A. & Witkowski, J. M. Immunosenescence is both functional/adaptive and dysfunctional/maladaptive. Semin. Immunopathol. 42, 521–536 (2020).
Sacha, J. B. et al. Gag-specific CD8+ T lymphocytes recognize infected cells before AIDS-virus integration and viral protein expression. J. Immunol. 178, 2746–2754 (2007).
Chen, H. et al. TCR clonotypes modulate the protective effect of HLA class I molecules in HIV-1 infection. Nat. Immunol. 13, 691–700 (2012).
Quinones-Parra, S. M. et al. Molecular determinants of cross-strain influenza A virus recognition by alphabeta T cell receptors. Sci. Immunol. 10, eadn3805 (2025).
Koutsakos, M. et al. Human CD8+ T cell cross-reactivity across influenza A, B and C viruses. Nat. Immunol. 20, 613–625 (2019).
Letourneau, S. et al. Design and pre-clinical evaluation of a universal HIV-1 vaccine. PLoS ONE 2, e984 (2007).
Gaiha, G. D. et al. Structural topology defines protective CD8+ T cell epitopes in the HIV proteome. Science 364, 480–484 (2019).
Nathan, A. et al. Structure-guided T cell vaccine design for SARS-CoV-2 variants and sarbecoviruses. Cell 184, 4401–4413.e10 (2021).
Moyo, N. et al. Long-term follow up of human T-cell responses to conserved HIV-1 regions elicited by DNA/simian adenovirus/MVA vaccine regimens. PLoS ONE 12, e0181382 (2017).
Santra, S. et al. Mosaic vaccines elicit CD8+ T lymphocyte responses that confer enhanced immune coverage of diverse HIV strains in monkeys. Nat. Med. 16, 324–328 (2010).
Zou, C. et al. Effective suppression of HIV-1 replication by cytotoxic T lymphocytes specific for Pol epitopes in conserved mosaic vaccine immunogens. J. Virol. 93, e02142-18 (2019).
Park, H. et al. Diversifying T-cell responses: safeguarding against pandemic influenza with mosaic nucleoprotein. J. Virol. 99, e0086724 (2025).
Ondondo, B. et al. Novel conserved-region T-cell mosaic vaccine with high global HIV-1 coverage is recognized by protective responses in untreated infection. Mol. Ther. 24, 832–842 (2016).
Chanda, C. et al. Safety and broad immunogenicity of HIVconsvX conserved mosaic candidate T-cell vaccines vectored by ChAdOx1 and MVA in HIV-CORE 006: a double-blind, randomised, placebo-controlled phase 1 trial in healthy adults living without HIV-1 in eastern and southern Africa. Lancet Microbe 6, 101041 (2025).