Abdel-Malek ZA, Jordan C, Ho T, Upadhyay PR, Fleischer A, Hamzavi I. The enigma and challenges of vitiligo pathophysiology and treatment. Pigment Cell Melanoma Res. 2020;33:778–87.
Chen J, Li S, Li C. Mechanisms of melanocyte death in vitiligo. Med Res Rev. 2021;41:1138–66.
Xuan Y, Yang Y, Xiang L, Zhang C. The role of oxidative stress in the pathogenesis of vitiligo: a culprit for melanocyte death. Oxid Med Cell Longev. 2022;2022:8498472.
Touni AA, Shivde RS, Echuri H, Abdel-Aziz RTA, Abdel-Wahab H, Kundu RV, et al. Melanocyte–keratinocyte cross-talk in vitiligo. Front Med. 2023;10:1176781.
Bastonini E, Bellei B, Filoni A, Kovacs D, Iacovelli P, Picardo M. Involvement of non-melanocytic skin cells in vitiligo. Exp Dermatol. 2019;28:667–73.
Richmond JM, Bangari DS, Essien KI, Currimbhoy SD, Groom JR, Pandya AG, et al. Keratinocyte-derived chemokines orchestrate T-cell positioning in the epidermis during vitiligo and may serve as biomarkers of disease. J Invest Dermatol. 2017;137:350–8.
Li S, Zhu G, Yang Y, Jian Z, Guo S, Dai W, et al. Oxidative stress drives CD8+ T-cell skin trafficking in patients with vitiligo through CXCL16 upregulation by activating the unfolded protein response in keratinocytes. J Allergy Clin Immunol. 2017;140:177–89.e9.
Li S, Kang P, Zhang W, Jian Z, Zhang Q, Yi X, et al. Activated NLRP3 inflammasome in keratinocytes promotes cutaneous T-cell response in patients with vitiligo. J Allergy Clin Immunol. 2020;145:632–45.
Chen X, Guo W, Chang Y, Chen J, Kang P, Yi X, et al. Oxidative stress-induced IL-15 trans-presentation in keratinocytes contributes to CD8+ T-cell activation via JAK–STAT pathway in vitiligo. Free Radic Biol Med. 2019;139:80–91.
Kang P, Wang Y, Chen J, Chang Y, Zhang W, Cui T, et al. TRPM2-dependent autophagy inhibition exacerbates oxidative stress-induced CXCL16 secretion by keratinocytes in vitiligo. J Pathol. 2024;262:441–53.
Newman LE, Shadel GS. Mitochondrial DNA release in innate immune signaling. Annu Rev Biochem. 2023;92:299–332.
Quan S, Fu X, Cai H, Ren Z, Xu Y, Jia L. The neuroimmune nexus: unraveling the role of the mtDNA–cGAS–STING signal pathway in Alzheimer’s disease. Mol Neurodegener. 2025;20:25.
Vaseghi H, Houshmand M, Jadali Z. Increased levels of mitochondrial DNA copy number in patients with vitiligo. Clin Exp Dermatol. 2017;42:749–54.
Sant’Anna-Silva ACB, Botton T, Rossi A, Dobner J, Bzioueche H, Thach N, et al. Vitiligo auto-immune response upon oxidative stress-related mitochondrial DNA release opens up new therapeutic strategies. Clin Transl Med. 2024;14:e1810.
Xu X, Lu X, Zheng Y, Xie Y, Lai W. Cytosolic mtDNA–cGAS–STING axis mediates melanocyte pyroptosis to promote CD8+ T-cell activation in vitiligo. J Dermatol Sci. 2025;117:61–70.
Wu T, Chen X, Fan J, Ye P, Zhang J, Wang Z, et al. Oxidative stress-induced release of mitochondrial DNA promotes vitiligo progression by activating the cGAS–STING pathway in monocytes. Free Radic Biol Med. 2025;235:43–55.
McArthur K, Whitehead LW, Heddleston JM, Li L, Padman BS, Oorschot V, et al. BAK/BAX macropores facilitate mitochondrial herniation and mtDNA efflux during apoptosis. Science. 2018;359:eaao6047.
Riley JS, Quarato G, Cloix C, Lopez J, O’Prey J, Pearson M, et al. Mitochondrial inner membrane permeabilisation enables mtDNA release during apoptosis. EMBO J. 2018;37:e99238.
He B, Yu H, Liu S, Wan H, Fu S, Liu S, et al. Mitochondrial cristae architecture protects against mtDNA release and inflammation. Cell Rep. 2022;41:111774.
Xian H, Watari K, Sanchez-Lopez E, Offenberger J, Onyuru J, Sampath H, et al. Oxidized DNA fragments exit mitochondria via mPTP- and VDAC-dependent channels to activate NLRP3 inflammasome and interferon signaling. Immunity. 2022;55:1370–85.e8.
Liu H, Fan H, He P, Zhuang H, Liu X, Chen M, et al. Prohibitin 1 regulates mtDNA release and downstream inflammatory responses. EMBO J. 2022;41:e111173.
Kim J, Gupta R, Blanco LP, Yang S, Shteinfer-Kuzmine A, Wang K, et al. VDAC oligomers form mitochondrial pores to release mtDNA fragments and promote lupus-like disease. Science. 2019;366:1531–6.
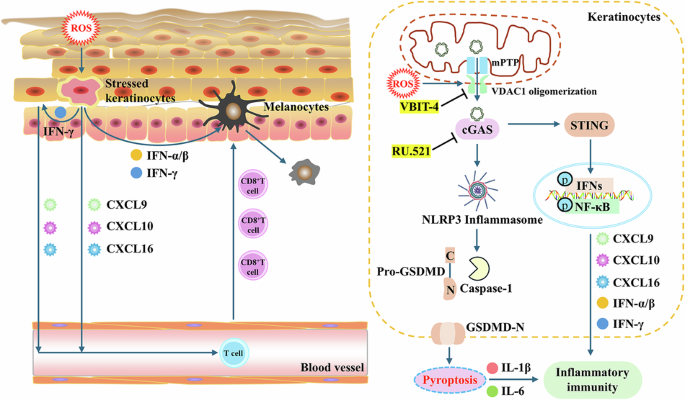
Gao R, Meng D, Zhao Z, Xue H, Hu N, Jiang P, et al. VDAC1 oligomerization-mediated mtDNA release under sublethal oxidative stress: a novel inflammatory mechanism in vitiligo. Free Radic Biol Med. 2025;241:1–13.
De Gaetano A, Solodka K, Zanini G, Selleri V, Mattioli AV, Nasi M, et al. Molecular mechanisms of mtDNA-mediated inflammation. Cells. 2021;10:2898.
Decout A, Katz JD, Venkatraman S, Ablasser A. The cGAS–STING pathway as a therapeutic target in inflammatory diseases. Nat Rev Immunol. 2021;21:548–69.
Zhang W, Li G, Luo R, Lei J, Song Y, Wang B, et al. Cytosolic escape of mitochondrial DNA triggers cGAS–STING–NLRP3 axis-dependent nucleus pulposus cell pyroptosis. Exp Mol Med. 2022;54:129–42.
Lv J, Zou K, Yin C, Xu W, Meng D, Zhang H, et al. Daidzin suppresses melanogenesis through ERK and AKT signaling pathways mediated MITF proteasomal degradation. Exp Mol Pathol. 2025;143:104986.
Lv J, Fu Y, Cao Y, Jiang S, Yang Y, Song G, et al. Isoliquiritigenin inhibits melanogenesis, melanocyte dendricity and melanosome transport via ERK-mediated MITF degradation. Exp Dermatol. 2020;29:149–57.
Lv J, Zhang X, An X, Cao Y, Meng D, Zou K, et al. Inhibition of VDAC1 oligomerization promotes pigmentation through the CaMK–CRTCs/CREB–MITF pathway. Exp Cell Res. 2024;434:113874.
Li J, Wu G, Su H, Liang M, Cen S, Liao Y, et al. Hsp90 C-terminal domain inhibition enhances ferroptosis by disrupting GPX4–VDAC1 interaction to increase HMOX1 release from oligomerized VDAC1 channels. Redox Biol. 2025;85:103672.
Hashiguchi K, Zhang-Akiyama QM. Establishment of human cell lines lacking mitochondrial DNA. Methods Mol Biol. 2009;554:383–91.
Han J, Zhang X, Cai M, Tian F, Xu Y, Chen H, et al. TSPO deficiency exacerbates acute lung injury via NLRP3 inflammasome-mediated pyroptosis. Chin Med J. 2024;137:1592–602.
Yao Y, Wang Z, Li J, Peng A, Cao Y, Liang N, et al. Pyroptosis and its role in autoimmune skin disease. Exp Dermatol. 2024;33:e15135.
Lian N, Chen Y, Chen S, Zhang Y, Chen H, Yang Y, et al. Gasdermin D–mediated keratinocyte pyroptosis as a key step in psoriasis pathogenesis. Cell Death Dis. 2023;14:595.
Liu H, Wang Y, Le Q, Tong J, Wang H. The IFN-γ–CXCL9/CXCL10–CXCR3 axis in vitiligo: pathological mechanism and treatment. Eur J Immunol. 2024;54:e2250281.
Jacquemin C, Rambert J, Guillet S, Thiolat D, Boukhedouni N, Doutre MS, et al. Heat shock protein 70 potentiates interferon-α production by plasmacytoid dendritic cells: relevance for cutaneous lupus and vitiligo pathogenesis. Br J Dermatol. 2017;177:1367–75.
Tulic MK, Cavazza E, Cheli Y, Jacquel A, Luci C, Cardot-Leccia N, et al. Innate lymphocyte-induced CXCR3B-mediated melanocyte apoptosis initiates T-cell autoreactivity in vitiligo. Nat Commun. 2019;10:2178.
Liu J, Zhou J, Luan Y, Li X, Meng X, Liao W, et al. cGAS–STING, inflammasomes and pyroptosis: an overview of crosstalk mechanism. Cell Commun Signal. 2024;22:22.
Luo X, Zhao Y, Luo Y, Lai J, Ji J, Huang J, et al. Cytosolic mtDNA–cGAS–STING axis contributes to sepsis-induced acute kidney injury via activating NLRP3 inflammasome. Clin Exp Nephrol. 2024;28:375–90.
Lai P, Liu L, Bancaro N, Troiani M, Cali B, Li Y, et al. Mitochondrial DNA released by senescent tumor cells enhances PMN-MDSC–driven immunosuppression through the cGAS–STING pathway. Immunity. 2025;58:811–25.e7.
Verma A, Pittala S, Alhozeel B, Shteinfer-Kuzmine A, Ohana E, Gupta R, et al. The role of mitochondrial protein VDAC1 in inflammatory bowel disease: a potential therapeutic target. Mol Ther. 2022;30:726–44.
Hu H, Guo L, Overholser J, Wang X. Mitochondrial VDAC1: a potential therapeutic target of inflammation-related diseases and clinical opportunities. Cells. 2022;11:3174.