Certain models of a popular family-friendly SUV have been recalled, and sales of the line have been halted as the manufacturer investigates a 2-year-old’s death, according to reports.
In other recent recall announcements, a pharmaceutical company has recalled nearly 90,000 bottles of children’s ibuprofen due to a mysterious contamination; a frozen product sold by Aldi may contain rodent hair; and federal regulators have added more distributors of vitamins and supplements to a growing list of “repeat offenders” who have failed to disclose active ingredients that are used in FDA-approved erectile dysfunction drugs.
Sources for this report are the Food and Drug Administration, the Consumer Product Safety Commission, the National Highway Traffic Safety Administration, Patch reporting, and other news outlets.
Find out what’s happening in Across Americafor free with the latest updates from Patch.Carmaker Halts Sales After Toddler’s Death
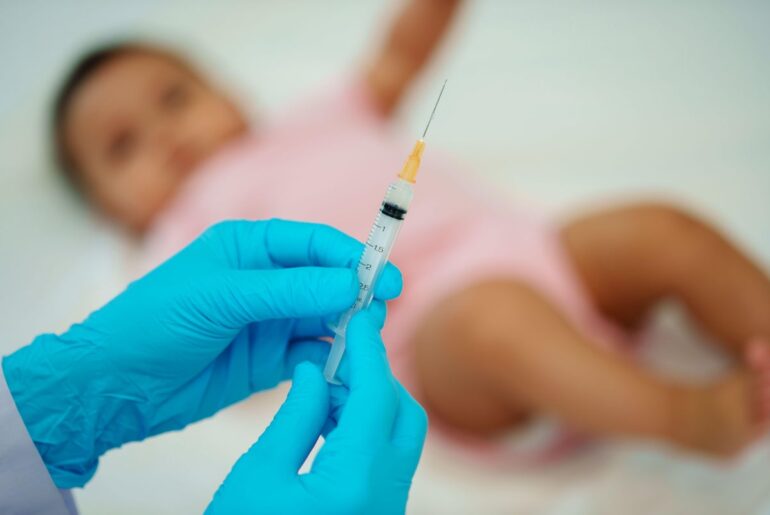
Hyundai is recalling about 61,000 of its model-year 2026 Palisade vehicles and halting sales of certain models after a fatal incident involving a child, according to multiple news outlets, including Reuters.
The action comes after a March 7 incident in which a 2-year-old girl from Ohio died. Hyundai said the circumstances remain under investigation and that it does not yet have full details of what happened, but that the problem involves second- and third-row power seats that may fail to detect an occupant or object.
Find out what’s happening in Across Americafor free with the latest updates from Patch.
“Hyundai is aware of a tragic incident involving a Palisade,” a spokesperson said in a statement. “While Hyundai does not yet have the full details and the incident is still under investigation, a young child lost her life. Hyundai extends its deepest sympathies to her family.”
Hyundai cautioned owners to make sure no children or objects are in the back two rows when using the power functions, advising, “When using the second-row one‑touch tilt‑and‑slide feature to access the third row, customers should avoid pressing the seatback button during entry or exit.”
The company is developing a free remedy, expected to include a software update to improve seat detection.
Both the Calligraphy and Limited trims are involved in the recall, which is being conducted in coordination with the National Highway Traffic Safety Administration. With vehicles from Canada included, the recall covers about 70,000 vehicles.
Previous complaints about the vehicle included one from December in which a passenger was injured while getting settled in a middle-row seat, according to Consumer Reports.
“A passenger got into the passenger-side middle-row seat, and the seat fold-up button was accidentally bumped while the person was trying to get situated and buckled,” according to the report. “The seat began moving and folding forward, pinning the passenger between the seat they were in and the back of the front seat.
“This incident hurt the passenger, who had the force of the seat against her, and it frightened her very badly. If the door had been locked, the passenger would not have been able to be helped out and could have sustained worse injuries.”
Hyundai also said that it will work with dealers to “offer interested customers a rental vehicle until a full remedy is available.”
Anyone with additional questions can call Hyundai at 800-633-5151.
Nearly 90,000 bottles of children’s ibuprofen have been recalled after customers reported finding a gel-like mass and black particles in the product, according to a Food and Drug Administration enforcement report.
Strides Pharma Inc., based in Bridgewater, New Jersey, recalled 82,592 bottles of Children’s Ibuprofen Oral Suspension, the FDA said. The agency classified it a Class II recall, which means “use of or exposure to a violative product may cause temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is remote.”
Possible Rodent Hair Triggers Recall
Simply Nature Spinach Bites sold in Aldi grocery stores in Pennsylvania and Maryland have been recalled because they may contain rodent hair, the Food and Drug Administration announced on March 16.
Dr. Prager’s Sensible Foods Inc. of New Jersey voluntarily recalled the products but didn’t disclose how the frozen spinach products may have become contaminated with rodent hair.
According to the FDA, the recall is considered Class II, which means exposure to the recalled item could cause temporary or medically reversible health consequences. The risk of long-term impacts is minimal. No illnesses or hospitalizations have been reported, according to the announcement.
The lot number of the recalled 12-ounce product is G25CF-02B. The product number is AL-SP UPC: 099100247992.
Ford Recalls More Than 840K Vehicles
Ford Motor Co. is recalling 849,310 Bronco and Edge models because of a defect that prevents the rearview camera image from properly displaying, according to a notice posted by the National Highway Traffic Safety Administration.
The accessory protocol interface module may overheat, preventing the image from displaying correctly while the car is in reverse, increasing the risk of a crash, according to the notice.
The affected vehicles are Ford Broncos from model years 2021 to 2026, and Ford Edge vehicles from model years 2021 to 2024.
Owners of vehicles included in the recall are expected to receive notifications on March 30. Software updates will be made at no charge to customers, whether at a dealership or remotely via the internet.
Vitamins and supplements sold under the Boner Bear Honey, Red Bull Extreme and Blue Bull Extreme labels have been recalled because they contain undeclared active ingredients used in erectile dysfunction drugs, according to the Food and Drug Administration.
The products contain sildenafil and/or tadalafil, which are active ingredients in the FDA-approved prescription drugs Viagra and Cialis, respectively. Their use is restricted to those under the care of a licensed health professional.
“These undeclared ingredients may interact with nitrates found in some prescription drugs, such as nitroglycerin, and may lower blood pressure to dangerous levels,” the FDA said. “People with diabetes, high blood pressure, high cholesterol or heart disease often take nitrates.”
No reports of adverse reactions have been reported.
The products were marketed as sexual enhancement honey products and are packaged in plastic tubes of single servings. More information is found here.
Consumers should stop using and discard the products. If they’ve had adverse reactions, they should consult their health care provider, the FDA said.
The distributor, Tampa, Florida-based Pure Vitamins and Natural Supplements, LLC, is listed in a database of fraudulent health care products. These companies have been cited in warning letters, online advisory letters, recalls, public notifications and news releases, according to the FDA.
In a related recall, Albuquerque, New Mexico-based Primal Supplements Group LLC is voluntarily recalling all orders of Primal Herbs Volume placed between July 2 and September 19, 2025, after an FDA lab analysis confirmed the presence sildenafil, which was not listed on the product label.
30 Burn Injuries From Name-Brand Gas Ranges
Some 174,800 Frigidaire gas ranges manufactured by Electrolux Group have been recalled after dozens of consumers reported they had been burned, according to the Consumer Product Safety Commission.
The recall affects ranges in which the oven’s bake burner can ignite with a delay, creating a risk of flames escaping unexpectedly and injuring users, according to the agency. At least 30 people were burned when the oven’s bake burner delayed ignition. More than 60 reports overall were taken.
Consumers are urged to check whether their appliance is included and to contact Electrolux for a free repair.
The affected units were sold nationwide at major appliance retailers, and model and serial numbers can typically be found on the range itself, often inside the oven door or storage drawer.
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.