Medical research.getty-images-KSRaPx2SAe4-unsplash.jpg
Medical research.getty-images-KSRaPx2SAe4-unsplash.jpg
In a discovery that reshapes fundamental concepts in immunology, researchers led by Pavan Reddy, MD, director of the Dan L Duncan Comprehensive Cancer Center at Baylor College of Medicine, have identified a novel way T-cells can attack cancer and foreign cells—opening new avenues for cancer immunotherapy and transplantation medicine.
The findings, published in Nature Immunology on March 24, 2026, emerged from a close collaboration between Reddy’s group and teams helmed by Dr. Arul M. Chinnaiyan, MD, Ph.D., and Marcin Cieslik, Ph.D., at the University of Michigan Rogel Cancer Center.
For decades, textbooks have described two distinct roles for molecules called major histocompatibility complexes (MHC): MHC class I was thought to exclusively present danger signals to CD8+ ‘killer’ T cells, while MHC class II molecules activated CD4+ ‘helper’ T cells. This clear division has shaped generations of research and clinical strategies in cancer and transplant immunology.

 Pavan Reddy, MD, director of the Dan L Duncan Comprehensive Cancer Center, Baylor College of Medicine, Houston, Texas. Photo courtesy © 2025 – 2026 Baylor College of Medicine, Houston, Texas. Used with permission
Pavan Reddy, MD, director of the Dan L Duncan Comprehensive Cancer Center, Baylor College of Medicine, Houston, Texas. Photo courtesy © 2025 – 2026 Baylor College of Medicine, Houston, Texas. Used with permission
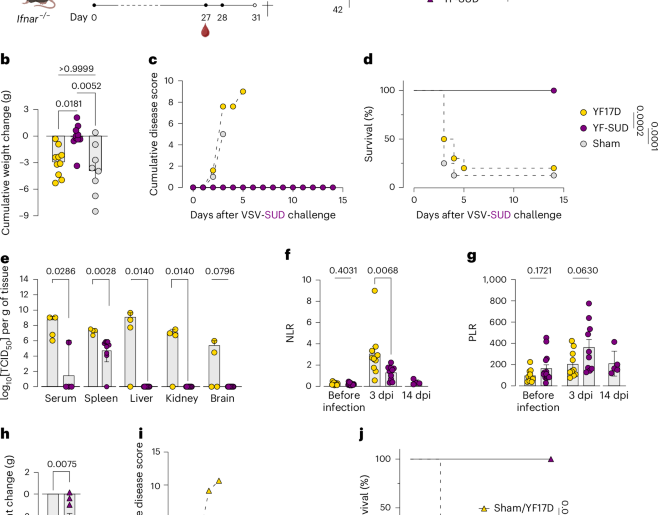
However, the new study, driven by graduate students Emma Lauder and Meng-Chih Wu at Baylor, and Mahnoor Gondal at Michigan, among others, and supported by grants from the National Institutes of Health and the Cancer Prevention and Research Institute of Texas, challenges this paradigm. Using sophisticated transcriptomic profiling and functional experiments in both mouse models and human cells, the researchers discovered that when cancer cells lose MHC class I (a common tactic for evading CD8+ T cell destruction), they unwittingly expose themselves to a different type of immune assault.
Susceptible to ferroptosis
The team found that cancer cells lacking MHC I become more susceptible to ferroptosis—a form of iron-dependent, oxidative cell death—induced by CD4+ T cells. This phenomenon was not limited to cancer, but also appeared in models of graft-versus-host disease, a major risk following bone marrow transplantation.
Advertisement #3
Further analysis by the University of Michigan team connected these findings to real-world outcomes by examining large clinical and genetic datasets from patients treated with immune checkpoint inhibitors for solid tumors. The results showed that reduced MHC I expression correlated with stronger CD4+ T cell-driven anti-tumor effects.
Downregulating MHC1
This research suggests that downregulation of MHC I, long considered a cancer cell’s shield against the immune system, actually increases vulnerability to CD4+ T cell attack. It also points to new therapeutic strategies—enhancing or harnessing CD4+ T cells could bolster immunotherapies, especially for tumors that have evaded CD8+ T cell responses. Conversely, understanding this mechanism could help prevent or manage unwanted immune reactions, such as those seen in transplant rejection.
“Our work, if further validated, will have implications for T cell-mediated immune responses beyond cancer and transplant immunology,” Reddy said.
“This may allow for the development of novel strategies that target MHC class I and CD4+ T cells to leverage the beneficial side of immunity or mitigate unwanted immune responses,” he concluded.
__
Note: The study was a collaborative effort involving Emma Lauder, Mahnoor Gondal, Meng-Chih Wu, Akira Yamamoto, Laure Maneix, Dongchang Zhao, and Yaping Sun, with institutional affiliations at Baylor College of Medicine, the University of Michigan, and the Howard Hughes Medical Institute.
Reference
[1] Lauder, E., Gondal, M., Wu, MC. et al. MHC class I on target cells regulates CD4+ T cell-mediated immunity. Nat Immunol (2026). https://doi.org/10.1038/s41590-026-02480-z
Featured image: Medical research. Photo courtesy © 2020 – 2026 Unsplash+ license. Used with permission
DOI
Advertisement #5