Photo Credit NIAID
#19,095
One of the key questions in our post-pandemic world is how much immunity have we gained against other emerging coronaviruses due to our continual exposure to SARS-CoV-2 and/or COVID vaccines over the past 6 years?
It is not an easy question to answer for a lot of reasons, including:
First and foremost, COVID is a sarbecovirus, which is just one type of coronavirus. MERS-CoV is another (Merbecovirus), which uses an entirely different receptor cell, making cross-immunity unlikely. Second, even among sarbecoviruses there are at least 4 lineages (clade 1a, 1b, 2, & 3); SARS-CoV was clade 1a, while SARS-CoV-2 was clade 1b. Clades 2 & 3 are more antigenically distant.Third, COVID immunity wanes relatively quickly. While re-exposures or vaccinations can `boost’ immunity, variant drift and/or declining vaccine uptake could erode immunity. In other words, there are a lot of moving parts. But in a world teeming with literally dozens of known coronaviruses circulating in the wild (see here, here, here, and here), any cross-immunity is better than none. All of which brings us to a new study, published in Nature Comms, where researchers tested cross‑neutralisation of four zoonotic sarbecovirus spikes using human sera from naïve, infected, vaccinated, and hybrid immunity groups.
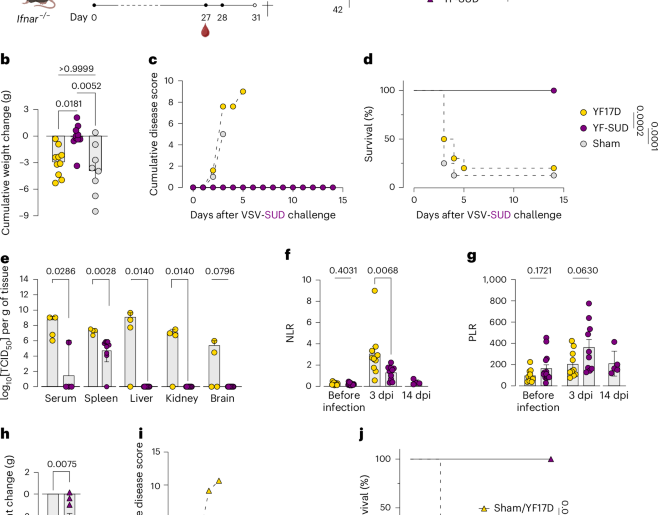
All four test viruses (SARS‑CoV, Rs4084, GX/P1E, RaTG13) belonged to either clade 1a or 1b, had relatively high spike similarity to SARS-CoV-2, and known ACE2 usage. Of the 4 tested, the 2002 SARS-CoV showed the least cross-neutralization (30%), while RaTG13 showed the most (79%).
Whether, and how much, cross immunity would extend to more antigenically distant – or non-ACE binding – coronaviruses is unknown.
What they found was cross‑neutralisation increases with number of vaccine doses and is highest in hybrid immunity (hx of infection & vaccination). They write:The highest levels of cross-neutralisation were consistently
observed in patients with hybrid immunity, suggesting that vaccine breakdown infections by immune evasive SARS-CoV-2 variants may
have a strong protective effect against SARS-CoV-X infection, and that
vaccination should be encouraged even in patients with a history of
prior infection. In unvaccinated individuals with a history of infection,
the strength of cross-neutralisation was lower than in patients with
hybrid immunity and was determined by the SARS-CoV-2 infecting
strain. This is consistent with findings that protection conferred by
natural infection varies over time and is influenced by the antigenic
evolution of SARS-CoV-2, with pre-Omicron infections offering durable
immunity, and immunity following Omicron infection waning more
rapidly, likely due to increased immune escape38. Using this data, the researchers then modeled how COVID and a hypothetical new “SARS‑CoV‑X” might spread in a population like Scotland’s, and determined that our current global immunity makes it more difficult for a SARS-like (ACE-receptor-using) virus to establish itself compared to a naïve community.
Not impossible, but apparently it provides a non-trivial barrier.
This is a detailed, complex, and nuanced study and deserves to be read in its entirety, as I’ve only scratched the surface. I’ll have a bit more after the break.
Ryan M. Imrie, Laura A. Bissett, Savitha Raveendran, Maria Manali, Julien A. R. Amat, Laura Mojsiejczuk, Nicola Logan, Andrew Park, Marc Baguelin, Mafalda Viana, Brian J. Willett & Pablo R. Murcia
Nature Communications volume 17, Article number: 2248 (2026) Cite this article
Abstract
Infections by endemic viruses, and the vaccines used to control them, often provide cross-protection against related viruses, potentially altering the transmission dynamics and likelihood of emergence of new zoonotic viruses with pandemic potential. Here, we investigate how population immunity after the COVID-19 pandemic has impacted the likelihood of emergence of a novel sarbecovirus, termed SARS-CoV-X.
To this end, we combined empirical cross-neutralisation data with mathematical modelling to identify key immunological and epidemiological factors shaping sarbecovirus emergence. We show that sera from individuals with different COVID-19 immunological histories contained cross-neutralising antibodies against the spike (S) protein of multiple zoonotic sarbecoviruses.
Simulations parameterised by these data predict that the likelihood of emergence of a novel sarbecovirus has been reduced significantly by population cross-immunity, with outcomes determined by the extent of cross-protection and R0 of the novel virus.
Preventative vaccination against SARS-CoV-X using available COVID-19 vaccines can help resist emergence even in the presence of co-circulating SARS-CoV-2. However, a theoretical vaccine with high specificity to SARS-CoV-2 can increase emergence probability by suppressing SARS-CoV-2 prevalence and, by extension, levels of natural cross-protection.
Overall, SARS-CoV-2 circulation and vaccination have generated widespread immunity against related sarbecoviruses, creating an immunological barrier to novel sarbecovirus emergence in humans.
While these findings appear to do little to negate the threat from MERS-CoV (see (Referral) Nature: Human MERS-CoV cases are falling but pose an ongoing pandemic threat), it does suggest that we likely now carry some degree of protection against a narrow – but important – range of sarbecoviruses.
While not exactly a get-out-of-pandemic-free card – when it comes to emerging viruses – any amount of immunity has to beat no immunity at all.