In contrast to chemokines, which are proteins, chemoattracting metabolites are small molecules, fats or ions that can attract various cell types, including immune cells, to specific locations in the body.
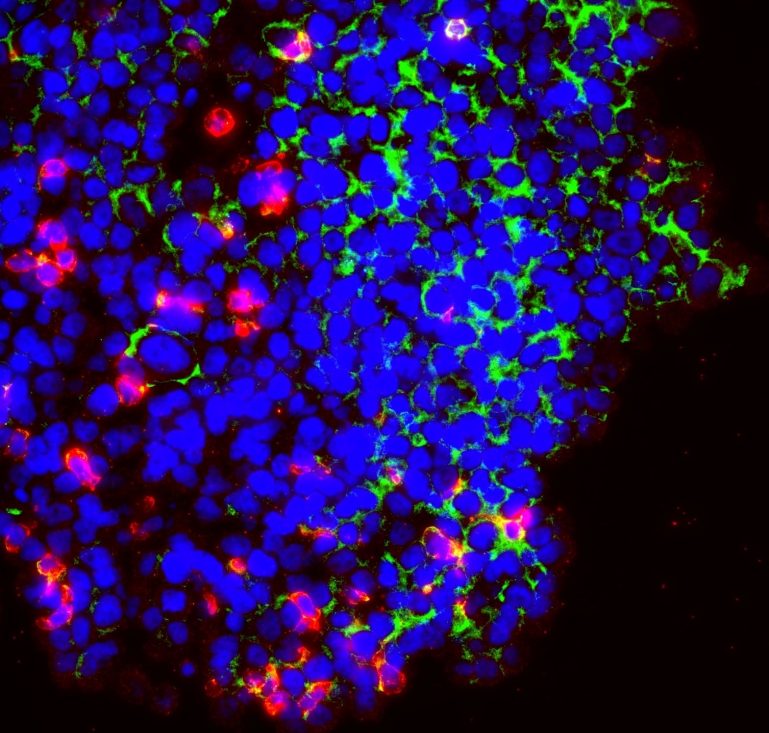
Jerby and her colleagues found that NK cells that had been engineered to express one of six genes were consistently better at infiltrating breast and ovarian tumors in the animals, migrating specifically to cancer cells and the factors they release. Because the genes they identified encode proteins belonging to a class of receptors called G-protein coupled receptors, or GPCRs, the researchers coined the term tumor-homing GPRs, or thGPRs.
The thGPRs identified are known to recognize and migrate toward specific types of phospholipids, fatty acids and derivatives of cholesterol, which are generated by cancer cells in their headlong dash to proliferate. The study’s analyses of patient data indicate that these metabolites recruit tumor-friendly immune cells, creating an environment that supports tumor growth and resists drug treatment. But they can also serve as a smoking gun, indicating a tumor is nearby.
“It’s been known for decades that cancer cells are metabolically unique in many ways,” Jerby said. “Clearly there are certain metabolic features that either directly aid tumor growth or are a byproduct of uncontrolled cell proliferation. These features are routinely exploited for cancer diagnostic scans such as PET imaging, which pinpoints areas in the body with high metabolic activity.”
A ‘follow the yellow brick road’ approach
Chemoattracting metabolites stimulate responding cells to migrate toward higher concentrations of the target to reach the signaling cells — a kind of “follow the yellow brick road” approach in which the path widens and becomes easier to follow as the destination is neared. The researchers exploited this feature to equip NK or killer T cells to track down and infiltrate tumors based on their telltale metabolite trails.
Examining this therapeutic approach the researchers focused on one of the thGPRs, GPR183, in breast cancer. GPR183 is a receptor of oxidized forms of cholesterol called GPR183. Engineering NK or T cells to express GPR183 on their surfaces markedly enhanced the ability of the cells to migrate toward cancer cells in laboratory dishes or in mice. Expressing GPR183 on the surface of NK cells, CAR-NK cells, CAR-T cells and other types of tumor reactive T cells led to significantly better tumor control and survival of laboratory mice with breast cancer tumors.
“We saw a more than doubling in the number of complete responses in the animals,” Jerby said. “T cells engineered to express GPR183 on their surfaces were far better at completely eradicating highly aggressive breast tumors. The tumors did not come back, and the mice went back to being healthy.”
Jerby and her lab members are now investigating whether thGPRs can be modified to recognize other tumor metabolites that are not normally chemoattracting as navigation cues, or to have immune cells interpret tumor metabolites as “on switches” to become killing machines only in the tumor. They are also moving toward testing the GPR183-engineered cells in clinical trials and testing the other thGPRs for their therapeutic potential.
“To the best of our knowledge, no one has tried to use cancer metabolism, a hallmark of drug resistance and aggressive tumor growth, to attract cancer-killing immune cells to the tumor,” Jerby said. “But our study uncovered the potential of this approach, and the results are quite promising.”
Jerby is a member of the Stanford Cancer Institute, Bio-X and a Chan Zuckerberg Biohub investigator.
The study was funded by the National Institutes of Health (grant U01HG012069), the Chan Zuckerberg Biohub, Under One Umbrella and the Stanford Cancer Institute, Stanford’s Discovery Innovation Funds, the Ovarian Cancer Research Alliance, the Burroughs Wellcome Fund, the National Research Foundation of Korea, the Ovarian Cancer Research Alliance, the Department of Defense, and Alba Tull Molecular Therapeutics Award for Innovative Medicines.
Stanford University has filed a patent based on the research in which Jerby and Kim are named as inventors.