The food babies eat in their first months of life does more than help them grow. It quietly trains the body for the future.
Scientists have discovered that the shift from milk to solid food can shape how the gut fights disease for years.
Researchers at Baylor College of Medicine, Tongji University, and other institutions found that this early change builds a strong foundation for lifelong health.
A big shift inside the gut
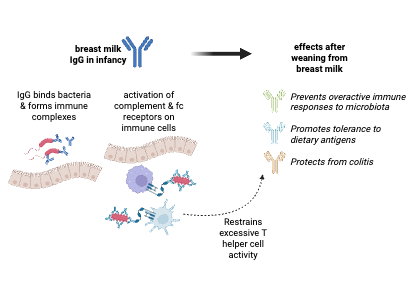
The transition from milk to solid food is called weaning. During this stage, the gut meets many new microbes. These microbes form the gut microbiome and play a key role in health.
“Weaning is a major transition for babies. As milk gives way to solid food, the gut is suddenly exposed to a much wider variety of microbes,” explained Dr. Lanlan Shen from Baylor College of Medicine.
This exposure increases microbial diversity and triggers a short immune response in the gut. This response is not random. It is a planned step that helps the body prepare for future challenges.
A short reaction with long effects
During weaning, the body produces signals like IFNγ and other immune molecules. These signals create a brief inflammatory response known as the weaning reaction.
Inflammation may sound harmful, but in this case, it helps. It acts like a training session for the immune system. It teaches the gut how to respond to microbes and infections.
This reaction happens only during a specific time window in early life. Even though it is short, it leaves long-lasting effects on the body.
The role of stem cells in the gut
The gut lining renews itself every few days. This process depends on intestinal stem cells. These cells live for a long time and keep producing new gut cells.
“Because these stem cells persist for life, durable changes to them could shape gut health for decades,” says Dr. Li Yang, co-author of the study. These cells can store information from early life and pass it on to future cells.
Scientists found that microbes during weaning send signals to these stem cells. These signals change how the cells behave in the future.
How genes get reprogrammed
The study showed that microbes can change gene activity without changing the DNA itself. This happens through a process called DNA methylation. It adds small chemical tags that turn genes on or off.
These changes mostly happen in specific regions of genes, especially those linked to immune functions. For example, MHC class II genes become easier to activate after weaning.
This change does not immediately turn the genes on. Instead, it prepares them to respond faster later. It is like setting up a system in advance.
Creating a memory in the gut
These changes in stem cells create a type of memory. Even when stem cells turn into regular gut cells, this memory stays.
“This process creates an epithelial immune memory that is embedded directly in the gut lining,” Yang said. When the body faces microbes again, these trained cells respond quickly and strongly.
Experiments also showed that cells exposed to early signals reacted faster when tested again later. This proves that the gut remembers its early training.
Why certain bacteria matter
Not all microbes help in the same way. Some bacteria play a stronger role in training the immune system. Gram positive bacteria are especially important because they trigger immune signals like IFNγ.
Other helpful microbes produce substances like short-chain fatty acids. These substances help control gene activity and support healthy gut development.
When these helpful microbes grow after weaning, they support proper immune training. Without them, the process becomes weaker.
What happens when antibiotics interfere
Researchers also studied what happens when antibiotics are used early in life. Antibiotics reduced many helpful bacteria in the gut.
“As adults, antibiotic-exposed mice had lower MHC class II expression in the gut, weaker immune defenses and far greater susceptibility to colitis and colon cancer when challenged,” Shen said.
The study also showed that antibiotic use reduced important bacteria that produce helpful compounds. This disrupted gene regulation and weakened the immune system.
This suggests that disturbing the gut microbiome early can have long term effects on health.
A critical window in early life
Timing plays a key role in this process. The study found that this immune training works best during early life. If similar changes happen later, the effect becomes much weaker.
Scientists also tested transferring healthy microbes at different ages. The results showed that early transfer worked best, while later attempts had limited impact.
This shows that early childhood is a critical window for building strong immunity.
What this means for long-term health
Even though this study was done in mice, it gives important clues about human health. Diseases like Crohn’s disease and ulcerative colitis often appear later in life. However, their roots may begin in early childhood.
“Epidemiological studies already link antibiotic use in infancy with higher risk of these diseases later,” Shen said.
This research suggests that early diet, healthy microbes, and careful use of antibiotics can shape lifelong health. In the future, scientists may design diets or treatments that support healthy gut training in early life.
The study is published in the journal Nature Microbiology.
—–
Like what you read? Subscribe to our newsletter for engaging articles, exclusive content, and the latest updates.
Check us out on EarthSnap, a free app brought to you by Eric Ralls and Earth.com.
—–