Our study highlights the complexity of nutrient deficiencies and their associations with metabolic and clinical complications in patients with CD. The findings emphasize the importance of folate and vitamin B12 in managing systemic complications, including cardiovascular risks and EIMs.
Previous studies have primarily examined B12 and folate deficiencies in relation to clinical activity or anatomical factors. Akbulut reported lower B12 levels with ileal involvement [26], and Huang et al. identified ileal resection as a risk factor for B12 deficiency and shorter disease duration for folate deficiency [27]. Gioxari et al. evaluated serum vitamin status and inflammatory/oxidative biomarkers, although dietary intake was analyzed only as a determinant of vitamin levels [28]. In contrast, our study integrates dietary intake, serum concentrations, systemic complications, and disease behavior according to the Montreal classification, using objective endoscopic or imaging criteria to define disease activity. This approach allowed us to identify associations between vitamin B12 and folate intake, inflammation, EIMs, metabolic risk, and stricturing/penetrating phenotypes, providing a more comprehensive assessment of nutritional and clinical determinants in CD.
We demonstrated that patients had a high protein intake. Patients with active disease require increased protein due to intestinal losses, reduction in lean body mass, drug interactions, and inflammatory processes [21]. However, 67.74% of patients in remission also consumed a high-protein diet, which is relevant since excessive protein intake, particularly from animal sources, may have a deleterious effect on the gut microbiota, an essential component for the development and management of IBD [29]. An excess of nitrogenous compounds in the intestinal lumen stimulates the microbiota to produce harmful substances and promotes the growth of pathogenic strains [30]. Additionally, Lylowska-Szuber et al. have shown that dysbiosis significantly impacts cardiovascular risk in IBD through the production of inflammatory byproducts, increased intestinal permeability, and the promotion of low-grade inflammation [31].
High protein intake may not prevent muscle loss when caloric intake is low, contributing to sarcopenic obesity [21]. In this study, carbohydrate consumption in the lowest tertile (T1) was below recommendations, and diets rich in protein but low in carbohydrates may not provide adequate energy, especially when total energy intake is insufficient. A systematic review found that patients with CD typically consume less energy than recommended while meeting or exceeding protein requirements [32]. This imbalance can lead the body to use protein as an energy source, limiting its availability for muscle maintenance and impairing protein metabolism, particularly in chronic or hypercatabolic conditions [33]. Another relevant aspect is that as protein consumption increases, the substrate is available to form Hcy [34]. Since vitamins B12 and folate are essential for Hcy metabolism, deficiencies in these nutrients can raise Hcy levels, which are associated with increased cardiovascular risk [5, 35].
Although most patients had adequate intake of vitamin B12 and folate, a large proportion of them were found to be at risk for elevated Hcy levels based on their serum B12 levels. It is important to note that adequacy was assessed using Dietary Reference Intakes (DRIs) for healthy individuals [19], as we do not yet have specific reference values for IBD. This is concerning as patients with CD often have increased nutritional requirements due to increased intestinal losses, reduced intake, and malabsorption [21], suggesting that nutrient adequacy data should be interpreted cautiously. Further research is needed to define the appropriate intake levels of micronutrients like folate and vitamin B12 in patients with IBD. While the latest guidelines include specific protein and energy intake recommendations, there are no established values for other nutrients, leaving a gap in the nutritional recommendations for this population [21, 36].
Our results highlight an important finding: the association between vitamin B12 and lower ESR, a marker of inflammation, as well as with the absence of EIMs. This suggests that adequate B12 may have a protective role against inflammation [37]. Since ESR commonly rises in inflammatory conditions, the association observed could reflect the role of vitamin B12 in reducing Hcy [38, 39]. Additionally, the positive association between the absence of EIMs and the highest tertile (T3) of vitamin B12 intake also indicates the potential of vitamin B12 in attenuating Hcy, which is associated with autoimmune cascades [40]. Studies have shown that elevated Hcy levels in IBD result from pharmacological, dietary, and lifestyle factors, associated with low levels of vitamin B12 and folate [39, 41]. Moreover, research indicates that hyperhomocysteinemia exacerbates symptoms and inflammation in patients with rheumatoid arthritis, a frequent EIM in patients with IBD [42, 43].
Similarly, our study showed that folate was associated with the consumption of nutrient-rich foods, such as meats and fruits, and with iron and riboflavin consumption, indicating that healthy dietary patterns can contribute to reducing the risk of deficiency. Furthermore, total folate showed a negative association with the absence of EIMs. This is noteworthy, as folate plays a central role in Hcy metabolism [41], yet it did not show clinical associations comparable to those observed for vitamin B12. Studies show that folate supplementation can reduce Hcy by 25%, compared with 7% for vitamin B12. A meta-analysis by Chen et al. associated folate intake, but not vitamin B12, with lower heart attack risk, highlighting the role of Hcy in cardiovascular pathology [44].
In our dietary intake analysis, we were unable to identify correlations consistent with previous literature. A possible explanation is that folate may have a greater capacity to reduce Hcy concentrations and be associated with clinical outcomes when supplemented. Dietary patterns and individual variability in nutrient absorption may also have contributed to the absence of associations. A review on Hcy metabolism reiterates the impact of folate supplementation (0.5–5 mg/day); however, it provides limited evidence regarding food-derived folate [45], which may explain why folate intake from diet did not show clinical effects in our study, unlike vitamin B12.
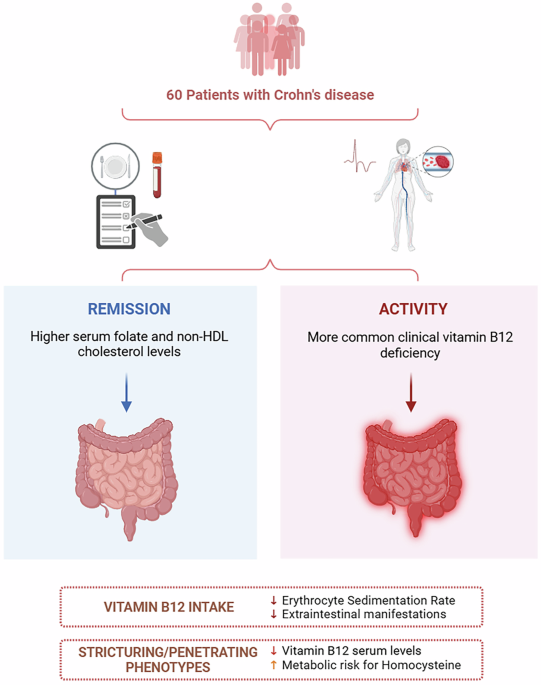
Our analysis of biochemical parameters demonstrated that patients in remission showed higher serum folate and non-HDL-C levels, whereas patients with active disease had higher rates of B12 deficiency. The active phase, especially in patients who have already undergone ileal resection, may predispose them to increase the risk of B12 malabsorption [10], which could explain a greater proportion of active patients in the category. Evidence also associates low B12 with higher levels of inflammatory markers, such as IL-6 and C-reactive protein [37, 46, 47] supporting the hypothesis that deficiency may contribute to systemic inflammation and potentially worsen disease activity.
Regarding folate, previous studies indicate that CD patients tend to have lower serum levels [48], influenced by inadequate intake, malabsorption, increased requirements, inflammation, and medication use [10, 49]. These factors may be intensified during active disease, when appetite and intake often decline [41].
Furthermore, the remission group had a significantly higher mean of non-HDL-C than the active group. This may reflect a nutritional transition in patients in remission, where overweight and obesity become more prevalent. Our group previously reported that 55% of patients in remission were overweight or obese [14]. Obesity is associated with altered lipid profiles and long-term cardiovascular risk [50], and shares mechanisms with IBD, such as chronic inflammation, mesenteric fat infiltration, and adipokine dysregulation, that may influence lipid metabolism [51]. Conversely, increased cardiovascular risk and lipid alterations have also been described in active disease, driven by inflammation and treatment-related effects [31]. Overall, our findings emphasize that active disease and advanced phenotypes (B2/B3) are associated with lower B12 levels, higher metabolic risk, and greater nutritional vulnerability, whereas patients in remission show higher folate and non-HDL-C levels, potentially influenced by nutritional transition and weight gain. Given the high prevalence of overweight and obesity among patients in remission, we also explored whether BMI was associated with the metabolic risk categories for Hcy used in this study; however, no significant associations were identified. These findings suggest that Hcy-related risk in this cohort is more closely associated with nutritional deficiencies and disease-related factors than with BMI.
The lower B12 values observed in the B2/B3 phenotype align with prior evidence showing that ileal inflammation, pre-stenotic dilatation, and resections >20 cm markedly impair B12 absorption [52, 53]. As the ileum is the primary absorption site, structural and inflammatory alterations in this region likely drive both the nutrient deficiency and the higher metabolic risk observed in these patients.
Most patients in our sample were in the metabolic deficiency risk range for Hcy (140–300 pmol/L), although no differences were observed between the groups. Increased Hcy may promote Th17 differentiation and enhance pro-inflammatory cytokine production, contributing to complications such as strictures and fistulas [54, 55]. Therefore, there is an association between nutritional insufficiency and cardiovascular risk, regardless of the stage of the disease or deficiency status.
Thus, the risk category of patients in the B2 + B3 group with lower serum levels of vitamin B12 may have a bidirectional causality. There may be some pathophysiological relationship between higher Hcy concentrations and a worse prognosis of the disease. In addition, complications characteristic of the stricturing or penetrating disease could increase the risk of B12 depletion due to involvement of the ileum, intestinal inflammatory activity, the need for intestinal resection, and loss of tissue function [10].
Although these findings are predictive rather than conclusive, they emphasize the importance of nutritional assessment as part of the clinical routine for these patients since elevated Hcy and micronutrient deficiencies may affect the course of the disease and overall health. Dietary strategies to improve micronutrient intake could play a crucial role in managing CD by addressing malabsorption deficiencies, reducing inflammation, and supporting metabolic health. Therefore, the study suggests that folate and B12 levels could serve as markers for clinical characteristics and be associated with metabolic risk in CD patients.
Despite these findings, some limitations of our study should be considered. Dietary assessment based on a single 24-hour recall may not fully capture habitual intake [56], although single-day recalls have been used effectively in similar research [57, 58]. Additionally, the absence of serum Hcyone, one of our primary variables of interest, required the use of validated metabolic risk categories. While informative, these categories represent indirect estimates and cannot establish causal relationships. Future studies incorporating direct biomarkers such as methylmalonic acid and Hcy would deepen understanding of micronutrient metabolism and its role in CD progression.