Soil characteristics
The physical and chemical properties of two contaminated soils are showed in Table 1, including pH, OM, and total concentrations of major elements. Both soil A and soil B had similar pH, 7.48 and 8.02, respectively. The OM content of soil A and B was 11.1% and 4.99%, respectively. Arsenic concentration was nearly 10-fold higher in soil A compared to soil B (1011 ± 21.6 mg kg–1 vs. 121 ± 9.58 mg kg–1), and Pb concentration was also higher in soil A than soil B (4738 ± 65.4 mg kg–1 vs. 1427 ± 21.8 mg kg–1). The other elements (Ca, Fe, and Mn) concentrations were also 2.64-, 6.96-, and 27.8-fold higher in soil A compared to soil B. In addition to the difference in total metal contents, there are also variations in the concentrations of different As and Pb fractions in soils (FigureS1). Specially, the amorphous Fe/Al oxide fraction (AF3, 22.0%), crystallized Fe/Al oxide fraction (CF4, 32.9%), and residual fraction (RS5, 35.6%) were the major As fractions for soil A, while AF3 (62.9%) was the main fraction of soil B. Different from the distribution of As fraction, Fe/Mn oxides fraction (F3, 80.2%) was the dominant form of Pb for soil A, and Pb in the carbonate fraction (C2, 54.8%) and F3 (29.8%) accounted for 84.6% of soil B, which was similar to the previous study17.
Table 1 Physical and chemical properties of soils (< 250 μm particle size).As and Pb bioaccessibility in soils
In this study, As and Pb bioaccessibility measured by SBRC assay varied considerably with soil types (Fig. 1). In the gastric phase (GP), the bioaccessibility of As (1.28 ± 0.04% and 73.7 ± 3.72%) and Pb (19.0 ± 0.74% and 92.3 ± 5.47%) in soil A and B showed significant differences (p < 0.05), respectively. In the intestinal phase (IP), Pb bioaccessibility in soil A and B decreased from 19.0 to 0.18%, 92.3 to 17.5%, respectively. From the gastric (pH ~ 1.5) to intestinal phase (pH ~ 7.0), the soluble Pb in the gastric phase was probably absorbed by soils and Fe oxides17.
Based on our data, As was less bioaccessible (mean 37.5%) than Pb (55.7%) in both soil A and B impacted by mining/smelting activities. This result was consisted with previous studies that As bioaccessibility lower than Pb in soils contaminated by mining/smelting activities18,26. Compared to Pb (0.42‒2.05%), higher portions of As were associated with RS5 (5.66‒35.6%), thereby leading to lower As bioaccessibility. Furthermore, non-specifically sorbed (NS1) + specifically sorbed (SS2) is usually considered to be a mobile component of As with high bioaccessibility27. Comparing the percentage of different bound As in soil A and B, it is found that NS1 + SS2 in soil A accounted for 9.51%, and NS1 + SS2 in soil B accounted for 19.28%. Previous work suggested that available Pb mainly existed in exchangeable state (E1) and carbonate bound state (C2)17. In this study, E1 + C2 bound Pb of soil A accounted for 7.60% and up to 58.9% in soil B. Therefore, the As and Pb bioaccessibility in soil A contaminated by mining activity was significantly lower than that in soil B contaminated by smelting activity.
Effect of inorganic calcium on as and Pb bioaccessibility
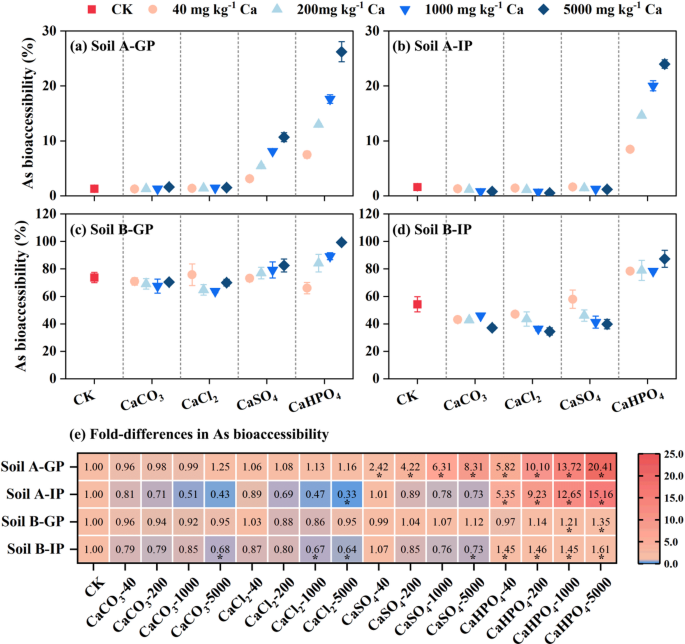
The effect of four inorganic Ca on As bioaccessibility in soils was shown in Fig. 1. In the gastric phase (GP), As bioaccessibility of soil A and B was increased from 1.28 to 73.7% to 1.23–99.3% after adding CaHPO4 and CaSO4, respectively. For these two kinds of inorganic calcium, the growth rates of As bioaccessibility in the two soil samples were higher under high-dose treatment (8.79‒25.6% with 5000 mg Ca kg‒1) compared with those under low-dose treatment (−2.17‒6.20% with 40 mg Ca kg‒1). Phosphate and arsenate have similar structure and chemical behavior, and both of them can be adsorbed on iron oxides28. In the gastric phase, the addition of CaHPO4 promoted the dissolution of iron in two soils (Figure S2), thereby reducing the adsorption sites on the surface of iron oxides. In this state, phosphate competes with arsenate, and this competition enhances with the increase of phosphate concentration24,29. Therefore, the addition of CaHPO4 increased the As bioaccessibility and showed a dose effect. Although previous studies showed that sulfate had an insignificant influence the arsenate adsorption under acidic conditions30, the addition of CaSO4 also promoted the dissolution of iron in gastric phase (Figure S2). Therefore, AF3 bound As in soils was partially released, thereby increasing As bioaccessibility.
Fig. 1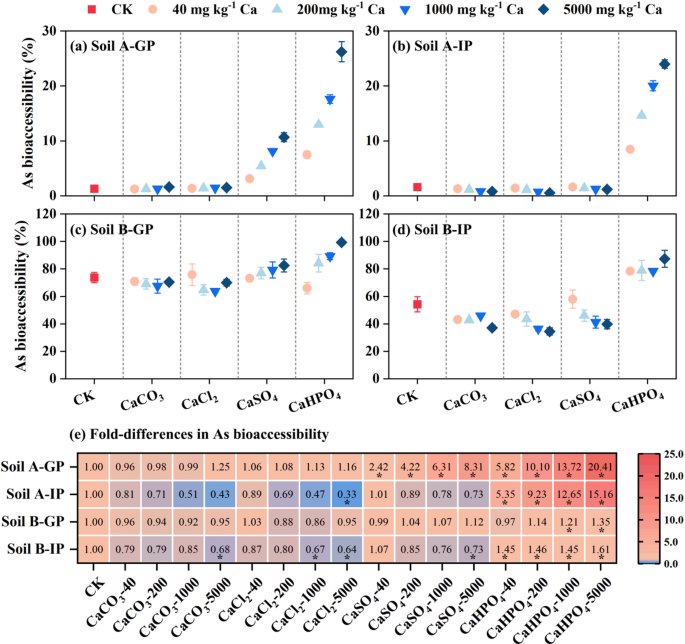
Effect of inorganic calcium on the As bioaccessibility in gastric phase (GP) and intestinal phase (IP) (a-d) and heat map (e) showed that compared with the control group. * p < 0.05.
Small reductions in the As bioaccessibility in soil B were observed with the addition of calcium carbonate (CaCO3) and calcium chloride (CaCl2) groups. Although chloride (Cl−) and carbonate/bicarbonate (CO32−/HCO3−) were found to be limited or insignificant effect on As adsorption29, this result was consistent with the fact that the addition of CaCO3 and CaCl2 slightly improved the dissolution of iron (Figure S2). Similar result was reported that Fe and Ca concentrations in rice being negatively correlated with As bioaccessibility in the gastric phase by31. Comparison with the control, our results indicated that the presence of CaCO3, CaCl2, and CaSO4 lead to a detectable reduction in As bioaccessibility of the two soil samples after gastrointestinal digestion. Under the neutral condition of the intestinal phase, Ca combined with As to form a precipitate, thereby fixing the dissolved As in the solution and reducing the As bioaccessibility32. However, the addition of CaHPO4 still improved As bioaccessibility for all soil samples in intestinal phase (IP). This phenomenon was consistent with the conclusion that there was still competition between phosphate and As in neutral environment33.
Different from As, Pb bioaccessibility in GP was decreased from 19.0 to 92.3% to 13.2‒88.1% with the addition of four inorganic Ca for two soil samples (Fig. 2), and varied with the inorganic Ca type. Due to the ion competition between Ca2+ and Pb2+, the dissolution of Pb in the GP was inhibited34. The higher the dosage of added Ca, the more significant inhibition of Pb dissolution in the GP, thereby reducing Pb bioaccessibility. Similarly, the addition of Ca compounds (CaCO3, CaCl2, and CaSO4) inhibited the precipitation of soluble Pb and Fe oxides in the IP, thus improving Pb bioaccessibility compared with control without additional Ca. However, CaHPO4 reduced Pb bioaccessibility from 0.18 to 17.53 to 0.00‒3.53% in IP. The XRD results showed that new Pb phosphate minerals (such as Pb5(PO4)3Cl and Pb2P2O7) were formed in soils after extracting by simulated gastrointestinal fluid, which reduced the Pb bioaccessibility (Figure S3). Previous studies have reported that the order of solubility of Pb compounds from high to low is: PbCO3 > PbSO4 > PbHPO4 > Pb5(PO4)3OH > Pb5(PO4)3Cl35. Compared with Pb-phosphate minerals, PbCO3 and PbSO4 were difficult to form minerals due to high solubility in gastrointestinal simulated solution, which was also the reason why CaCO3 and CaSO4 did not significantly reduce the bioaccessibility of Pb in the IP.
Fig. 2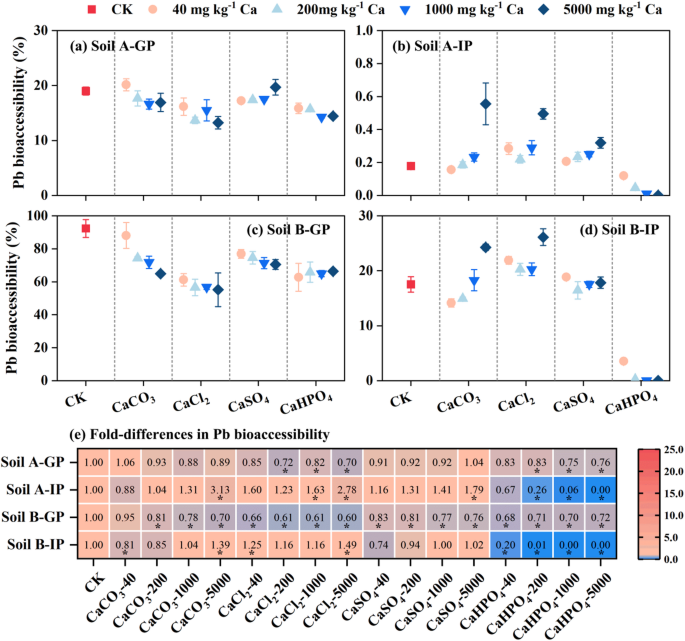
Effect of inorganic calcium on the Pb bioaccessibility in gastric phase (GP) and intestinal phase (IP) (a-d) and heat map (e) showed that compared with the control group. * p < 0.05.
Effect of organic calcium on the as and Pb bioaccessibility
The effect of four organic Ca (Ca lactate, Ca gluconate, Ca citrate and Ca acetate) on As bioaccessibility in soils was shown in Fig. 3. In the gastric phase, As bioaccessibility increased 1.27‒4.13, 1.32‒9.14 and 2.39‒8.79 times after adding different doses (40‒5000 mg Ca kg‒1) Ca lactate, Ca gluconate and Ca citrate to soil (A) The difference analysis showed that Ca citrate had significant difference in improving As bioaccessibility, while Ca lactate and Ca gluconate had a significant difference only at high doses (Fig. 3E). To explain this phenomenon, we measured the concentration of Fe in gastric phase, and the results showed that the concentration of extractable Fe and As had similar increasing trend by addition the three organic acid (Ca lactate, Ca gluconate and Ca citrate) (Figure S2). It was reported that low-molecular-mass organic acids (such as citric acid) led to the release of As from mineral oxides, thereby increasing As bioaccessibility in a dose-dependent manner36. Therefore, we speculated that the organic acid in three different types of Ca salts promoted the desorption of As from Fe minerals, thereby increasing As bioaccessibility. However, the addition of different doses of Ca acetate did not change the concentration of Fe in the gastric phase, which was consistent with the As bioaccessibility. In addition, the As bioaccessibility was almost unchanged with adding the four organic calcium for soil (B) From gastric phase to intestinal phase, the As bioaccessibility decreased gradually from 53.0% to 42.0% with the increase of Ca acetate addition (from 40 to 5000 mg Ca kg‒1). The above data indicated that Ca acetate had the potential to reduce the dissolution of As from the soil in gastrointestinal tract.
Fig. 3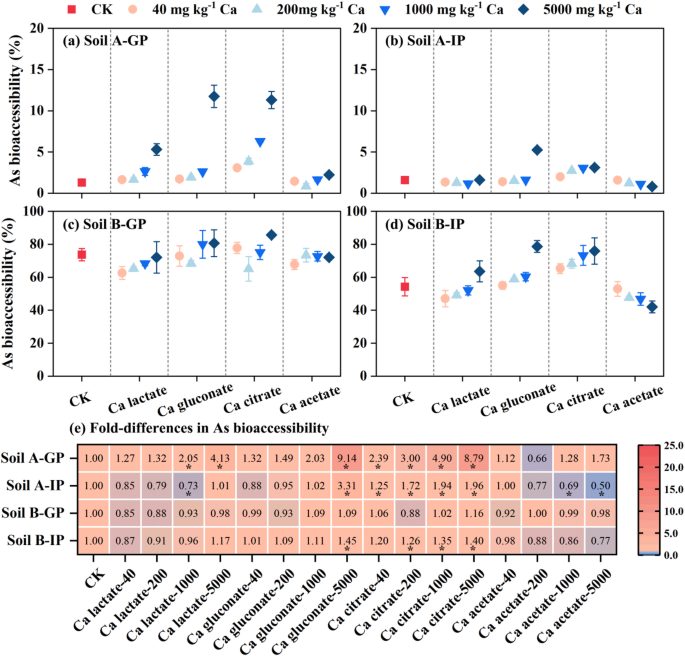
Effect of organic calcium on the As bioaccessibility in gastric phase (GP) and intestinal phase (IP) (a-d) and heat map (e) showed that compared with the control group. * p < 0.05.
Different from As bioaccessibility, the addition of four organic Ca resulted in a significant decrease of Pb bioaccessibility (from 19.0% to 9.55% and from 92.3% to 56.2% for soil A and B, respectively) in the GP due to the competition between Pb and Ca (Fig. 4). In addition, low concentrations of organic acids can inhibit the desorption of Pb, but it can alleviate the inhibition and even enhance the desorption of Pb at higher concentrations37. Therefore, the Pb bioaccessibility showed a trend of decreasing first and then increasing, especially in soil A. During neutral intestinal phase, soluble Fe in the gastric phase precipitates as Fe oxides, which then adsorbs soluble Pb and thereby reduce its solubility17. The present study revealed that the decreased bioaccessibility of Pb in the intestinal phase was closely associated with a sharp reduction in soluble Fe content (Figure S2). However, with the increase of organic calcium concentration, the concentration of dissolved Fe in the intestinal simulated solution increased significantly, which may be the key factor leading to the increase of Pb bioaccessibility in IP. Among the four organic Ca, the addition of Ca acetate had the best effect on reducing the Pb bioaccessibility in the two soils.
Fig. 4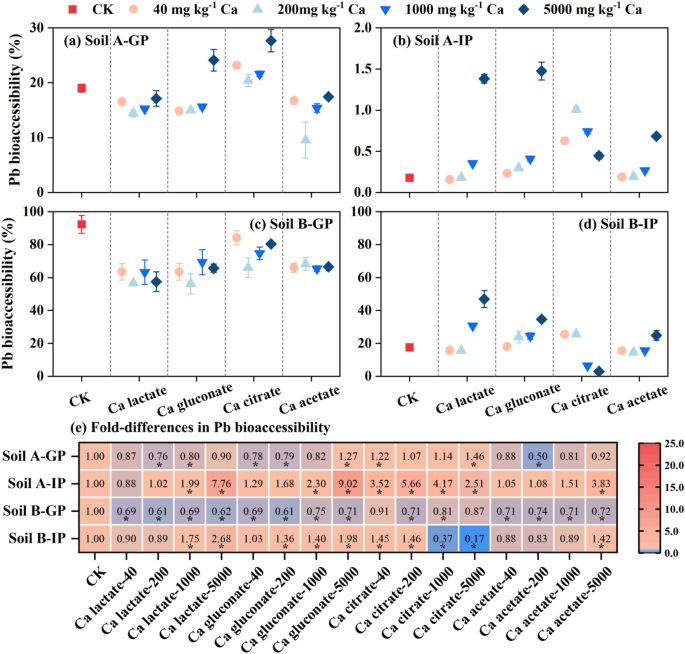
Effect of organic calcium on the Pb bioaccessibility in gastric phase (GP) and intestinal phase (IP) (a-d) and heat map (e) showed that compared with the control group. * p < 0.05.
Effect of CaHPO4 and Ca acetate on As- and Pb-RBA
The effects of different calcium compounds on the As and Pb bioaccessibility showed that Ca acetate and CaHPO4 significantly reduced the As and Pb bioaccessibility, respectively. Therefore, Ca acetate and CaHPO4 were selected to study the regulation of As and Pb relative bioavailability (RBA) via mouse model. The accumulation of As/Pb in mouse tissues exposed to sodium arsenate/Pb acetate showed a strong correlation with the administered dose (R2 = 0.95 − 0.96), indicating that a good linear response can be obtained by using the liver and kidneys as the exposure endpoint (Figure S4).
Fig. 5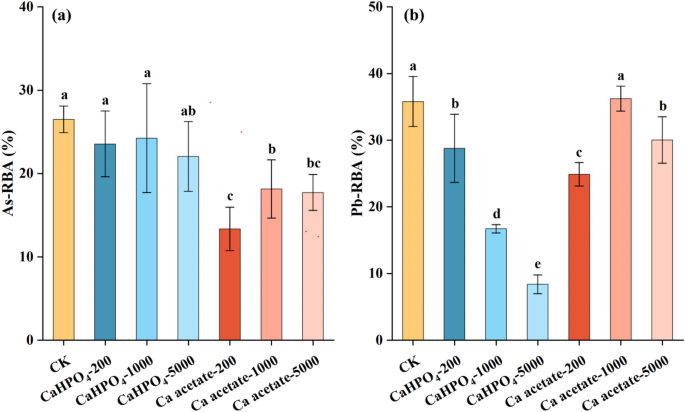
Effects of Ca compounds (CaHPO4 and Ca acetate) on the relative bioavailability of As (a) and Pb (b) contained in orally ingested soil B in mice.
The effects of Ca acetate and CaHPO4 on As- or Pb-RBA were determined on the basis of As or Pb accumulation in liver and kidneys of the mice fed with soil B for 10 days (Fig. 5). In mice exposed to soil B without Ca supplementation, As and Pb relative bioavailability was 26.5 ± 2.51% and 35.8 ± 5.03%, respectively. Compared to soil B without Ca supplements, mice fed CaHPO4 had 8.50%−16.8% and 19.6%−76.6% lower As-RBA and Pb-RBA, respectively. With the addition of CaHPO4, Ca and As can form low-soluble calcium arsenate in the intestinal neutral environment, thereby reducing the dissolution of As38,39. This idea was supported by our current findings, which showed 9.56%−18.4% lower As bioaccessibility of soil B with the presence of calcium compounds (5000 mg Ca L–1 from CaCO3, CaCl2, and CaSO4) in intestinal fluid. However, the competition between phosphate and arsenate leads to the re-release and activation of As40, so the addition of CaHPO4 did not significantly reduce the As-RBA. Different from As, the higher CaHPO4 concentration corresponded to lower Pb-RBA. Recent studies have demonstrated that Pb-RBA is reduced in the presence of phosphate, owing to the reaction between soluble Pb and phosphate in the gastrointestinal tract that generates insoluble lead phosphate precipitates41,42. This conclusion was consisted with our finding, which showed 99% lower Pb bioaccessibility of intestinal phase with CaHPO4 (5000 mg Ca kg‒1) (Fig. 2d). In addition, the competition between Ca2+ and Pb for calcium transporters may be another important reason for reducing Pb-RBA8.
Compared to the control, mice fed Ca acetate had 31.5%−49.6% and 0%−30.5% lower As- and Pb-RBA, respectively. In comparison with mice fed CaHPO4, those fed with Ca acetate exhibited higher Pb-RBA. The difference was possibly due to the ability of organic ligands to enhance Pb solubility in the intestine (Fig. 4d). Calcium acetate was more effective than CaHPO4 in reducing As-RBA. Especially, 200 mg kg‒1 Ca acetate was the most effective at reducing As-RBA in soil B (49.6% reductions in As RBA). In this study, Ca acetate was associated with lower As solubility of intestinal phase in contrast with Ca lactate, Ca gluconate, or Ca citrate (Fig. 3). Arsenic dissolved in the gastrointestinal tract is absorbed through phosphate transporters, such as the type IIb sodium phosphate (NaPi-IIb) cotransporters located on the apical membrane of enterocytes43.Lower mRNA expression of the P transporter NaPi-IIb was observed in the duodenum of mice fed organic Ca (Ca gluconate, Ca lactate, Ca aspartate, and Ca citrate)8. Therefore, the two processes (Ca-decreased As solubility and Ca-decreased P transporters) interacted synergistically, leading to decrease As-RBA with Ca acetate.