STONY BROOK, NY, April 2, 2026 – Systemic candidiasis is an opportunistic fungal infection that has been difficult to treat effectively. Research published in a paper in the April edition of Cell Host & Microbe suggests that immune metabolic reprogramming could be a new strategy to fight the infection rather than developing another specific antifungal medication.
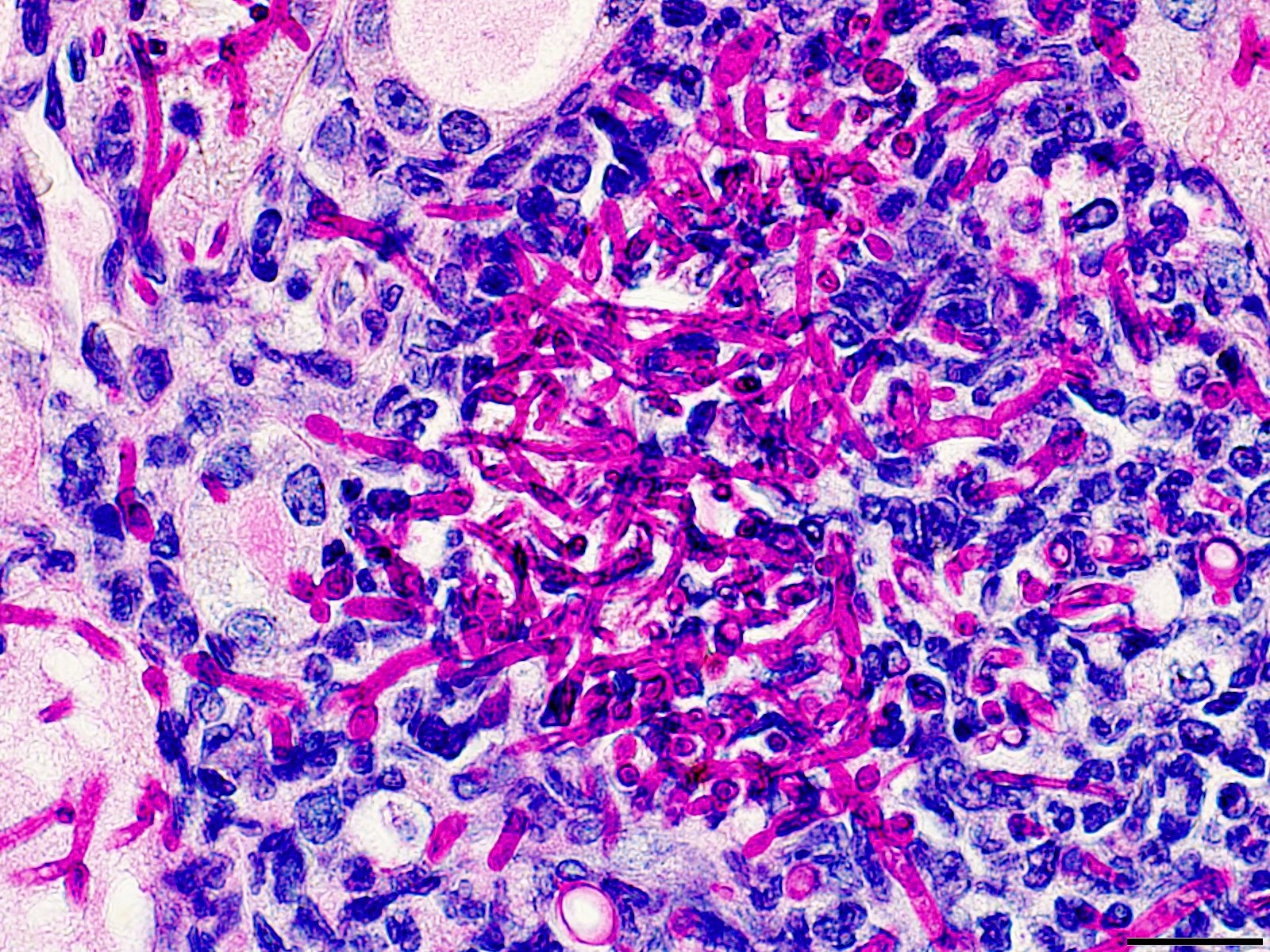
The fungus Candida albicans causes infections that range from superficial on the skin and nails to invasive into organs and the bloodstream. In recent decades, systemic candidiasis has increased due to more patients with immunosuppression from disease or treatments, prolonged antibiotic exposure and certain conditions such as kidney disease. Management of systemic candidiasis has become more difficult because of antifungal drug resistance, limited early diagnostic tools and absence of approved fungal vaccines.
According to Partha Biswas, DVM, PhD, lead author of the paper, and a Professor in the Department of Microbiology and Immunology in the Renaissance School of Medicine (RSOM) at Stony Brook University, these challenges have become roadblocks to treating systemic candidiasis and illustrate the need for new and different therapeutic strategies.
Dr. Biswas and his coinvestigators used a murine model of candidiasis to look at neutrophils, which are the primary effector cells responsible for rapid fungal clearance during candidiasis. However, this natural antifungal activity depends largely on extracellular glucose, which becomes scarce in this infection process because C. albicans competes for the nutrient. What usually occurs is that metabolic competition within the host is a hostile environment that limits neutrophil survival and function, especially when underlying metabolic impairments (disease) are present.
The research team demonstrated in the model that neutrophils overcome glucose deprivation by activating the glycogen phosphorylase liver form (PYGL) enzyme. This enzyme mobilizes intracellular glycogen stores that fuel antifungal activity.
They further found that by a neutrophil-specific deletion of PYGL in the model, the mice had an increased susceptibility to candidiasis. But then by using a clinically approved PYGL activator, beta 2 adrenergic receptor agonist, the host defense against candidiasis greatly improved by way of increased antifungal functions. Additionally, by using neutrophils from the peripheral blood of healthy volunteers, the authors demonstrated that human neutrophils similarly upregulate PYGL function to control fungal infections.
Beta 2 adrenergic receptor agonists, such as Albuterol, are currently used to treat asthma and other respiratory conditions, as they relax muscles in airways to improve breathing.
“Our results uncover a previously unrecognizable metabolic pathway that has the potential to be therapeutically targeted against candidiasis,” explains Dr. Biswas. “If we are able to identify and use the PYGL enzyme to essentially reprogram neutrophil function in people against C. albicans that cause infection, this could significantly change the course of future treatments against candidiasis and perhaps other invasive fungal infections.”
This collaborative work involved medical researchers from both Stony Brook Medicine and the University of Pittsburgh. The research is supported in part by numerous grants from federal agencies, including three National Institutes of Health (NIH) grants to Dr. Biswas, and another NIH grant and a Department of Defense (DOD) grant to a co-author, Charles K. Vorkas, MD, of Stony Brook Medicine.
###