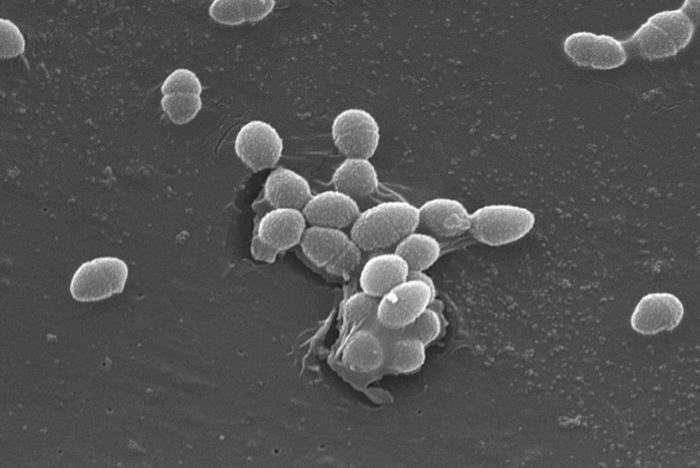
Chronic wound infections are notoriously difficult to manage because some bacteria can actively interfere with the body’s immune defenses. In wounds, Enterococcus faecalis (E. faecalis) is particularly resilient – it can survive inside tissues, alter the wound environment, and weaken immune signals at the injury site. This disruption creates conditions where other microbes can easily establish themselves, resulting in multi-species infections that are complex and slow to resolve. Such persistent wounds, including diabetic foot ulcers and post-surgical infections, place a heavy burden on patients and health care systems, and sometimes lead to serious complications such as amputations.
Now, researchers have discovered how E. faecalis releases lactic acid to acidify its surroundings and suppresses the immune-cell signal needed to start a proper response to infection. By silencing the body’s defenses, the bacterium can cause persistent and hard-to-treat wound infections. This explains why some wounds struggle to heal, even with treatment, and why infections involving multiple bacteria are especially difficult to eradicate.
The work was led by researchers from the Singapore-MIT Alliance for Research and Technology (SMART) Antimicrobial Resistance ( AMR ) interdisciplinary research group, alongside collaborators from the Singapore Centre for Environmental Life Sciences Engineering at Nanyang Technological University (NTU Singapore), MIT, and the University of Geneva in Switzerland.
Researchers found that E. faecalis uses a two‑step mechanism to achieve this. Lactic acid enters the macrophages through a lactate transporter called MCT‑1 and also binds to a lactate-sensing receptor, GPR81, on the cell surface. By engaging both pathways, the bacterium effectively shuts down downstream immune signalling and blocks the macrophage’s inflammatory response, allowing E. faecalis to persist in the wound much longer than it should. Specifically, the lactic acid prevents a key immune alarm signal, known as NF-κB, from switching on inside these cells.
This was proven in a mouse wound model, where strains of E. faecalis that could not make lactic acid were cleared much more quickly, and the wounds also showed stronger immune activity. In wounds infected with both E. faecalis and Escherichia coli, the weakened immune response caused by lactic acid also allowed E. coli to grow better. This explains why wound infections often involve multiple species of bacteria and become harder to treat over time, particularly since E. faecalis is among the most common bacteria found in chronic wounds.
“Chronic wound infections often fail not because antibiotics are powerless, but because the immune system has effectively been ‘switched off’ at the infection site. We found that E. faecalis floods the wound with lactic acid, lowering pH and muting the NF‑κB alarm inside macrophages – the very cells that should be calling for help. By pinpointing how acidity rewires immune signalling, we now have clear targets to reactivate the immune response,” says first author Ronni da Silva, research scientist at SMART AMR, former postdoc in the lab of co-author and MIT professor of biology Jianzhu Chen, and SCELSE-NTU visiting researcher.
“This discovery strengthens our understanding of host-pathogen interactions and offers new directions for developing treatments and wound care that target the bacteria’s immunosuppressive strategies. By revealing how the immune response is shut down, this research may help improve infection management and support better recovery outcomes for patients, especially those with chronic wounds or weakened immunity,” says Kimberly Kline, principal investigator at SMART AMR, SCELSE-NTU visiting academic, professor at the University of Geneva, and corresponding author of the paper.
By identifying lactic‑acid‑driven immune suppression as a root cause of persistent wound infections, this work highlights the potential of treatment approaches that support the immune system, rather than rely on antibiotics alone. This could lead to therapies that help wounds heal more reliably and reduce the risk of complications. Potential directions include reducing acidity in the wound or blocking the signals that lactic acid uses to switch off immune cells.
Building on their study, the researchers plan to explore validation in additional pathogens and human wound samples, followed by assessments in advanced preclinical models ahead of any potential clinical trials.
The research was partially supported by the National Research Foundation Singapore under its Campus for Research Excellence and Technological Enterprise program.
/University Release. This material from the originating organization/author(s) might be of the point-in-time nature, and edited for clarity, style and length. Mirage.News does not take institutional positions or sides, and all views, positions, and conclusions expressed herein are solely those of the author(s).View in full here.