Metchnikov, E. Untersuchungen ueber die mesodermalen Phagocyten einiger Wirbeltiere. Biologisches Centralblatt 3, 560–565 (1883).
Browicz, T. Ueber intravasculäre Zellen in den Blutcapillaren der Leberacini. Arch. f. mikrosk. Anat. 55, 420–426 (1899).
Sierro, F. et al. A liver capsular network of monocyte-derived macrophages restricts hepatic dissemination of intraperitoneal bacteria by neutrophil recruitment. Immunity 47, 374–388.e6 (2017).
Guilliams, M. et al. Spatial proteogenomics reveals distinct and evolutionarily conserved hepatic macrophage niches. Cell 185, 379–396.e38 (2022). This work integrated spatial transcriptomics and proteomics to map hepatic macrophage niches, showing that different subsets occupy evolutionarily conserved microenvironments that determine their identity and function.
Remmerie, A. et al. Osteopontin expression identifies a subset of recruited macrophages distinct from Kupffer cells in the fatty liver. Immunity 53, 641–657.e14 (2020).
Tran, S. et al. Impaired Kupffer cell self-renewal alters the liver response to lipid overload during non-alcoholic steatohepatitis. Immunity 53, 627–640.e5 (2020).
Wang, J. & Kubes, P. A reservoir of mature cavity macrophages that can rapidly invade visceral organs to affect tissue repair. Cell 165, 668–678 (2016).
Blériot, C. & Ginhoux, F. Understanding the heterogeneity of resident liver macrophages. Front. Immunol. 10, 2694 (2019).
Hoeffel, G. et al. C-Myb+ erythro-myeloid progenitor-derived fetal monocytes give rise to adult tissue-resident macrophages. Immunity 42, 665–678 (2015).
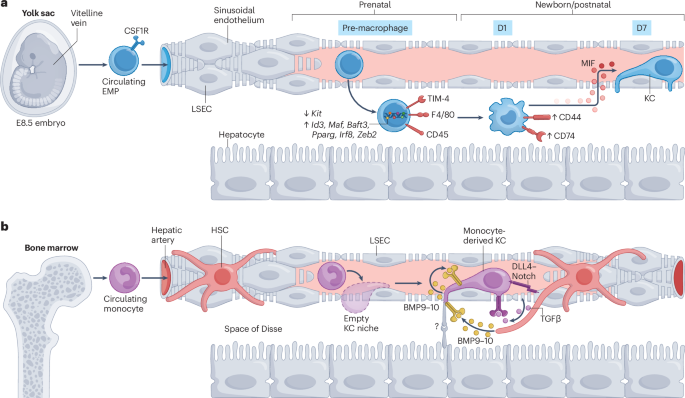
Gomez Perdiguero, E. et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 518, 547–551 (2015). This study overturned the dogma that tissue macrophages come from bone marrow, proving that yolk sac-derived erythro-myeloid progenitors give rise to a first wave of tissue macrophages during embryogenesis.
Schulz, C. et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science 336, 86–90 (2012).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013).
Mass, E. et al. Specification of tissue-resident macrophages during organogenesis. Science 353, aaf4238 (2016).
Liu, Z. et al. Fate mapping via Ms4a3-expression history traces monocyte-derived. Cell 178, 1509–1525.e19 (2019).
Scott, C. L. et al. Bone marrow-derived monocytes give rise to self-renewing and fully differentiated Kupffer cells. Nat. Commun. 7, 10321 (2016). This work provided functional evidence loss of embryonic KCs after liver injury opens the niche to circulating monocytes, which can engraft, self-renew and acquire the full identity of canonical KCs.
Sakai, M. et al. Liver-derived signals sequentially reprogram myeloid enhancers to initiate and maintain Kupffer cell identity. Immunity 51, 655–670.e8 (2019).
Bonnardel, J. et al. Stellate cells, hepatocytes, and endothelial cells imprint the Kupffer cell identity on monocytes colonizing the liver macrophage niche. Immunity 51, 638–654.e9 (2019). This work showed that KCs do not acquire their identity on their own but are instructed by signals from LSECs, hepatocytes and HSCs, highlighting the central role of this niche in shaping immune cell fate.
David, B. A. et al. Combination of mass cytometry and imaging analysis reveals origin, location, and functional repopulation of liver myeloid cells in mice. Gastroenterology 151, 1176–1191 (2016).
Krenkel, O. & Tacke, F. Liver macrophages in tissue homeostasis and disease. Nat. Rev. Immunol. 17, 306–321 (2017).
Woltman, A. M., Boonstra, A., Naito, M. & Leenen, P. J. M. in Macrophages: Biology and Role in the Pathology of Diseases (eds Biswas, S. K. & Mantovani, A.) 217–247 (Springer, 2014).
Wong, C. H. Y., Jenne, C. N., Petri, B., Chrobok, N. L. & Kubes, P. Nucleation of platelets with blood-borne pathogens on Kupffer cells precedes other innate immunity and contributes to bacterial clearance. Nat. Immunol. 14, 785–792 (2013).
Blériot, C. et al. Liver-resident macrophage necroptosis orchestrates type 1 microbicidal inflammation and type-2-mediated tissue repair during bacterial infection. Immunity 42, 145–158 (2015).
Zeng, Z. et al. Sex-hormone-driven innate antibodies protect females and infants against EPEC infection. Nat. Immunol. 19, 1100–1111 (2018).
Alemany, R., Suzuki, K. & Curiel, D. T. Blood clearance rates of adenovirus type 5 in mice. J. Gen. Virol. 81, 2605–2609 (2000).
Deppermann, C. et al. Tacrolimus impairs Kupffer cell capacity to control bacteremia: why transplant recipients are susceptible to infection. Hepatology 73, 1967–1984 (2021).
Sitia, G. et al. Kupffer cells hasten resolution of liver immunopathology in mouse models of viral hepatitis. PLoS Pathog. 7, e1002061 (2011).
van Furth, R. et al. The mononuclear phagocyte system: a new classification of macrophages, monocytes, and their precursor cells. Bull. World Health Organ. 46, 845–852 (1972).
Deimann, W. & Fahimi, H. D. Peroxidase cytochemistry and ultrastructure of resident macrophages in fetal rat liver. Dev. Biol. 66, 43–56 (1978).
Sorokin, S. P., Hoyt, R. F. & Grant, M. M. Development of macrophages in the lungs of fetal rabbits, rats, and hamsters. Anat. Rec. 208, 103–121 (1984).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Heinz, S., Romanoski, C. E., Benner, C. & Glass, C. K. The selection and function of cell type-specific enhancers. Nat. Rev. Mol. Cell Biol. 16, 144–154 (2015).
Bian, Z. et al. Deciphering human macrophage development at single-cell resolution. Nature 582, 571–576 (2020).
Holt, P. G. & Jones, C. A. The development of the immune system during pregnancy and early life. Allergy 55, 688–697 (2000).
Popescu, D.-M. et al. Decoding human fetal liver haematopoiesis. Nature 574, 365–371 (2019).
Surewaard, B. G. J. & Kubes, P. Measurement of bacterial capture and phagosome maturation of Kupffer cells by intravital microscopy. Methods 128, 12–19 (2017).
McDonald, B. et al. Programing of an intravascular immune firewall by the gut microbiota protects against pathogen dissemination during infection. Cell Host Microbe 28, 660–668.e4 (2020).
Hirako, I. C. et al. Uptake of Plasmodium chabaudi hemozoin drives Kupffer cell death and fuels superinfections. Sci. Rep. 12, 19805 (2022).
Araujo David, B. et al. Kupffer cell reverse migration into the liver sinusoids mitigates neonatal sepsis and meningitis. Sci. Immunol. 9, eadq9704 (2024). This study challenged the dogma that KCs are sessile by showing that in neonates they can detach from the sinusoidal wall and migrate back into the lumen, where they intercept bacteria and protect against systemic infections.
Ginhoux, F. & Guilliams, M. Tissue-resident macrophage ontogeny and homeostasis. Immunity 44, 439–449 (2016).
Bittmann, I. et al. The role of graft-resident Kupffer cells and lymphocytes of donor type during the time course after liver transplantation—a clinico-pathological study. Virchows Arch. 443, 541–548 (2003).
Pallett, L. J. et al. Longevity and replenishment of human liver-resident memory T cells and mononuclear phagocytes. J. Exp. Med. 217, e20200050 (2020).
MacParland, S. A. et al. Single cell RNA sequencing of human liver reveals distinct intrahepatic macrophage populations. Nat. Commun. 9, 4383 (2018).
Andrews, T. S. et al. Single-cell, single-nucleus, and spatial RNA sequencing of the human liver identifies cholangiocyte and mesenchymal heterogeneity. Hepatol. Commun. 6, 821–840 (2022).
Seidman, J. S. et al. Niche-specific reprogramming of epigenetic landscapes drives myeloid cell diversity in nonalcoholic steatohepatitis. Immunity 52, 1057–1074.e7 (2020).
Klein, I. et al. Kupffer cell heterogeneity: functional properties of bone marrow derived and sessile hepatic macrophages. Blood 110, 4077–4085 (2007).
Beattie, L. et al. Bone marrow-derived and resident liver macrophages display unique transcriptomic signatures but similar biological functions. J. Hepatol. 65, 758–768 (2016).
Guilliams, M. & Scott, C. L. Liver macrophages in health and disease. Immunity 55, 1515–1529 (2022).
Yang, C.-Y. et al. CLEC4F is an inducible C-type lectin in F4/80-positive cells and is involved in alpha-galactosylceramide presentation in liver. PLoS ONE 8, e65070 (2013).
Lavin, Y. et al. Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell 159, 1312–1326 (2014).
Scott, C. L. et al. The transcription factor ZEB2 is required to maintain the tissue-specific identities of macrophages. Immunity 49, 312–325.e5 (2018).
Aizarani, N. et al. A human liver cell atlas reveals heterogeneity and epithelial progenitors. Nature 572, 199–204 (2019).
Ramachandran, P. et al. Resolving the fibrotic niche of human liver cirrhosis at single-cell level. Nature 575, 512–518 (2019).
Jiang, Y. et al. Kupffer cell receptor CLEC4F is important for the destruction of desialylated platelets in mice. Cell Death Differ. 28, 3009–3021 (2021).
Blériot, C. et al. A subset of Kupffer cells regulates metabolism through the expression of CD36. Immunity 54, 2101–2116.e6 (2021). This study identifies a new discrete Kupffer cell population that expands during metabolic challenges and displays distinct metabolic programs.
De Simone, G. et al. Identification of a Kupffer cell subset capable of reverting the T cell dysfunction induced by hepatocellular priming. Immunity 54, 2089–2100.e8 (2021). This study showed that a Kupffer cell population can cross-present hepatocyte-derived antigens in response to IL-2, thereby sustaining the function of intrahepatic dysfunctional CD8⁺ T cells.
Hume, D. A., Offermanns, S. & Bonnavion, R. Contamination of isolated mouse Kupffer cells with liver sinusoidal endothelial cells. Immunity 55, 1139–1140 (2022).
Millard, S. M. et al. Fragmentation of tissue-resident macrophages during isolation confounds analysis of single-cell preparations from mouse hematopoietic tissues. Cell Rep. 37, 110058 (2021).
De Ponti, F. F. et al. Spatially restricted and ontogenically distinct hepatic macrophages are required for tissue repair. Immunity 58, 362–380.e10 (2025). This work uncovered that both embryo-derived and monocyte-derived KCs can acquire a lipid-associated macrophage phenotype promoting tissue clearance and repair.
Xu, Y. et al. Identification of conserved and tissue-restricted transcriptional profiles for lipid associated macrophages. Commun. Biol. 8, 953 (2025).
Molgora, M. et al. TREM2 modulation remodels the tumor myeloid landscape enhancing anti-PD-1 immunotherapy. Cell 182, 886–900.e17 (2020).
Dunsmore, G. et al. Timing and location dictate monocyte fate and their transition to tumor-associated macrophages. Sci. Immunol. 9, eadk3981 (2024).
Ulland, T. K. et al. TREM2 maintains microglial metabolic fitness in Alzheimer’s disease. Cell 170, 649–663.e13 (2017).
Jaitin, D. A. et al. Lipid-associated macrophages control metabolic homeostasis in a Trem2-dependent manner. Cell 178, 686–698.e14 (2019).
Liang, Y. et al. Temporal analyses of postnatal liver development and maturation by single-cell transcriptomics. Dev. Cell 57, 398–414.e5 (2022).
Miyamoto, Y. et al. Periportal macrophages protect against commensal-driven liver inflammation. Nature 629, 901–909 (2024). This study discovered a specialized population of periportal macrophages that sense gut-derived microbial signals and act as a frontline barrier preventing commensal-driven inflammation from spreading into the liver.
Martini, T., Naef, F. & Tchorz, J. S. Spatiotemporal metabolic liver zonation and consequences on pathophysiology. Annu. Rev. Pathol. 18, 439–466 (2023).
Ben-Moshe, S. et al. Spatial sorting enables comprehensive characterization of liver zonation. Nat. Metab. 1, 899–911 (2019).
Dobie, R. et al. Single-cell transcriptomics uncovers zonation of function in the mesenchyme during liver fibrosis. Cell Rep. 29, 1832–1847.e8 (2019).
Inverso, D. et al. A spatial vascular transcriptomic, proteomic, and phosphoproteomic atlas unveils an angiocrine Tie-Wnt signaling axis in the liver. Dev. Cell 56, 1677–1693.e10 (2021).
Iannacone, M. et al. Response to contamination of isolated mouse Kupffer cells with liver sinusoidal endothelial cells. Immunity 55, 1141–1142 (2022).
Sleyster, E. C. & Knook, D. L. Relation between localization and function of rat liver Kupffer cells. Lab. Invest. 47, 484–490 (1982).
Bouwens, L., Baekeland, M., De Zanger, R. & Wisse, E. Quantitation, tissue distribution and proliferation kinetics of Kupffer cells in normal rat liver. Hepatology 6, 718–722 (1986).
Bouwens, L., De Bleser, P., Vanderkerken, K., Geerts, B. & Wisse, E. Liver cell heterogeneity: functions of non-parenchymal cells. Enzyme 46, 155–168 (1992).
Itoh, Y. et al. Functional heterogeneity of rat liver macrophages: interleukin-1 secretion and Ia antigen expression in contrast with phagocytic activity. Liver 12, 26–33 (1992).
Gola, A. et al. Commensal-driven immune zonation of the liver promotes host defence. Nature 589, 131–136 (2021). This work demonstrated that commensal-derived endotoxins activate LSECs to remodel the glycocalyx, enabling CXCL9 retention and positioning Kupffer cells around the portal tract through CXCR3-dependent recruitment.
Feng, D. et al. Characterisation of macrophages in healthy and diseased livers in mice: identification of necrotic lesion-associated macrophages. eGastroenterology 3, e100189 (2025).
Ioannou, G. N. Epidemiology and risk-stratification of NAFLD-associated HCC. J. Hepatol. 75, 1476–1484 (2021).
Riazi, K. et al. The prevalence and incidence of NAFLD worldwide: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 7, 851–861 (2022).
Alonso, C. et al. Metabolomic identification of subtypes of nonalcoholic steatohepatitis. Gastroenterology 152, 1449–1461.e7 (2017).
Younossi, Z. M. et al. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology 77, 1335–1347 (2023).
Younossi, Z. M. et al. Global epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64, 73–84 (2016).
Diniz, A. B. et al. Imaging and immunometabolic phenotyping uncover changes in the hepatic immune response in the early phases of NAFLD. JHEP Rep. 2, 100117 (2020).
Govaere, O. et al. Macrophage scavenger receptor 1 mediates lipid-induced inflammation in non-alcoholic fatty liver disease. J. Hepatol. 76, 1001–1012 (2022).
Huang, H. et al. Kupffer cell programming by maternal obesity triggers fatty liver disease. Nature 644, 790–798 (2025). This study showed that maternal obesity epigenetically reprograms fetal KCs towards glycolytic metabolism, creating a long-lasting imprint that promotes lipid accumulation and fatty liver disease in offspring.
Mulder, K. et al. Cross-tissue single-cell landscape of human monocytes and macrophages in health and disease. Immunity 54, 1883–1900.e5 (2021).
Guo, W. et al. Notch signaling regulates macrophage-mediated inflammation in metabolic dysfunction-associated steatotic liver disease. Immunity 57, 2310–2327.e6 (2024).
Daemen, S. et al. Dynamic shifts in the composition of resident and recruited macrophages influence tissue remodeling in NASH. Cell Rep. 34, 108626 (2021).
Krenkel, O. et al. Myeloid cells in liver and bone marrow acquire a functionally distinct inflammatory phenotype during obesity-related steatohepatitis. Gut 69, 551–563 (2020).
Morinaga, H. et al. Characterization of distinct subpopulations of hepatic macrophages in HFD/obese mice. Diabetes 64, 1120–1130 (2015).
Morgantini, C. et al. Liver macrophages regulate systemic metabolism through non-inflammatory factors. Nat. Metab. 1, 445–459 (2019).
Kohyama, M. et al. Role for Spi-C in the development of red pulp macrophages and splenic iron homeostasis. Nature 457, 318–321 (2009).
Haldar, M. et al. Heme-mediated SPI-C induction promotes monocyte differentiation into iron-recycling macrophages. Cell 156, 1223–1234 (2014).
Willy, P. J. et al. LXR, a nuclear receptor that defines a distinct retinoid response pathway. Genes Dev. 9, 1033–1045 (1995).
Spann, N. J. & Glass, C. K. Sterols and oxysterols in immune cell function. Nat. Immunol. 14, 893–900 (2013).
Calkin, A. C. & Tontonoz, P. Transcriptional integration of metabolism by the nuclear sterol-activated receptors LXR and FXR. Nat. Rev. Mol. Cell Biol. 13, 213–224 (2012).
Spann, N. J. et al. Regulated accumulation of desmosterol integrates macrophage lipid metabolism and inflammatory responses. Cell 151, 138–152 (2012).
Pehkonen, P. et al. Genome-wide landscape of liver X receptor chromatin binding and gene regulation in human macrophages. BMC Genomics 13, 50 (2012).
Costet, P., Luo, Y., Wang, N. & Tall, A. R. Sterol-dependent transactivation of the ABC1 promoter by the liver X receptor/retinoid X receptor. J. Biol. Chem. 275, 28240–28245 (2000).
Venkateswaran, A. et al. Control of cellular cholesterol efflux by the nuclear oxysterol receptor LXRα. Proc. Natl Acad. Sci. USA 97, 12097–12102 (2000).
Westerterp, M. et al. Deficiency of ATP-binding cassette transporters A1 and G1 in macrophages increases inflammation and accelerates atherosclerosis in mice. Circ. Res. 112, 1456–1465 (2013).
Odegaard, J. I. et al. Alternative M2 activation of Kupffer cells by PPARdelta ameliorates obesity-induced insulin resistance. Cell Metab. 7, 496–507 (2008).
Wan, J. et al. M2 Kupffer cells promote M1 Kupffer cell apoptosis: a protective mechanism against alcoholic and nonalcoholic fatty liver disease. Hepatology 59, 130–142 (2014).
Francque, S. et al. Nonalcoholic steatohepatitis: the role of peroxisome proliferator-activated receptors. Nat. Rev. Gastroenterol. Hepatol. 18, 24–39 (2021).
Francque, S. M. et al. A randomized, controlled trial of the pan-PPAR agonist lanifibranor in NASH. N. Engl. J. Med. 385, 1547–1558 (2021).
Huang, S. C.-C. et al. Cell-intrinsic lysosomal lipolysis is essential for alternative activation of macrophages. Nat. Immunol. 15, 846–855 (2014).
Pfeffer, S. R. NPC intracellular cholesterol transporter 1 (NPC1)-mediated cholesterol export from lysosomes. J. Biol. Chem. 294, 1706–1709 (2019).
Kanamori, Y. et al. Iron-rich Kupffer cells exhibit phenotypic changes during the development of liver fibrosis in NASH. iScience 24, 102032 (2021).
Theurl, I. et al. On-demand erythrocyte disposal and iron recycling requires transient macrophages in the liver. Nat. Med. 22, 945–951 (2016). This work showed that, during acute haemolysis, monocyte-derived macrophages transiently colonize the liver to clear damaged erythrocytes and recycle iron, revealing a flexible backup system that complements the homeostatic role of KCs.
Willekens, F. L. A. et al. Liver Kupffer cells rapidly remove red blood cell-derived vesicles from the circulation by scavenger receptors. Blood 105, 2141–2145 (2005).
Lee, S.-J., Park, S.-Y., Jung, M.-Y., Bae, S. M. & Kim, I.-S. Mechanism for phosphatidylserine-dependent erythrophagocytosis in mouse liver. Blood 117, 5215–5223 (2011).
Park, M. D., Silvin, A., Ginhoux, F. & Merad, M. Macrophages in health and disease. Cell 185, 4259–4279 (2022).
Christ, A. et al. Western diet triggers NLRP3-dependent innate immune reprogramming. Cell 172, 162–175.e14 (2018).
Hegde, S. et al. Myeloid progenitor dysregulation fuels immunosuppressive macrophages in tumours. Nature 646, 1214–1222 (2025).
English, K. et al. A hepatic network of dendritic cells mediates CD4 T cell help outside lymphoid organs. Nat. Commun. 15, 1261 (2024).
Iannacone, M. & Guidotti, L. G. Immunobiology and pathogenesis of hepatitis B virus infection. Nat. Rev. Immunol. 22, 19–32 (2022).
Venzin, V. et al. CD4 + T cells license Kupffer cells to reverse CD8 + T cell dysfunction induced by hepatocellular priming. Nat. Immunol. https://doi.org/10.1038/s41590-025-02199-3 (2025). This study demonstrated that CD4⁺ T cells directly interact with KCs in the liver through CD40–CD40L, inducing IL-12 and IL-27 production, with IL-27 being essential to sustain antiviral CD8⁺ T cell function.
Knolle, P. et al. Human Kupffer cells secrete IL-10 in response to lipopolysaccharide (LPS) challenge. J. Hepatol. 22, 226–229 (1995).
Li, H. et al. Hepatitis B virus particles preferably induce Kupffer cells to produce TGF-β1 over pro-inflammatory cytokines. Dig. Liver Dis. 44, 328–333 (2012).
Tian, Y. et al. Kupffer cell-dependent TNF-alpha signaling mediates injury in the arterialized small-for-size liver transplantation in the mouse. Proc. Natl Acad. Sci. USA 103, 4598–4603 (2006).
Ait Ahmed, Y. et al. Kupffer cell restoration after partial hepatectomy is mainly driven by local cell proliferation in IL-6-dependent autocrine and paracrine manners. Cell. Mol. Immunol. 18, 2165–2176 (2021).
Tian, Y., Kuo, C.-F., Akbari, O. & Ou, J.-H. J. Maternal-derived hepatitis b virus e antigen alters macrophage function in offspring to drive viral persistence after vertical transmission. Immunity 44, 1204–1214 (2016).
Lee, W.-Y. et al. An intravascular immune response to Borrelia burgdorferi involves Kupffer cells and iNKT cells. Nat. Immunol. 11, 295–302 (2010).
Liew, P. X., Lee, W.-Y. & Kubes, P. iNKT cells orchestrate a switch from inflammation to resolution of sterile liver injury. Immunity 47, 752–765.e5 (2017).
Van Der Meijden, P. E. J. & Heemskerk, J. W. M. Platelet biology and functions: new concepts and clinical perspectives. Nat. Rev. Cardiol. 16, 166–179 (2019).
Deppermann, C. et al. Macrophage galactose lectin is critical for Kupffer cells to clear aged platelets. J. Exp. Med. 217, e20190723 (2020).
Helmy, K. Y. et al. CRIg: a macrophage complement receptor required for phagocytosis of circulating pathogens. Cell 124, 915–927 (2006). This seminal work identified CRIg as a KC-specific receptor and a dominant component of the phagocytic system mediating rapid clearance of C3-opsonized particles from the circulation.
Holers, V. M. Complement and its receptors: new insights into human disease. Annu. Rev. Immunol. 32, 433–459 (2014).
He, J. Q. et al. CRIg mediates early Kupffer cell responses to adenovirus. J. Leukoc. Biol. 93, 301–306 (2013).
Zeng, Z. et al. CRIg functions as a macrophage pattern recognition receptor to directly bind and capture blood-borne Gram-positive bacteria. Cell Host Microbe 20, 99–106 (2016).
Canton, J., Neculai, D. & Grinstein, S. Scavenger receptors in homeostasis and immunity. Nat. Rev. Immunol. 13, 621–634 (2013).
Surewaard, B. G. J. et al. Identification and treatment of the Staphylococcus aureus reservoir in vivo. J. Exp. Med. 213, 1141–1151 (2016).
Surewaard, B. G. J. et al. α-Toxin induces platelet aggregation and liver injury during Staphylococcus aureus sepsis. Cell Host Microbe 24, 271–284.e3 (2018).
Jorch, S. K. et al. Peritoneal GATA6+ macrophages function as a portal for Staphylococcus aureus dissemination. J. Clin. Invest. 129, 4643–4656 (2019).
Hommes, J. W. & Surewaard, B. G. J. Intracellular habitation of Staphylococcus aureus: molecular mechanisms and prospects for antimicrobial therapy. Biomedicines 10, 1804 (2022).
Egen, J. G. et al. Macrophage and T cell dynamics during the development and disintegration of mycobacterial granulomas. Immunity 28, 271–284 (2008).
An, H. et al. Functional vulnerability of liver macrophages to capsules defines virulence of blood-borne bacteria. J. Exp. Med. 219, e20212032 (2022).
Huang, X. et al. Capsule type defines the capability of Klebsiella pneumoniae in evading Kupffer cell capture in the liver. PLoS Pathog. 18, e1010693 (2022).
An, H. et al. Splenic red pulp macrophages eliminate the liver-resistant Streptococcus pneumoniae from the blood circulation of mice. Sci. Adv. 11, eadq6399 (2025).
Deniset, J. F., Surewaard, B. G., Lee, W.-Y. & Kubes, P. Splenic Ly6Ghigh mature and Ly6Gint immature neutrophils contribute to eradication of S. pneumoniae. J. Exp. Med. 214, 1333–1350 (2017).
Wang, J. et al. Liver macrophages and sinusoidal endothelial cells execute vaccine-elicited capture of invasive bacteria. Sci. Transl. Med. 15, eade0054 (2023).
Tian, X. et al. Natural antibodies to polysaccharide capsules enable Kupffer cells to capture invading bacteria in the liver sinusoids. J. Exp. Med. 222, e20240735 (2025).
Lopes, M. E. et al. Susceptibility to infections during acute liver injury depends on transient disruption of liver macrophage niche. Front. Immunol. 13, 892114 (2022).
Triantafyllou, E. et al. PD-1 blockade improves Kupffer cell bacterial clearance in acute liver injury. J. Clin. Invest. 131, e140196 (2021).
Peiseler, M. et al. Kupffer cell-like syncytia replenish resident macrophage function in the fibrotic liver. Science 381, eabq5202 (2023). This work discovered that, in liver fibrosis, monocyte-derived macrophages can fuse into multinucleated syncytia that occupy the Kupffer cell niche and partially restore its filtering functions.
Borst, K. et al. Type I interferon receptor signaling delays Kupffer cell replenishment during acute fulminant viral hepatitis. J. Hepatol. 68, 682–690 (2018).
Liu, W. et al. In situ expansion and reprogramming of Kupffer cells elicit potent tumoricidal immunity against liver metastasis. J. Clin. Invest. 133, e157937 (2023).
Brown, G. D. et al. Hidden killers: human fungal infections. Sci. Transl. Med. 4, 165rv13 (2012).
Sun, D. et al. Fungal dissemination is limited by liver macrophage filtration of the blood. Nat. Commun. 10, 4566 (2019).
Lin, Y.-Y., Shiau, S. & Fang, C.-T. Risk factors for invasive Cryptococcus neoformans diseases: a case-control study. PLoS ONE 10, e0119090 (2015).
Singh, N. et al. Cryptococcosis in patients with cirrhosis of the liver and posttransplant outcomes. Transplantation 99, 2132–2141 (2015).
Spec, A., Raval, K. & Powderly, W. G. End-stage liver disease is a strong predictor of early mortality in cryptococcosis. Open. Forum Infect. Dis. 3, ofv197 (2016).
Beattie, L. et al. Dynamic imaging of experimental Leishmania donovani-induced hepatic granulomas detects Kupffer cell-restricted antigen presentation to antigen-specific CD8 T cells. PLoS Pathog. 6, e1000805 (2010).
Moyo, D. et al. Macrophage transactivation for chemokine production identified as a negative regulator of granulomatous inflammation using agent-based modeling. Front. Immunol. 9, 637 (2018).
Pessenda, G. et al. Kupffer cell and recruited macrophage heterogeneity orchestrate granuloma maturation and hepatic immunity in visceral leishmaniasis. Nat. Commun. 16, 3125 (2025).
Musrati, M. A. et al. Infection history imprints prolonged changes to the epigenome, transcriptome and function of Kupffer cells. J. Hepatol. 81, 1023–1039 (2024).
Kourtzelis, I., Hajishengallis, G. & Chavakis, T. Phagocytosis of apoptotic cells in resolution of inflammation. Front. Immunol. 11, 553 (2020).
Doran, A. C., Yurdagul, A. & Tabas, I. Efferocytosis in health and disease. Nat. Rev. Immunol. 20, 254–267 (2020).
Liebold, I. et al. Apoptotic cell identity induces distinct functional responses to IL-4 in efferocytic macrophages. Science 384, eabo7027 (2024). This study revealed that macrophages interpret IL-4 signals differently depending on the type of apoptotic cell they engulf, showing that efferocytosis imprints a context-specific transcriptional and functional state that shapes tissue repair and immune regulation.
Horst, A. K., Tiegs, G. & Diehl, L. Contribution of macrophage efferocytosis to liver homeostasis and disease. Front. Immunol. 10, 2670 (2019).
Hochreiter-Hufford, A. & Ravichandran, K. S. Clearing the dead: apoptotic cell sensing, recognition, engulfment, and digestion. Cold Spring Harb. Perspect. Biol. 5, a008748 (2013).
Boada-Romero, E., Martinez, J., Heckmann, B. L. & Green, D. R. The clearance of dead cells by efferocytosis. Nat. Rev. Mol. Cell Biol. 21, 398–414 (2020).
Sitia, G., Iannacone, M., Müller, S., Bianchi, M. E. & Guidotti, L. G. Treatment with HMGB1 inhibitors diminishes CTL-induced liver disease in HBV transgenic mice. J. Leukoc. Biol. 81, 100–107 (2007).
Dong, S. et al. Mechanisms of CCl4-induced liver fibrosis with combined transcriptomic and proteomic analysis. J. Toxicol. Sci. 41, 561–572 (2016).
Miyazaki, H. et al. Fatty acid binding protein 7 regulates phagocytosis and cytokine production in Kupffer cells during liver injury. Am. J. Pathol. 184, 2505–2515 (2014).
Shi, H. et al. Impaired TIM4-mediated efferocytosis by liver macrophages contributes to fibrosis in metabolic dysfunction–associated steatohepatitis. Sci. Transl. Med. 17, eadv2106 (2025).
Rumgay, H. et al. Global burden of primary liver cancer in 2020 and predictions to 2040. J. Hepatol. 77, 1598–1606 (2022).
Horn, S. R. et al. Epidemiology of liver metastases. Cancer Epidemiol. 67, 101760 (2020).
Sharma, A. et al. Onco-fetal reprogramming of endothelial cells drives immunosuppressive macrophages in hepatocellular carcinoma. Cell 183, 377–394.e21 (2020).
Sharma, A., Blériot, C., Currenti, J. & Ginhoux, F. Oncofetal reprogramming in tumour development and progression. Nat. Rev. Cancer 22, 593–602 (2022).
Kimura, Y. et al. The innate immune receptor Dectin-2 mediates the phagocytosis of cancer cells by Kupffer cells for the suppression of liver metastasis. Proc. Natl Acad. Sci. USA 113, 14097–14102 (2016). This work showed that KCs protect the liver at the onset of metastatic dissemination by using Dectin-2 to phagocytose circulating cancer cells, in contrast to their immunosuppressive role once the tumour is established.
Gül, N. et al. Macrophages eliminate circulating tumor cells after monoclonal antibody therapy. J. Clin. Invest. 124, 812–823 (2014).
Deng, Z. et al. The nuclear factor ID3 endows macrophages with a potent anti-tumour activity. Nature 626, 864–873 (2024). This study showed that ID3 programs a Kupffer cell transcriptional state that enhances phagocytosis of metastatic cells and recruitment of cytotoxic lymphocytes, uncovering a core mechanism by which KCs suppress liver tumour growth.
Yang, P. et al. CD36-mediated metabolic crosstalk between tumor cells and macrophages affects liver metastasis. Nat. Commun. 13, 5782 (2022).
Wang, G. et al. Tumour extracellular vesicles and particles induce liver metabolic dysfunction. Nature 618, 374–382 (2023).
Wortzel, I. et al. Unique structural configuration of EV-DNA primes Kupffer cell-mediated antitumor immunity to prevent metastatic progression. Nat. Cancer 5, 1815–1833 (2024).
Halpern, K. B. et al. Single-cell spatial reconstruction reveals global division of labour in the mammalian liver. Nature 542, 352–356 (2017).
Halpern, K. B. et al. Paired-cell sequencing enables spatial gene expression mapping of liver endothelial cells. Nat. Biotechnol. 36, 962–970 (2018).
Hildebrandt, F. et al. Spatial transcriptomics to define transcriptional patterns of zonation and structural components in the mouse liver. Nat. Commun. 12, 7046 (2021).
Blériot, C., Dunsmore, G., Alonso-Curbelo, D. & Ginhoux, F. A temporal perspective for tumor-associated macrophage identities and functions. Cancer Cell https://doi.org/10.1016/j.ccell.2024.04.002 (2024).
Michels, D. A., Parker, M. & Salas-Solano, O. Quantitative impurity analysis of monoclonal antibody size heterogeneity by CE-LIF: example of development and validation through a quality-by-design framework. Electrophoresis 33, 815–826 (2012).
Breitkopf-Heinlein, K. et al. BMP-9 interferes with liver regeneration and promotes liver fibrosis. Gut 66, 939–954 (2017).
Zhao, D. et al. ALK1 signaling is required for the homeostasis of Kupffer cells and prevention of bacterial infection. J. Clin. Invest. 132, e150489 (2022).
van de Laar, L. et al. Yolk sac macrophages, fetal liver, and adult monocytes can colonize an empty niche and develop into functional tissue-resident macrophages. Immunity 44, 755–768 (2016).
Ganguly, S. et al. Lipid-associated macrophages’ promotion of fibrosis resolution during MASH regression requires TREM2. Proc. Natl Acad. Sci. USA 121, e2405746121 (2024).