A herb found in popular cold and flu supplements in Australia has officially been linked with life-threatening anaphylaxis.
The Therapeutic Goods Administration (TGA) has completed a safety review of Andrographis paniculata, which is used in approximately 84 listed medicines that are marketed to relieve cold and flu symptoms and boost immunity.
Listed medicines are regulated as low-risk, can only contain ingredients that are classified as low-risk and can be bought from retail outlets including pharmacies.
The TGA’s safety review has found that people taking supplements containing Andrographis have experienced “unpredictable” reactions to the herb, regardless of whether they have taken the product for the first time or have used it previously.
Since 2005, the regulator has received 1,368 reports of adverse reactions related to medicines containing Andrographis, including 287 incidents of anaphylaxis.
“The sustained high reporting pattern since 2019 indicates that Andrographis is associated with risk of life-threatening anaphylaxis which is inconsistent with the low-risk regulatory framework for listed medicines,” the TGA’s report stated.
“Current risk mitigation strategies, including stronger label warnings, have not reduced the risk of anaphylaxis from Andrographis for it to be suitable for use in listed medicines.”

Connie Katelaris says products containing Andrographis paniculata should be removed from the shelves of pharmacies. (ABC News: Jerry Rickard)
Connie Katelaris AM is a professor of immunology and allergy at Western Sydney University and has been warning the TGA that the risk posed by the herb outweighs any perceived benefits.
“This stuff causes harm, it has caused death and this is a global thing,” Professor Katelaris said.
“We’ve got data from many countries showing adverse responses to this product.
“I am in complete agreement with the [TGA’s] conclusions that it’s unsafe and that despite warnings and stickers on bottles, these things have been inadequate in stopping people using it and still having reactions.”
In 2019, products containing Andrographis had to carry a warning label stating that the ingredient may cause allergic reactions in some people.
In August 2024, BioCeuticals, which manufactures ArmaForce, one of the most popular cold and flu supplements containing the herb, updated its warning label to say that the product may cause anaphylaxis.
ArmaForce is one of dozens of products containing the herb Andrographis paniculata. (ABC News: Jerry Rickard)
“The number of anaphylaxis reports for this medicine in 2025 remains consistent with the high number of anaphylaxis reports for this medicine in 2022 and 2023 prior to the introduction of the strengthened label warning in 2024,” the TGA’s review said.
The regulator has concluded that stronger label warnings would be inadequate.
“For consumers experiencing these symptoms, the label warning becomes irrelevant,” the report said.
“A label warning is also not helpful for people who did not have quick access to emergency treatment including adrenaline.”
Patient’s frustration
Carmen Wells is among the people who experienced an adverse reaction after taking a product containing Andrographis.
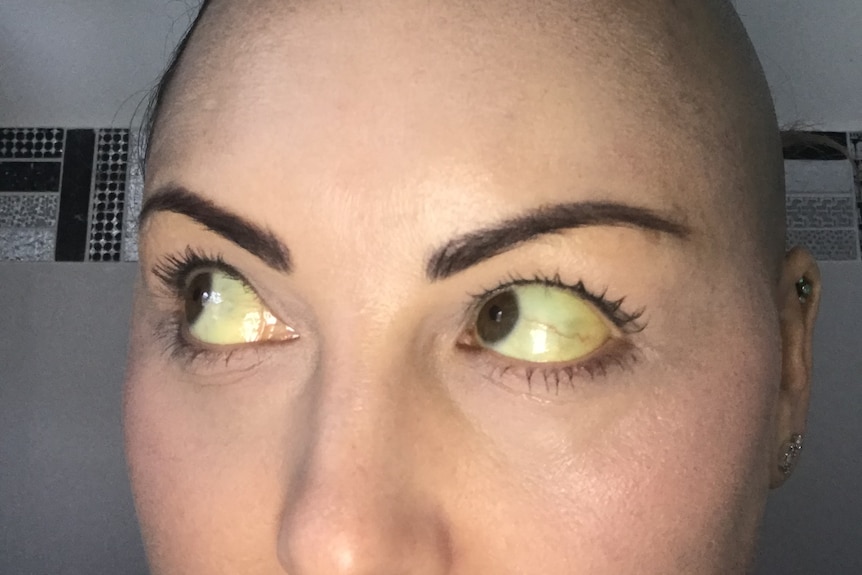
Carmen Wells’s doctor initially diagnosed her with a drug-induced liver injury after taking a cold and flu supplement containing the herb. (Supplied: Carmen Wells)
In 2019 she used a supplement to help treat a cold, and in the days afterwards, she realised something was wrong when she noticed her eyes and skin turning yellow.
Ms Wells was initially diagnosed with drug-induced liver injury, which was revised to drug-induced autoimmune hepatitis.
“For me, six years down the track, I’m having to manage my health still, so I’m back on regular blood testing,” she said.
“It’s always in the background, it’s always something I have to think about.”
Ms Wells said she was pleased with the results of the TGA’s safety review.
“I think it’s probably what people have been saying and it’s great to have that identified through the research,” she said.
“It was good to see it concluded what we’ve known but also frustration that it’s taken this long to actually have this data out.”
Further consultation
The TGA started this safety review in July 2024, a month after it received a report of a fatal anaphylactic reaction in Queensland.
Professor Katelaris said many people had suffered adverse reactions while the regulator was conducting its review.

An updated warning label was applied to one of the cold and flu supplements in August 2024. (ABC News: Jerry Rickard)
“I’m totally frustrated by the length of time [it’s taken],” she said.
“The data has been accumulating, it’s very clear-cut.
“[The TGA’s] own statement says that their warnings to date have been inadequate in preventing harm and it’s taken so long to get to this point and this stuff is still being sold to the public.”
The TGA has announced it will now consult health professionals, manufacturers and consumer groups on its proposal to remove Andrographis from the list of low-risk ingredients.
In a statement, a spokesperson for the TGA said the consultation period closed on April 30, and the regulator would then review and consider the responses.
“Should the TGA decide to remove Andrographis from the Permissable Ingredients Determination, it will become unavailable for use as an ingredient in the low-risk listed medicine products,” the spokesperson said.
The TGA said Andrographis was also available for use as an ingredient in prescription and registered non-prescription medicines, and there was no proposal to remove the herb from use in these other medicine categories.
Professor Katelaris has criticised the TGA for starting a consultation process.
“I think it’s completely unnecessary. [The TGA’s] own expert committees have met over eight months ago, documents have come out and been publishing stating … this stuff is unsafe,” Professor Katelaris said.
“I don’t understand what is going to be gained by putting it out for stakeholder comment at this point? The TGA should just act and remove it from the shelves.”
Carmen Wells also does not believe further consultation is required.
“What is concerning for me is that, why do you need to do more consultation?
“We have the data, we know that the adverse reactions are happening.
“What more do you need to determine that this is not a low-risk product or ingredient. What are they actually looking for when they say, ‘we’re going to consult’?”
In a statement, the TGA said it was required under legislation to “undertake meaningful consultation with persons who may be affected” by any changes.
“Given its role as a risk-based regulator, the TGA must, where possible, understand all risks associated with the regulatory decisions it takes,” the spokesperson said.
“In addition to safety and quality consideration, this includes impact on sponsors, markets, and individuals who use these products.”
In a statement, BioCeuticals said consumer safety and wellbeing were its priority and that all of its products met “current TGA regulatory requirements”.
“We are reviewing the consultation and will take any steps required following the outcome of the TGA consultation.”