The present study investigated whether an increased focus on nutrition in EPT infants contributed to the increase in NEC incidence in Sweden between Epoch 1 (2004–2007) and Epoch 2 (2014–2016). The primary finding of the multivariable logistic model indicated no association between more active enteral nutrition during the first week or the first two postnatal weeks and the risk of late NEC (>7 days). Additionally, we demonstrated that active enteral nutrition policies were implemented more frequently in Epoch 2, resulting in an observed increase in growth at postnatal day 28 and 36 weeks PMA.
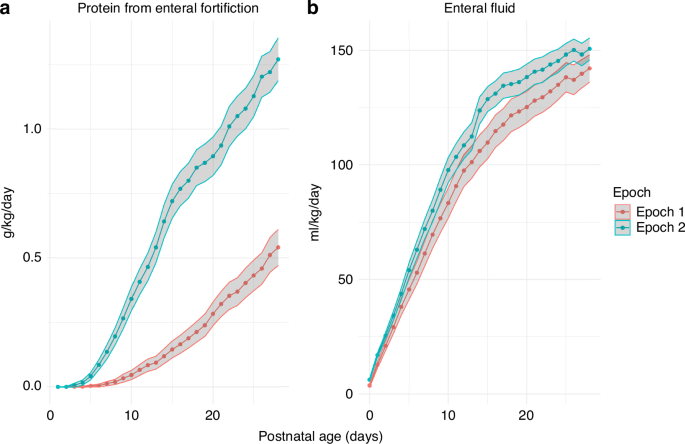
Since our study from 2013 showed that EPT infants during Epoch 1 received energy and macronutrient intake far below the recommended levels and had postnatal growth impairment,12 quality improvement projects, educational activities, and more widespread use of nutrition calculation software were implemented. These changes in clinical practice probably contributed to our observation that during Epoch 2, infants received more active parenteral and enteral nutrition and an increased intake of energy and macronutrients. The energy and protein intakes in Epoch 2 were more aligned with the European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN) 2022 recommendations,2 which likely accounted for the observed increase in weight and length growth in Epoch 2 than in Epoch 1. Notably, similar improvements in growth outcomes have been reported by a Californian quality improvement collaborative that optimised nutritional practices in very low birth weight infants, resulting to a significant reduction in extrauterine growth restriction.21 The ESPGHAN 2022 recommendations for energy and macronutrient intakes have not changed substantially from the 2010 ESPGHAN recommendations present during Epoch 2.22
During Epoch 2, the rate of feeding advancement was more rapid than that in Epoch 1, resulting in faster achievement of full enteral feeding. Despite the advancements in enteral feeding in Epoch 2, almost all infants were below the maximum volume of enteral fluid suggested in the recent ESPGHAN guidelines (200 ml/kg/d).2
It is important to note that during Epochs 1 and 2, evidence regarding the correlation between feeding advancement and NEC was inconclusive. There were concerns that a more rapid rate of feeding advancement might be associated with NEC. An American historical case-control study (2003–2012) examined a slow incremental protocol involving a 2 ml/meal increase every second day (≈35 days to full enteral feeds), compared with a faster advancement of 10–15 ml/kg/d.4 A slower feeding protocol was associated with a reduced NEC incidence among infants with a birth weight <750 g.4 Nonetheless, other variables also changed during the study period, such as the transition from indomethacin to ibuprofen for PDA treatment, and not all infants were exclusively fed human breast milk.4 A British multicentre case-control study conducted in 2004–2005 reached the same conclusion as Viswanathan et al., since infants with NEC were fully fed significantly earlier than controls.3 Owing to the heterogeneity of results from observational studies in the early 2000s, variability in feeding protocols is unsurprising.23 An international web survey conducted in 2012 that included 124 neonatal intensive care units (NICUs) revealed significant differences between countries in the initiation of fortification and nutrition in very preterm infants.23 Scandinavia was more proactive during the initial postnatal days, whereas in Canada, it was common to employ minimal enteral feeding for four to five days. The target volumes also vary among countries.23
The results of our study are compatible with more recent studies, suggesting that the initiation of small volumes of breast milk on the first day of life is considered safe compared to fasting or minimal enteral feeds,24 and that a more rapid feeding advancement does not elevate the risk of NEC.25,26 A recent large multicentre cohort study from the US even reported a protective association between feed advancement and NEC.27 These studies were supported by Cochrane reports.28,29,30 A significant randomised controlled trial (RCT) within the meta-analysis was the SIFT study, a large multicentre RCT involving 2804 very preterm infants, which compared 30 ml/kg/d vs. 18 ml/kg/d increases in feeding advancement, affirming that a rapid increase does not increase the risk of NEC, despite it not being the primary outcome.26
Fortification was more commonly used in the second epoch, with an earlier start and a higher dose. The ESPGHAN recommends that early fortification is as safe as late fortification against the adverse effects of NEC, albeit with a lower degree of evidence.2 A systematic review conducted in 2020 identified only two RCTs, encompassing 171 patients, that compared early and late fortification.31 No adverse effects of NEC were reported with early fortification. However, there was significant heterogeneity among the studies, and the conclusion was graded as having a low level of evidence.31 A Cochrane report from 2020 examined the benefits and risks of multinutrient fortification and included 18 trials involving 1456 preterm infants. The studies were of low methodological quality, and the results suggested that fortification improved growth with low-to-moderate certainty. However, the conclusion that fortification was not harmful and had no effect on NEC could only be made with low certainty based on 13 studies and 1110 infants.32
In the absence of definitive evidence, opinions and practices regarding fortification vary. In a study utilising data from the National Neonatal Research Database for England and Wales, only 40% of very preterm infants received fortification during their NICU stay.33 Arguments against fortification include the absence of clear evidence for optimal growth or protein intake in EPT infants and the possibility of achieving adequate protein levels with higher feeding volumes (180–200 ml/kg/d).33 However, the protein content in human breast milk at four weeks postpartum averages 1.6 g/100 ml,34 which translates to a protein intake of 2.88 g/kg/d at enteral feeds of 180 ml/kg/d. This amount of protein is less than the ESPGHAN recommendations of 3.5–4.0 g protein/kg/d. In the present study, we observed no association between NEC risk and the amount of fortification.
During both epochs, breast milk analysis was performed in the majority of NICUs; however, general routines for targeted fortification and nutrition calculations were not in place until the second epoch. Adjustable and targeted fortification strategies can compensate for variations in human breast milk; still, the optimal strategy remains unclear.2 A Cochrane review from 2020, including eight publications and 521 participants, indicates with low-median certainty that both targeted and adjusted fortification improves weight and length growth in infants with birth weight <1500 g.35
NEC increased 2.5-fold between the epochs,9 and further investigation found that only late NEC (>7 days) increased, and that the increased survival of the most vulnerable infants was the strongest driving factor of the increase.10 Thus, there is no evidence that more active nutrition policies in Epoch 2 were associated with a higher incidence of early NEC, and the present study found no association between more active enteral nutrition policies and the incidence of late NEC (>7 days).
A strength of this dual national cohort study is that it covers 58% of all live-born infants below 27 GW in Sweden over a total period of six years. All NEC diagnoses were rigorously validated, 10,19 and the outcomes and characteristic variables were highly complete.11 The growth and nutritional data were of high resolution and rigorously collected for Epoch 1. The study limitations include reliance on partially validated clinical prescription data in Epoch 2 and a greater extent of imputation than in Epoch 1, with the risk of introducing systematic bias and attenuating short-term variability. The prescription nutritional data were not available from all healthcare regions, resulting in a smaller study population than the original cohorts. This may also have introduced selection bias and may have limited the ability to detect regional differences in nutritional practices and outcomes. The relatively low number of NEC cases in both cohorts, resulting from the exclusion of infants who died early or had missing nutritional data limits the statistical power of the study. Furthermore, the 10-year interval between Epoch 1 and Epoch 2 introduces the potential for temporal confounding due to unmeasured changes in clinical care. Exposure to other NEC risk factors, which were not examined in this study, may have varied between epochs. One such risk factor is exposure to antibiotics.25,36