New series:
Insights on Inflammation
Edited by Bhawan Minhas, OD, and Rami Aboumourad, OD
To help optometrists strengthen their understanding of the ocular inflammatory response, we have created a new seven-part series. Beginning this month and continuing through August, experts will methodically describe how these complex processes manifest in the eye from front to back, starting at the eyelids and moving posteriorly to the optic nerve. Here’s what you can expect over the course of the series:
February: Eyelid and adnexal inflammation
March: Conjunctivitis and episcleritis
April: Keratitis
May: Scleritis
June: Anterior and intermediate uveitis
July: Posterior and panuveitis
August: Optic neuritis
The series will be edited by two prominent ODs with extensive experience in both clinical care and education: Bhawan Minhas, OD, and Rami Aboumourad, OD. Each will also write one installment (Dr. Minhas this month, Dr. Aboumourad in July). We hope you enjoy this tour of all the
-itis diseases of the eye!
—The editors
The ocular surface is a complex ecosystem that serves as the first line of defense for the eye. It consists of the eyelids, conjunctiva, tear film and cornea, each of which is exposed to a range of microorganisms. The ocular surface has the ability to recognize, neutralize and eliminate pathogenic organisms. This ecosystem functions based on a combination of mechanical, anatomical, chemical and immunological defense mechanisms. The eyes’ defense system is twofold, consisting of both innate and adaptive immunity: innate immunity is a nonspecific defense that protects the ocular surface, and adaptive (acquired) immunity is responsible for antigen-specific immune responses once the innate defense has been breached.1
Follow along as we review the ocular surface’s anatomy, functions and anti-infectious defense mechanisms for a thorough understanding of how it operates.
Tear Film Anatomy
The tear film forms a protective barrier between the ocular surface and the external environment. Its primary functions are to lubricate the ocular surface, maintain a smooth surface for refraction, preserve the health of the conjunctiva and cornea and protect the eye from the environment. The tear film is approximately 3µL to 10µL in volume, 3µm thick and secreted at a rate of 1µL/min to 2µL/min.2
Most tears are produced from the main lacrimal glands, and a small amount is produced by the goblet cells in the conjunctiva and accessory lacrimal glands.3 The three types produced are basal tears, reflex tears and closed-eye tears.4 While the tear film has traditionally been described as having three distinct layers (inner: mucin, middle: aqueous and outer: lipid), current evidence suggests a more complex structure. Rather than being separate, the aqueous and mucin components are now understood to form a mucoaqueous gel with a decreasing gradient of mucin concentration from the epithelial surface toward the aqueous phase, covered by an outer lipid layer.5
Defense mechanism. The tear film serves to protect the cornea by removing irritants and pathogens while also weakening toxins and allergens. Every time we blink, the lids promote tear film turnover, removing potential dangers.6
The aqueous layer of the tear film provides a chemical barrier for the ocular surface that contains the following key antimicrobial factors: lysozyme, lactoferrin, transferrin, ceruloplasmin, immunoglobulin (Ig) A, IgG, IgE, complement, glycoprotein and anti-proteinase.4
Each of these factors plays a large part in defending the cornea:
Lysozymes are bacteriolytic and can hydrolyze bacterial peptidoglycan cell walls.Lactoferrin chelates iron and sequesters it from bacteria that need it to grow and metabolize.Immunoglobulins are used in the defense against bacterial, viral and parasitic infections.IgA levels increase during infectious and inflammatory states (e.g., graft rejection, bacterial conjunctivitis, blepharoconjunctivitis).Goblet cells produce mucin and glycoproteins that have decoy receptors for bacteria, making them unable to attach to ocular tissue and trapping bacteria or foreign bodies.3Antimicrobial peptides are endogenous peptides that act on the ocular surface and play a key role in tear film innate immunity via their antimicrobial and wound healing properties.7Host immune cells secrete lipocalin, which binds to siderophores (small ferric iron-binding molecules) secreted by bacteria and fungi to block their reuptake of iron-containing siderophores.8Corneal Structure and Function
The intricacies of the corneal layers continue to be studied; the first descriptions emerged in the mid-19th century, and more recently, the discussion of a novel acellular, strong layer called Dua’s layer was proposed in 2007. The cornea is an aspheric, transparent avascular tissue composed of six layers (from superficial to deep): epithelium, Bowman’s layer, stroma, Dua’s layer, Descemet’s membrane and endothelium.9
The average corneal diameter at birth is 9.5mm to 10.5mm, increasing to 11mm to 12mm horizontally and 10mm to 11mm vertically in adults, with an average central corneal thickness of 535µm to 555µm that increases toward the periphery. The cornea is one of the most densely innervated structures in the body, as it is 20 to 40 times more innervated than tooth pulp and 300 to 600 times more innervated than skin.10 The central corneal nerve density is approximately 7,000 nociceptors per square millimeter.11
The cornea is supplied mainly by sensory fibers of the ophthalmic branch of the trigeminal nerve as well as sympathetic and parasympathetic fibers. The nerves enter the cornea at the corneoscleral limbus, where they form a moderately dense plexus in the mid-stroma and a denser subepithelial plexus in the anterior stroma beneath Bowman’s membrane. These nerves penetrate Bowman’s membrane and form a dense subbasal nerve plexus with branches terminating throughout all layers of the corneal epithelium. One study found increased corneal sensitivity was associated with greater density and number of sub-basal nerves.12
The cornea relies on the diffusion of glucose from the aqueous humor as well as active transport into the corneal epithelium.13 Additionally, the cornea relies on diffusion of atmospheric oxygen through the tear film to maintain transparency in the open eye, whereas closed-eye oxygen is supplied by palpebral conjunctival blood vessels.14
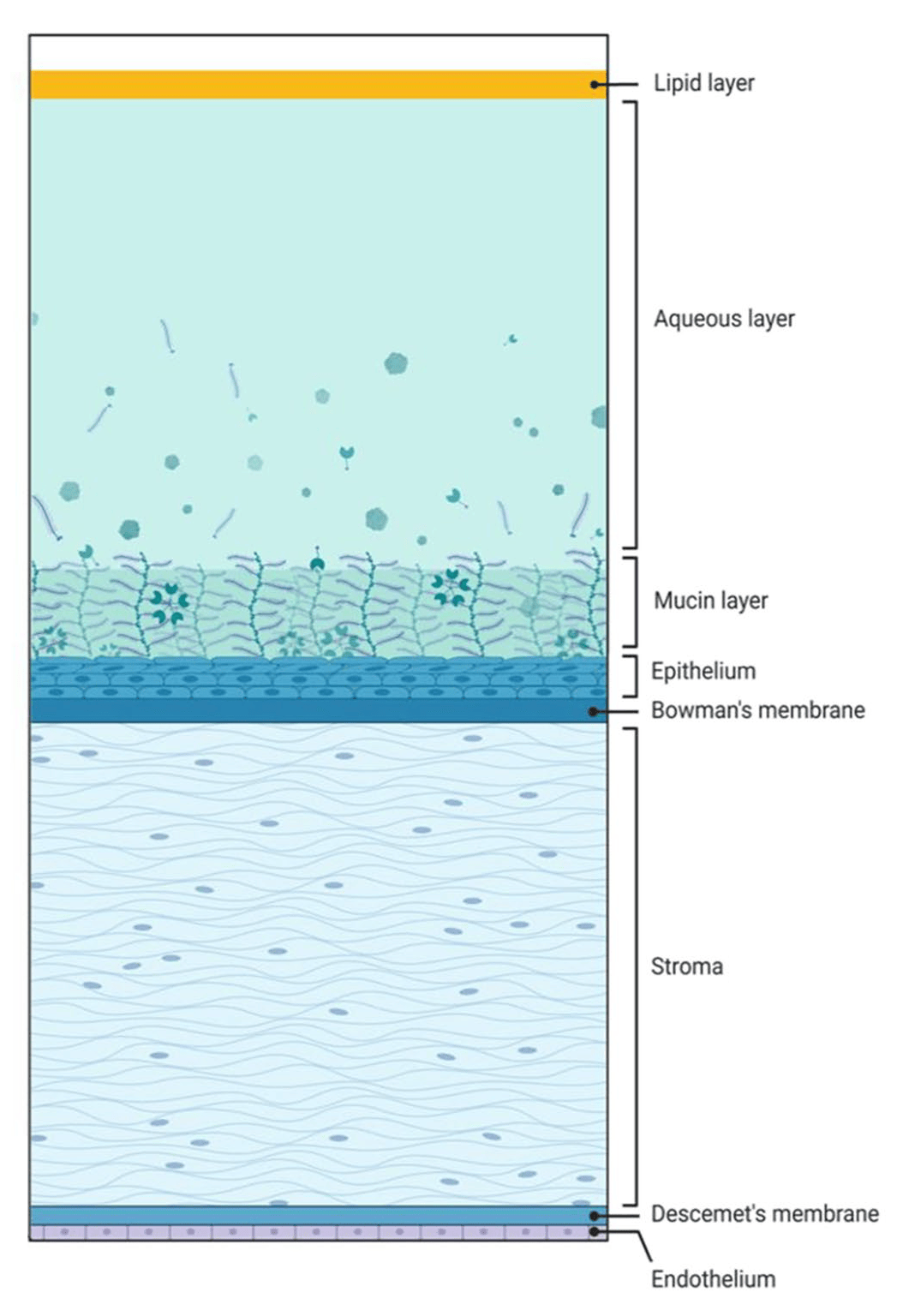
The first line of defense against external threats to the eye is the multilayered tear film and cornea. In addition to these physical barriers, a variety of immune responses can be triggered to summon antimicrobial agents. Photo: Adapted from Kelly DS, Sabharwal S, Ramsey DJ, Morkin MI. The effects of female sex hormones on the human cornea across a woman’s life cycle. BMC Ophthalmol. 2023;23:358 per Creative Commons 4.0. Click image to enlarge.
The hydrophobic corneal epithelium consists of four to six layers; aided by the tear film, the corneal epithelium creates an optimally optically smooth refracting surface for incoming light. Tight junctions between superficial epithelial cells provide an anatomic defense from the environment, as well as preventing diffusion of tear film into the stroma. Corneal clarity depends on the corneal epithelial cells being as tightly packed as possible, creating a layer of almost uniform refractive index with minimal light scatter.
The corneal limbus, which houses the limbal stem cells, is responsible for wound repair of the corneal epithelium. Bowman’s layer lies between the corneal epithelium and the anterior stroma. It was previously described as a membrane, but it’s actually a biomolecular condensate—a tough non-cellular sheet of randomly interwoven collagen fibrils. It is approximately 15µm thick and crucial for corneal shape but does not regenerate after injury.
Beneath Bowman’s layer is the corneal stroma, which makes up approximately 90% of the total corneal thickness. It is composed of keratocytes, collagen fibers and an extracellular matrix. Corneal transparency is dependent on the maintenance of the stromal water content at 78%. Descemet’s membrane, unlike Bowman’s layer, is a true basement membrane for the corneal endothelium. It is secreted by the corneal endothelium and gradually thickens throughout life, with an average adult thickness of 10µm to 12µm. The corneal endothelium is the final posterior surface of the cornea. It is a monolayer of interdigitated cells that are arranged in a mosaic pattern and is essential in maintaining transparency by controlling hydration and maintaining stromal deturgescence.15
Defense mechanism. Corneal defense consists of both innate and acquired immunity. Innate immunity is the cornea’s first line of defense against infection. This immunity is present from birth and provides continuous surveillance of the eye, the orbit and the eyelids. The cornea’s innate immunity is composed of the tear film, epithelial cells, keratocytes, corneal nerves, complement, interferons and other cells, each playing a significant part in defending the cornea, as detailed below:1
Epithelial cells secrete cytokines to activate the immune defense.
– Cytokine interleukin-1α is released when a membrane is ruptured by infectious agents or trauma. It acts as an integral signal for innate immunity. It is responsible for the production of inflammation.
Stromal keratocytes secrete IL-1α.
– Defensins have broad-spectrum antimicrobial activity against bacteria, viruses and fungi by disrupting microbial cell membranes and/or inhibiting growth. They also accelerate epithelial healing.
Corneal nerves relay sensory information, causing reflexive movements to protect the eye. Ocular sensations cause the release of peptides that induce cytokine activity.16
– Ocular surface epithelial cells, nerve endings at inflammatory sites and lacrimal gland tissues can release various peptides, including substance P, vasoactive intestinal peptide, neuropeptide Y and calcitonin gene-related peptide to mediate immune cell activation and infiltration as well as reflex tearing and ocular discomfort.16
Complement (effector and regulatory proteins) generate biologically active molecules.17Interferons are proteins released in response to viral infections, causing a generalized antiviral state in surrounding cells.18Cells of innate immunity include neutrophils, eosinophils, macrophages and natural killer cells:
– Neutrophils play a significant role in phagocytosis and microbial killing.
– Eosinophils play a large role in parasitic toxicity.
– Macrophages carry out phagocytosis, have antigen-presenting capabilities and secrete inflammatory cytokines.
– Natural killer cells destroy compromised cells without prior sensitization.1
The body’s first line of corneal defense (innate immunity) is often strong enough to dispel most infections; however, some organisms can penetrate this line and cause infection, or the innate defense may fail, allowing for opportunistic infection on the ocular surface (an area typically without harmful bacteria).19
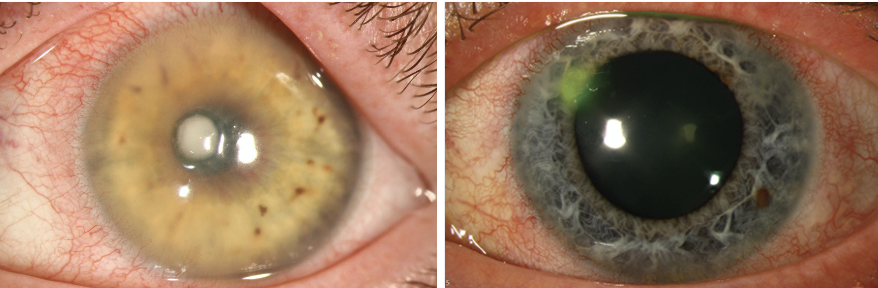
Microbial keratitis tends to be more aggressive when it affects the central cornea (left) vs. the periphery (right), as the central cornea lacks the defensive complement proteins that the peripheral cornea possesses to fight off infections. Photos: Christine Sindt, OD. Click image to enlarge.
Adaptive, or acquired, immunity is called into action when innate immunity fails and/or when it is overwhelmed by a persistent threat. As a second layer of defense, it may take several days to activate a response to specific pathogens. Cell-mediated immunity can help control microbial replication. While acquired immunity has many benefits, it can produce a disproportionate response, leading to irreversible tissue damage by way of scarring or disorganization of the extracellular matrix.
Langerhans cells are antigen-presenting cells that bridge innate and adaptive immunity and are responsible for recognizing, processing and presenting antigens.20 Cytokines produced by T-helper cells can signal alarms and trigger inflammation, while others can prevent overreaction. They are crucial for targeting specific cells and creating memory.
Keratitis
With this condition, inflammatory cells infiltrate layers of the cornea in response to noxious stimuli. These harmful agents can be exogenous (infectious, UV, chemical/exposure, thermal) or endogenous (self-antigens, autoimmune disease). Corneal inflammation can present with variable signs, including infiltration of white blood cells and ciliary congestion. The most common complaints of patients with keratitis are redness, pain and irritation in the eye; however, they may often have photophobia, visual decline and more.
Keratitis is commonly described by distribution, depth, location and shape. Each of these descriptors helps clinicians calibrate the level of attack on the eye and judge how to proceed.
Distribution: diffuse, focal or multifocalDepth: epithelial, subepithelial, anterior stromal, deep stromal or endothelialLocation: central, paracentral or peripheralShape: dendritic, disciform or geographic
Location matters: peripheral vs. central cornea. The peripheral cornea is proximal to the conjunctiva and therefore has all the immunologic machinery to generate an immune response to infection. Antigen-antibody complexes coming from the tears, aqueous humor and/or limbal vessels can activate complement proteins more effectively in the peripheral cornea.1 These are defensive advantages that the central cornea does not have. Therefore, the central cornea is more susceptible to infection. However, the peripheral cornea’s proximity to the corneal vascular arch and concentration of complement and immunoglobulins contribute to a higher risk of autoimmune reaction compared to the central cornea.21
Epithelial inflammation. Superficial keratitis involves the corneal epithelium and superficial stromal inflammation along with conjunctival hyperemia. Punctate lesions (microscopic, nonopaque epithelial erosions) that demonstrate intense fluorescein dye staining can be present, as well as small gray epithelial opacities that can produce a hazy appearance on the cornea, which can cause visual disturbances and discomfort. Superficial keratitis may result from various etiologies, including topical medications, viral conjunctival infection, ultraviolet light exposure, contact lens wear, blepharitis, eyelid abnormalities and dry eyes.22
Stromal inflammation. Inflammation here can occur due to a compromised epithelium or due to infectious agents penetrating an intact epithelium. Inflammatory cells enter the stroma through the tear film, which is normally obstructed. The entrance is usually through an epithelial defect. The stroma can receive inflammatory cells from varying providers depending on the location of the assault on the cornea. If there is endothelial cell damage, inflammatory cells can enter the corneal stroma via the aqueous humor. If the cornea has been vascularized, inflammatory cells can enter directly into the stroma from infiltrating blood vessels. When describing stromal inflammation, the depth and degree of inflammation are important to note.
Stromal inflammation can be devastating to vision because it can lead to opacification and neovascularization. When stromal keratocytes are altered, the new collagen fibers made are disorganized, scatter light and cause scarring.
Endothelial inflammation. This can cause inflammatory pseudoguttae, which are swollen endothelial cells visible on specular reflection. Keratic precipitates—clumps of inflammatory cells—can develop and adhere to the back of the endothelium. The presence of keratic precipitates demonstrates a disruption of the blood-aqueous barrier that occurs during the inflammatory process.23
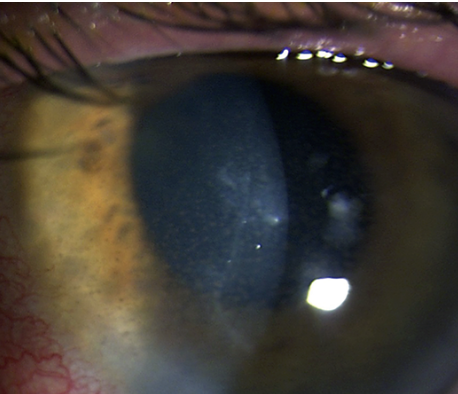
Keratic precipitates with anterior chamber reaction and stromal haze—all hallmarks of HSV endothelial keratitis. Photo: Lisa Martén, MD. Click image to enlarge.
Infectious vs. Autoimmune
The mechanisms and treatment of keratitis depend on whether the underlying etiology is infectious or autoimmune. Below, we’ll break down the pathophysiology of each corneal infection type.
Infectious keratitis. This is classified as microbial keratitis (bacterial, fungal or protozoal) or viral keratitis (herpes). Various pathogens can cause infectious keratitis, including bacteria like Pseudomonas aeruginosa and Streptococcus pneumoniae, fungi like Candida, protozoa like Acanthamoeba and viruses like herpes simplex virus.
Infectious keratitis is the fifth leading cause of blindness globally. Ocular injury, ocular surface diseases and contact lens wear are the major predisposing factors to infectious keratitis.24 However, infectious keratitis can also present without clinically demonstrable corneal abrasions. Tiny corneal injuries that are undetectable may allow bacterial attachment to the corneal surface, which can lead to infectious keratitis.25 Culturing corneal scrapes is important in diagnosing the causative organism, along with the use of other diagnostic tools like polymerase chain reaction and in vivo confocal microscopy.24
Many organisms cannot penetrate an intact corneal epithelium and are unable to progress to infectious keratitis without a pathway. However, there are a few organisms that can penetrate an intact corneal epithelium, resulting in infectious keratitis: Neisseria meningitides, N. gonorrhea, Corynebacterium diphtheria, Haemophilus influenzae and Listeria species.26
A wide range of pathogens can cause infectious keratitis, and each has different mechanisms by which they activate the host’s immune system. The key players of the corneal innate immune response are toll-like receptors (TLRs) and nucleotide-binding oligomerization domains (NOD)-like receptors, which recognize pathogen-associated molecular patterns on fungi, protozoa, bacteria and viruses as part of the innate immune system; this leads to the activation of inflammatory pathways via cytokines and chemokines.27,28
Macrophages and dendritic cells express pattern-recognition receptors such as TLRs and NOD-like receptors.28 These receptors play a protective role in the corneal innate immune response by detecting pathogens and triggering innate immunity and inflammation to protect the cornea from damage by infection. However, these receptors also play a destructive role when the innate immune response goes too far, resulting in excess inflammation, which can cause tissue destruction, corneal damage, dry eye disease and blindness.27,28
Infectious keratitis uses the adaptive immune system, which consists of B-lymphocytes and T-lymphocytes. Each of these cells carries a large repertoire of structurally unique antigen receptors; once activated, they trigger activation and proliferation of the B-cell or T-cell. This mechanism is called clonal selection and is the basis of the adaptive immune system. Adaptive immune responses develop more slowly than innate immune responses, taking three to five days to develop.
Dendritic cells are antigen-presenting cells that can induce primary immune responses in infection as well as acquired immune responses. The healthy cornea was previously believed to be an immune-privileged tissue lacking dendritic cells, but it is now known to contain them at various maturity levels (based on where they reside in the tissue). In infectious keratitis, immature dendritic cells capture antigens and microbial molecules in peripheral tissues and travel to nearby lymph nodes, where they mature. Mature dendritic cells can produce cytokines, upregulate co-stimulatory molecules and more effectively activate T-cells as a consequence of their TLRs being activated and the upregulated expression of molecules that impart efficient antigen presentation, T-cell priming and co-stimulation of T-cells.29 In summary, the adaptive immune system uses mature dendritic cells to activate T-cells and make them work better against infection.
Remember that when it comes to infectious keratitis, location matters. The infection involves both innate and adaptive immunity. For example, in central corneal infection, one way to bring more immune cells to the center of the cornea to fight infection is via conjunctival flaps. Although innate and adaptive immunity are robust, as previously mentioned, central corneal infection lacks the peripheral limbal resources.1
Conjunctival flaps in central corneal infection help bring the immune response from the periphery to the center of the cornea. Conjunctival flaps are especially useful in infectious keratitis refractory to antimicrobial therapy. These flaps bring blood and lymphatic vessels to the affected area of the central cornea and supply various growth factors, anti-inflammatory cells, cytokines and nutrients to the affected corneal tissue. Additionally, the conjunctival flap provides a physical barrier that may limit exposure of the corneal ulcer to proteolytic enzymes and proinflammatory mediators. One study found that out of nine patients receiving complete flaps, the rate of surgical success—the resolution of the refractive infectious keratitis after flap surgery—was 56%, and out of 12 patients receiving partial flaps, the surgical success rate was 66.7%.30
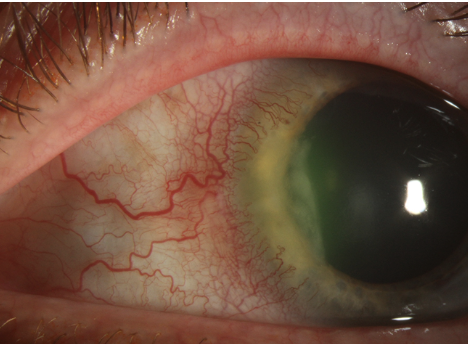
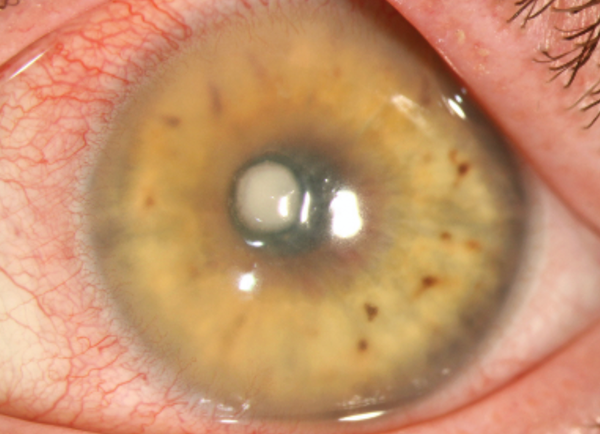
Peripheral ulcerative keratitis, a noninfectious form of keratitis, causes severe inflammation, progressive thinning and crescent-shaped ulcerations, often yellowish or gray, at the stromal layer. Photo: Christine Sindt, OD. Click image to enlarge.
Immune system presence (via blood vessels in conjunctival flaps) is important in helping heal refractory infectious keratitis as it brings peripheral resources to the central cornea. However, in noninfectious and autoimmune keratitis, there is overactivation of immune cells in the periphery.21
Noninfectious keratitis. Causes of noninfectious keratitis include autoimmune reactions, self-antigens and immune complex deposition.21 An example of immune complex deposition is the type III hypersensitivity reaction, which arises when antigen-antibody complexes develop in the bloodstream and are insufficiently removed, resulting in tissue deposition.31 This immune complex deposition activates the classical complement pathway, leading to the production of various chemotactic factors that recruit inflammatory cells such as neutrophils.
Peripheral ulcerative keratitis (PUK) pathogenesis is mediated by cellular and humoral immune pathways. It involves autoimmune reactions to corneal antigens, immune complex deposition and hypersensitivity reactions to foreign antigens. The classical complement pathway is activated due to immune complex deposition in the limbic vessels. This pathway leads to recruitment of inflammatory cells (plasma cells, neutrophils, mast cells, eosinophils) to the peripheral cornea and proinflammatory cytokine release, which promotes production and release of matrix metalloproteinases (MMPs). These enzymes can pathologically disrupt the extracellular matrix and type IV collagen (in basement membranes) but normally participate in tissue remodeling.
The proinflammatory cytokines can activate keratocytes, often causing them to release more cytokines such as neutrophil calgranulin C. An autoimmune response consisting of antibody- and cell-mediated hyperimmune reaction occurs in response to neutrophil calgranulin C in the peripheral cornea.21 Overall, the buildup of immune complexes in the limbic vessels triggers the classical complement system, releases enzymes that break down the extracellular matrix and activates an autoimmune response in the peripheral cornea.
Another autoimmune mechanism involves the failure of the blood-ocular barriers, which can lead to compromise of the eye’s immune privilege, allowing self-antigens to enter. Additionally, the eye regulates the immune system via a downregulatory immune environment to avoid permanent tissue damage. Regulatory T-cells help suppress any excessive immune activity. When these mechanisms fail, autoimmune disease can occur, and the immune system can be activated towards self-antigens.32 Autoimmune syndromes can cause visual morbidity. In uncontrolled autoimmune disease, sterile corneal ulcers are a common finding.33 Immune reaction to self-antigens results in the body attacking itself and damaging the cornea, possibly resulting in ulcerations.
Mooren’s ulcer is an uncommon, severe, chronic, progressive, idiopathic and painful variant of autoimmune PUK. It is characterized by a high risk of corneal perforation and vision loss. There are many possible triggers of this autoimmune response, including hepatitis C virus, ocular trauma, surgery and parasitic infections, but the underlying pathogenesis is uncertain, so clinical diagnosis is based predominantly on exclusion. Diagnosis involves excluding diseases capable of causing PUK, such as rheumatoid arthritis, systemic lupus erythematosus, malignancy and infections. Mooren’s ulcer is characterized by the absence of systemic autoimmune diseases.34 However, systemic autoimmune diseases can also result in PUK.
Rheumatoid arthritis and systemic lupus erythematosus (systemic autoimmune diseases) can have ophthalmic manifestations, including PUK.35,36 The upregulated inflammatory cascade due to T-cells, B-cells and monocytes, as well as associated cytokines such as tumor necrosis factor and IL-6, can result in inflammatory disease in the eyes.35 These systemic autoimmune diseases may cause excess inflammation and tissue breakdown in the eyes, resulting in PUK, an autoimmune keratitis.
Autoimmune interstitial keratitis involves non-ulcerating inflammation of the corneal stroma, with additional cellular infiltration and neovascularization. The corneal stroma is thinned and opacified, worsening vision. The precise pathophysiology remains unclear and can differ based on etiology, but it is broadly believed to be a T-cell-mediated immune reaction to corneal stromal antigens.37
Thygeson superficial punctate keratitis (SPK) is described by periods of remission and exacerbation of gray-white opacities in the corneal epithelium (these are often bilateral but can be asymmetric). The precise etiology is still unknown, but it is likely autoimmune and not infectious. Langerhans cells (a type of dendritic cell in the corneal epithelium) are positively correlated with PSK exacerbations. An increase in Langerhans cells may serve a protective role in the cornea via mitigating the cornea’s inflammatory response, although the exact inflammatory triggers and relapses in PSK remain unclear.38 A study consisting of five patients with SPK found Langerhans cells present in the basal epithelial layer of the cornea in all patients.39
Shared Mechanisms of Infectious and Noninfectious Keratitis
Despite the range of molecular and cellular mediators involved in corneal immunity, infectious and noninfectious keratitis share common mechanisms. These include the activation and migration of local antigen-presenting cells such as Langerhans cells, increased expression of proinflammatory cytokines and increased chemokine production. These responses are nonantigen-specific inflammatory responses to both endogenous and exogenous agents.40
Infectious and noninfectious keratitis trigger corneal inflammatory cascades and proinflammatory cytokines, leading to corneal tissue damage and possible ulceration. Both infectious and non-infectious keratitis may demonstrate localized white infiltrates, inflammation of the underlying stroma, corneal thinning and stromal edema. Additionally, mucopurulent discharge and hypopyon may be present, which can lead to corneal scarring or perforation. Corneal opacity (clouding) is a complication of keratitis and can lead to blindness.41
Noninfectious keratitis can become infectious by either pathogenic or nonpathogenic microbes. Complications can occur that threaten vision. This can occur because damaged corneal tissue may become susceptible to microbes (pathogenic or nonpathogenic), resulting in infection and, consequently, infectious keratitis.42
Takeaways
The cornea consists of various layers that play distinct roles in protection. The cornea relies on various mechanisms to protect the eye, including the tear film and corneal immunity. The tear film consists of molecules that protect the eye from toxins and allergens. Corneal immunity consists of both innate and adaptive immune responses. These responses are vital in protecting the cornea in various types of keratitis.
Although infectious keratitis is caused by pathogenic invasion of the cornea and noninfectious keratitis is caused by autoimmune-mediated or inflammatory reactions, both can result in corneal damage and destruction. The mechanisms by which these diseases can cause corneal damage share many similarities. Recognizing the distinct etiologies of these diseases and their common mechanisms is vital to timely diagnosis and treatment and, ultimately, to preserve vision.
Dr. Shaw is a medical student at the University of South Florida (USF) Health Morsani College of Medicine in Tampa, Florida. Dr. Sherman is an associate professor of optometry at USF Health. They have no financial disclosures.