A month before Roo began his course of vitamin D3 drops, Kayan Khan from Sheffield had also been prescribed the same brand of supplements.
Kayan, who is now 13, was already living with several complex medical conditions including chronic kidney disease, and the eight-week course of vitamin D prescribed by his GP was in line with recommendations.
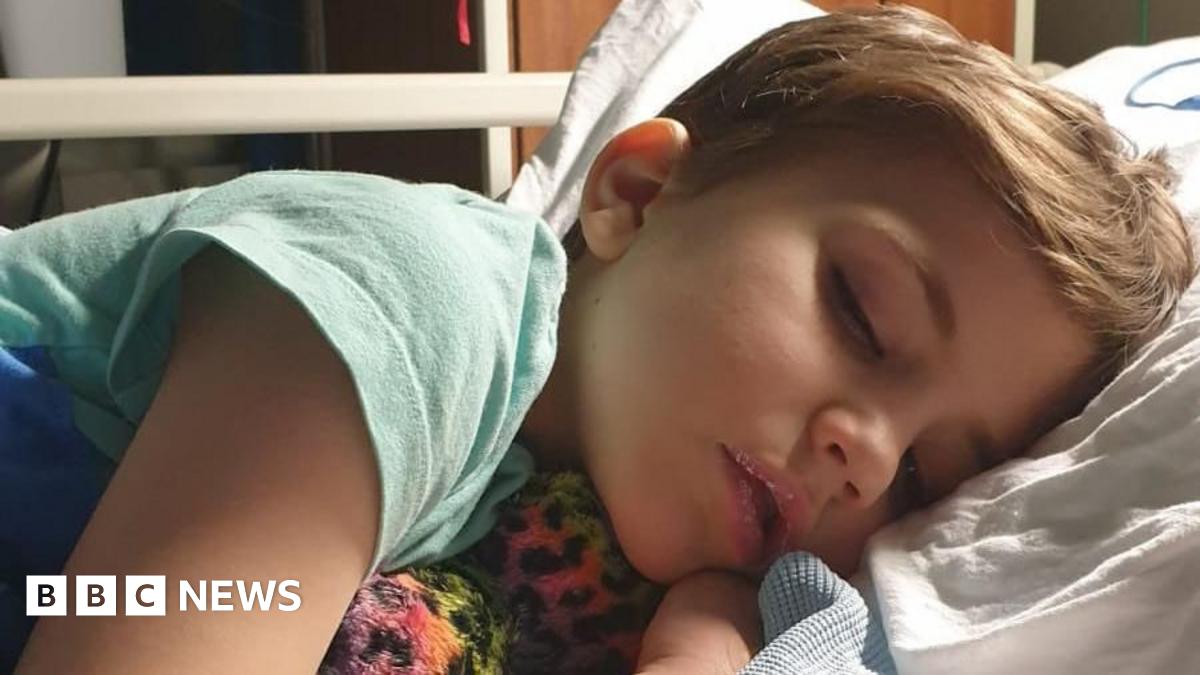
In February 2025, Kayan was admitted to hospital in Sheffield with high levels of calcium and reduced kidney function.
A letter to his mum Alaina from Sheffield Children’s NHS Foundation Trust said doctors had struggled to control his calcium levels and found he had vitamin D intoxication “far above expectation for standard supplementation”.
Alaina had not kept the bottle from his vitamin drops, so the exact batch could not be confirmed but the letter said that the “logical explanation” for his toxic level was a raised intake of vitamin D.
Once again, news of the FSA recall had not reached Alaina, although it had been passed on to hospital doctors and local pharmacies.
She said she only found out about the recall in April 2025.
Kayan suffered a significant deterioration in his kidney function, and although it has now recovered slightly, he may need a transplant in future.
“As a parent, as a mum, I’ve been let down,” she said.
“I can’t help but feel that for eight weeks, I’d been slowly poisoning my son.”
Dr Jeff Perring, executive medical director at Sheffield Children’s NHS Foundation Trust, said its standard practice was to ask GPs to prescribe medication, but that they followed strict procedures to inform patients and families about recalls of medication prescribed directly.
TriOn Pharma, the manufacturers of the drops, said that they had initiated a prompt recall and notified the FSA and the MHRA in line with requirements, after an issue was identified.
“Recall notifications were issued to all direct customers through established channels for onward communication across the supply chain,” they said.
“We continue to work with authorities to support patient safety.”