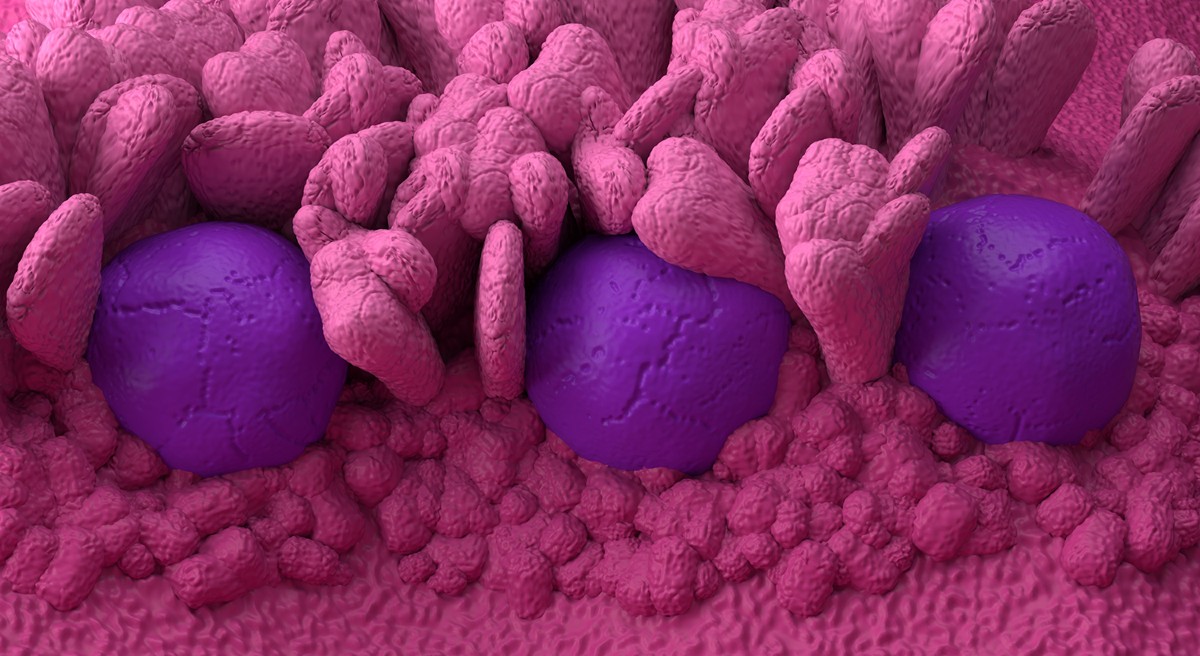
Illustration of Peyer’s patches in the wall of the small intestine. Peyer’s patches are part of the immune system and act as the first line of defence in this part of the gut. The oral cancer vaccine developed by Immunorock may activate the intestinal immune system via these patches. Credit: Nemes Laszlo/Science Photo Library/Getty
Cancer treatment usually conjures up images of major surgical operations, intravenous drips and intimidating radiotherapy machines. But what if a cancer therapy could be realized that is as straightforward as taking capsules at home? That’s the question being asked by researchers in Japan who are exploring the possibility of an orally administered therapeutic cancer treatment.
Immunotherapies are generating interest because they recruit the body’s immune system in the fight against cancer. They are mostly administered by intravenous drips. And while they have come a long way over the past decade, the responses rates remain fairly low, with only about 10–25% of patients responding to the most common form of immunotherapy — immune checkpoint inhibitors — for many cancers.
Researchers are seeking to build up the immunotherapy arsenal by developing alternative therapies that employ different strategies and are easier to administer. Scientists led by Toshiro Shirakawa — a professor in translational research for biologics at Kobe University’s Graduate School of Science, Technology and Innovation, and the CEO of research startup Immunorock — have been working on a series of preclinical studies and recently a clinical trial.
Immunity via the gut
In mouse and other lab work, Shirakawa’s team has been exploring a novel way of activating the immune system against cancer.
The approach would differ significantly from most conventional immunotherapies. Rather than being delivered by subcutaneous or intramuscular injection, which activates immune responses at the injection site, it would be taken orally as a capsule. It leverages the largest branch of the immune system in the body — that of the intestines.
Certain gut bacteria such as Bifidobacterium can activate the intestinal immune system via immune cells known as dendritic cells. Shirakawa and his team have been exploring the genetic modification of Bifidobacterium longum so that it displays an antigen protein for Wilms tumour 1 (WT1), a gene which often fuels tumour growth when it is overexpressed. This presentation of an antigen to dendritic cells in the intestines primes killer T cells to hunt and destroy cancer cells that produce the WT1 protein.
Since WT1 is frequently overexpressed across multiple cancer types, this approach could have potential as a method for targeting a wider range of cancers than existing immunotherapies.
Targeting WT1

Toshiro Shirakawa (right) conducting an experiment with a co-worker.
While other research groups have tried to develop approaches that target WT1, the results have generally been disappointing, either because the delivery system was not effective or because only a short portion of the antigen was used. Shirakawa’s teams say they have overcome both these problems and have demonstrated in mouse models that Bifidobacterium can deliver a full-length version of the WT1 antigen to the intestinal immune system1.
“Our strategy has the potential to transform the immunotherapy landscape for cancers that express WT1,” Shirakawa argues. “It represents a new modality in immunotherapy, combining the convenience of oral administration with the potential to overcome immunotherapy resistance.”
The approach has the potential to be used as a therapeutic vaccine, namely one administered after a cancer diagnosis. This is in contrast to prophylactic vaccines that protect against contracting a disease.
The biggest challenge was convincing others that the approach would work, Shirakawa says. “Initially, nobody believed that this oral cancer vaccine could really induce systemic immunity,” he says. “That’s why we needed to do many preclinical studies to show that it really does work.”
Seeking answers
The researchers found that the oral cancer vaccine can suppress tumour growth in mouse models of bladder cancer and prostrate cancer1,2. These early-stage results hint that a future possible treatment might be used alone or in combination with an immune-checkpoint inhibitor to treat cancers that originate in urothelial cells that line the urinary tract such as bladder cancer.
To determine the exact mechanism of the oral cancer vaccine, the team gave it to mice with cancerous tumours3. Mice treated with it exhibited a significant reduction in tumour size. They also had a higher proportion of active dendritic cells in their Peyer’s patches — specialized structures of lymphatic tissue in the small intestine — suggesting that this is the path to activating T cells via the intestinal immune system.
Finally, Shirakawa and his team found evidence that in mouse models with renal cell carcinoma the method has synergistic effects when combined with immune-checkpoint inhibitors4.
Tumours are described as cold when they are not responsive to immune-checkpoint inhibitors. “The vaccine can convert a cold tumour into a hot one,” Shirakawa says. “That’s a key finding.”
The researchers are now collaborating on a physician-led clinical trial to take the research further5.
Future outlook

An oral vaccine would be much easier to administer than other forms of immunotherapy. Credit: MJ_Prototype/iStock /Getty
The oral vaccine has several advantages over other forms of immunotherapy. “For a start, patients can self-administer the vaccine at home,” Shirakawa says. “There’s no need for them to go to a hospital to receive an injection.”
Another advantage is that these kinds of treatments would be cheap and easy to produce since they simply involve culturing bacteria and then freeze drying and encapsulating them. Since the vaccine doesn’t use live bacteria, cold storage isn’t needed, making transportation and storage easier.
The researchers further argue that using gut bacteria as vehicles to transport antigens offers a gentler way to activate the immune system than messenger RNA (mRNA) vaccines, such as those used to protect against COVID-19.
“Unlike mRNA vaccines, which can generate very high levels of antibodies, [this method] gently induces antigen-specific cellular immunity, which alters the microenvironments of tumour tissues and lymph nodes,” Shirakawa says.
Shirakawa and his team are now investigating the effect of using the oral vaccine in combination with an immune checkpoint inhibitor in patients with mesothelioma, a cancer that can be caused by exposure to asbestos. Immunorock is also in talks with a pharmaceutical company to develop a commercial oral vaccine, Shirakawa says.
In addition to targeting WT1-expressing cancers, the approach could potentially be expanded to different types of cancers and even to other diseases simply by attaching different antigens to Bifidobacterium.
“Using the gut immune system to induce cellular immunity is a unique approach,” Shirakawa notes. “And it should be very easy to extend it to other antigen proteins and also infectious diseases.”