Komaroff AL, Lipkin WI. ME/CFS and Long COVID share similar symptoms and biological abnormalities: road map to the literature. Front Med (Lausanne) 2023;10:1187163.
Al-Aly Z, Topol E. Solving the puzzle of long Covid. Science. 2024;383:830–2.
Mirin AA, Dimmock ME, Jason LA. Updated ME/CFS prevalence estimates reflecting post-COVID increases and associated economic costs and funding implications. Fatigue: Biomed Health Behav. 2022;10:83–93.
Reuken PA, Besteher B, Finke K, Fischer A, Holl A, Katzer K, et al. Longterm course of neuropsychological symptoms and ME/CFS after SARS-CoV-2-infection: a prospective registry study. Eur Arch Psychiatry Clin Neurosci. 2024;274:1903–10.
Ryabkova VA, Rubinskiy AV, Marchenko VN, Trofimov VI, Churilov LP. Similar patterns of dysautonomia in myalgic encephalomyelitis/chronic fatigue and post-COVID-19 syndromes. Pathophysiology. 2024;31:1–17.
Jamal A, Dalhuisen T, Gallego Márquez N, Dziarski AD, Uy J, Walch SN, et al. Post-SARS-CoV-2 Onset Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Symptoms in Two Cohort Studies of COVID-19 Recovery. medRxiv 2024.
Bonilla H, Quach TC, Tiwari A, Bonilla AE, Miglis M, Yang PC, et al. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome is common in post-acute sequelae of SARS-CoV-2 infection (PASC): Results from a post-COVID-19 multidisciplinary clinic. Front Neurol. 2023;14:1090747.
Tate W, Walker M, Sweetman E, Helliwell A, Peppercorn K, Edgar C, et al. Molecular Mechanisms of Neuroinflammation in ME/CFS and Long COVID to Sustain Disease and Promote Relapses. Front Neurol. 2022;13:877772.
Wirth KJ, Löhn M. Microvascular capillary and precapillary cardiovascular disturbances strongly interact to severely affect tissue perfusion and mitochondrial function in myalgic encephalomyelitis/chronic fatigue syndrome evolving from the post COVID-19 syndrome. Medicina. 2024;60:194.
Wirth KJ, Löhn M. Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and comorbidities: linked by vascular pathomechanisms and vasoactive mediators?. Medicina. 2023;59:978.
Kell DB, Pretorius E. The potential role of ischaemia–reperfusion injury in chronic, relapsing diseases such as rheumatoid arthritis, Long COVID, and ME/CFS: evidence, mechanisms, and therapeutic implications. Biochem J. 2022;479:1653–708.
Nunes M, Vlok M, Proal A, Kell DB, Pretorius E. Data-independent LC-MS/MS analysis of ME/CFS plasma reveals a dysregulated coagulation system, endothelial dysfunction, downregulation of complement machinery. Cardiovasc Diabetol. 2024;23:254.
Apostolou E, Rosén A. Epigenetic reprograming in myalgic encephalomyelitis/chronic fatigue syndrome: a narrative of latent viruses. J Intern Med. 2024;296:93–115.
Buonsenso D, Piazza M, Boner AL, Bellanti JA, editors. Long COVID: a proposed hypothesis-driven model of viral persistence for the pathophysiology of the syndrome. Allergy and Asthma Proceedings. OceanSide Publications; 2022.
Proal AD, VanElzakker MB, Aleman S, Bach K, Boribong BP, Buggert M, et al. SARS-CoV-2 reservoir in post-acute sequelae of COVID-19 (PASC). Nat Immunol. 2023;24:1616–27.
Bansal AS, Kraneveld AD, García EJ Oltra, Carding S. What causes ME/CFS: the role of the dysfunctional immune system and viral infections. J Immunol Allergy. 2022;3:1–15.
Wirth K, Scheibenbogen C. A unifying hypothesis of the pathophysiology of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): recognitions from the finding of autoantibodies against ß2-adrenergic receptors. Autoimmun Rev. 2020;19:102527.
Kell DB, Pretorius E. Are fibrinaloid microclots a cause of autoimmunity in Long Covid and other post-infection diseases?. Biochem J. 2023;480:1217–40.
Morris G, Anderson G, Maes M. Hypothalamic-pituitary-adrenal hypofunction in myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS) as a consequence of activated immune-inflammatory and oxidative and nitrosative pathways. Mol Neurobiol. 2017;54:6806–19.
Eaton-Fitch N, Rudd P, Er T, Hool L, Herrero L, Marshall-Gradisnik S. Immune exhaustion in ME/CFS and long COVID. JCI Insight 2024;9.
Saito S, Shahbaz S, Osman M, Redmond D, Bozorgmehr N, Rosychuk RJ, et al. Diverse immunological dysregulation, chronic inflammation, and impaired erythropoiesis in long COVID patients with chronic fatigue syndrome. J Autoimmun. 2024;147:103267.
Nunes JM, Kruger A, Proal A, Kell DB, Pretorius E. The occurrence of hyperactivated platelets and fibrinaloid microclots in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Pharmaceuticals. 2022;15:931.
Pretorius E, Venter C, Laubscher GJ, Kotze MJ, Oladejo SO, Watson LR, et al. Prevalence of symptoms, comorbidities, fibrin amyloid microclots and platelet pathology in individuals with long COVID/post-acute sequelae of COVID-19 (PASC). Cardiovasc Diabetol. 2022;21:148.
Dalton CF, de Oliveira MI, Stafford P, Peake N, Kane B, et al. Increased fibrinaloid microclot counts in platelet-poor plasma are associated with Long COVID. medRxiv:2024.04.04.24305318 [Preprint]. 2024.
Vu LT, Ahmed F, Zhu H, Iu DSH, Fogarty EA, Kwak Y, et al. Single-cell transcriptomics of the immune system in ME/CFS at baseline and following symptom provocation. Cell Rep Med. 2024;5:101373.
Giloteaux L, Glass KA, Germain A, Franconi CJ, Zhang S, Hanson MR. Dysregulation of extracellular vesicle protein cargo in female myalgic encephalomyelitis/chronic fatigue syndrome cases and sedentary controls in response to maximal exercise. J Extracell Vesicles. 2024;13:12403.
Aggarwal A, Singh TK, Pham M, Godwin M, Chen R, McIntyre TM, et al. Dysregulated platelet function in patients with postacute sequelae of COVID-19. Vasc Med. 2024;29:125–34.
Liu J, Li Y, Liu Q, Yao Q, Wang X, Zhang H, et al. SARS-CoV-2 cell tropism and multiorgan infection. Cell Discov. 2021;7:17.
Haffke M, Freitag H, Rudolf G, Seifert M, Doehner W, Scherbakov N, et al. Endothelial dysfunction and altered endothelial biomarkers in patients with post-COVID-19 syndrome and chronic fatigue syndrome (ME/CFS). J Transl Med. 2022;20:138.
Kuchler T, Günthner R, Ribeiro A, Hausinger R, Streese L, Wöhnl A, et al. Persistent endothelial dysfunction in post-COVID-19 syndrome and its associations with symptom severity and chronic inflammation. Angiogenesis. 2023;26:547–63.
Alfaro, Díaz-García E, García-Tovar E, Galera R S, Casitas R, Torres-Vargas M, et al. Endothelial dysfunction and persistent inflammation in severe post-COVID-19 patients: implications for gas exchange. BMC Med. 2024;22:242.
Wu X, Xiang M, Jing H, Wang C, Novakovic VA, Shi J. Damage to endothelial barriers and its contribution to long COVID. Angiogenesis. 2024;27:5–22.
Ståhlberg M, Fischer K, Tahhan M, Zhao A, Fedorowski A, Runold M, et al. Post-acute covid-19 syndrome: prevalence of peripheral microvascular endothelial dysfunction and associations with NT-ProBNP dynamics. Am J Med. 2025;138:1019−28.
Muys M, Demulder A, Besse-Hammer T, Ghorra N, Rozen L. Exploring hypercoagulability in post-COVID syndrome (PCS): an attempt at unraveling the endothelial dysfunction. J Clin Med. 2025;14:789.
Smadja DM, Günther S, Cavagna P, Renaud B, Salmon D, Hermann B, et al. Circulating endothelial cells: a key biomarker of persistent fatigue after hospitalization for COVID-19. Angiogenesis. 2024;28:8.
Thomas D, Noishiki C, Gaddam S, Wu D, Manhas A, Liu Y, et al. CCL2-mediated endothelial injury drives cardiac dysfunction in long COVID. Nat Cardiovasc Res. 2024;3:1249–65.
Perico L, Benigni A, Remuzzi G. SARS-CoV-2 and the spike protein in endotheliopathy. Trends Microbiol. 2024;32:53–67.
Scherbakov N, Szklarski M, Hartwig J, Sotzny F, Lorenz S, Meyer A, et al. Peripheral endothelial dysfunction in myalgic encephalomyelitis/chronic fatigue syndrome. ESC Heart Fail. 2020;7:1064–71.
Sørland K, Sandvik MK, Rekeland IG, Ribu L, Småstuen MC, Mella O, et al. Reduced endothelial function in myalgic encephalomyelitis/chronic fatigue syndrome-results from open-label cyclophosphamide intervention study. Front Med (Lausanne). 2021;8:642710.
Blauensteiner J, Bertinat R, León LE, Riederer M, Sepúlveda N, Westermeier F. Altered endothelial dysfunction-related miRs in plasma from ME/CFS patients. Sci Rep. 2021;11:10604.
Sandvik MK, Sørland K, Leirgul E, Rekeland IG, Stavland CS, Mella O, et al. Endothelial dysfunction in ME/CFS patients. PLoS ONE. 2023;18:e0280942.
McLaughlin M, Sanal-Hayes NEM, Hayes LD, Berry EC, Sculthorpe NF. People with long COVID and myalgic encephalomyelitis/chronic fatigue syndrome exhibit similarly impaired vascular function. Am J Med. 2025;138:560−6.
Cambras T, Zerón-Rugerio MF, Díez-Noguera A, Zaragozá MC, Domingo JC, Sanmartin-Sentañes R, et al. Skin temperature circadian rhythms and dysautonomia in myalgic encephalomyelitis/chronic fatigue syndrome: the role of endothelin-1 in the vascular tone dysregulation. Int J Mol Sci. 2023;24:4835.
Domingo JC, Battistini F, Cordobilla B, Zaragozá MC, Sanmartin-Sentañes R, Alegre-Martin J, et al. Association of circulating biomarkers with illness severity measures differentiates myalgic encephalomyelitis/chronic fatigue syndrome and post-COVID-19 condition: a prospective pilot cohort study. J Transl Med. 2024;22:343.
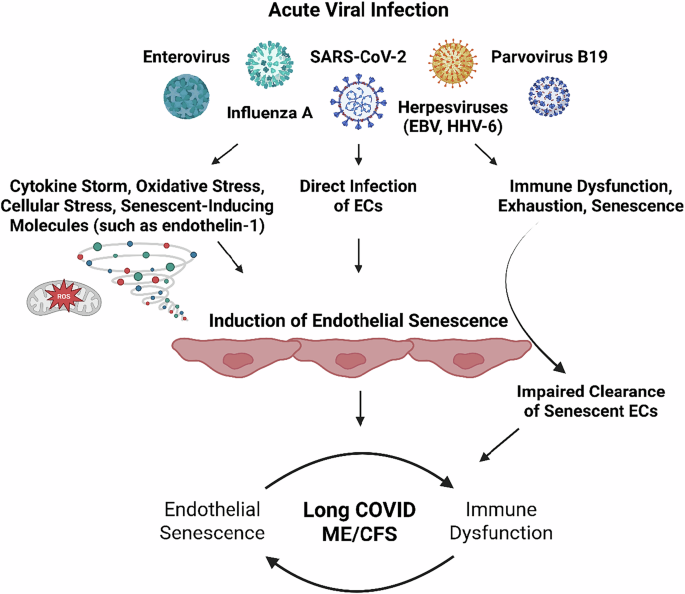
Sfera A, Osorio C, Zapata Martín Del Campo CM, Pereida S, Maurer S, Maldonado JC, et al. Endothelial senescence and chronic fatigue syndrome, a COVID-19 based hypothesis. Front Cell Neurosci. 2021;15:673217.
Nunes JM, Kell DB, Pretorius E. Herpesvirus infection of endothelial cells as a systemic pathological axis in myalgic encephalomyelitis/chronic fatigue syndrome. Viruses. 2024;16:572.
de Rooij LPMH, Becker LM, Carmeliet P. A role for the vascular endothelium in post–acute COVID-19?. Circulation. 2022;145:1503–5.
van Campen CMC, Verheugt FWA, Rowe PC, Visser FC. The cardiac output–cerebral blood flow relationship is abnormal in most myalgic encephalomyelitis/chronic fatigue syndrome patients with a normal heart rate and blood pressure response during a tilt test. Healthcare. 2024;12:2566.
van Campen CMC, Verheugt FWA, Rowe PC, Visser FC. Cerebral blood flow is reduced in ME/CFS during head-up tilt testing even in the absence of hypotension or tachycardia: a quantitative, controlled study using Doppler echography. Clin Neurophysiol Pr. 2020;5:50–8.
van Campen CMC, Rowe PC, Visser FC. Orthostatic symptoms and reductions in cerebral blood flow in long-haul COVID-19 patients: similarities with myalgic encephalomyelitis/chronic fatigue syndrome. Medicina. 2022;58:28.
Thapaliya K, Marshall-Gradisnik S, Barth M, Eaton-Fitch N, Barnden L. Brainstem volume changes in myalgic encephalomyelitis/chronic fatigue syndrome and long COVID patients. Front Neurosci. 2023;17:1125208.
Chien C, Heine J, Khalil A, Schlenker L, Hartung TJ, Boesl F, et al. Altered brain perfusion and oxygen levels relate to sleepiness and attention in post-COVID syndrome. Ann Clin Transl Neurol. 2024;11:2016–29.
Lee J-S, Sato W, Son C-G. Brain-regional characteristics and neuroinflammation in ME/CFS patients from neuroimaging: a systematic review and meta-analysis. Autoimmun Rev. 2024;23:103484.
Li X, Julin P, Li T-Q. Limbic perfusion is reduced in patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Tomography. 2021;7:675–87.
Pizzuto DA, Buonsenso D, Morello R, De Rose C, Valentini P, Fragano A, et al. Lung perfusion assessment in children with long-COVID: a pilot study. Pediatr Pulmonol. 2023;58:2059–67.
van Deursen JM. The role of senescent cells in ageing. Nature. 2014;509:439–46.
Harley CB, Futcher AB, Greider CW. Telomeres shorten during ageing of human fibroblasts. Nature. 1990;345:458–60.
Narita M, Young AR, Arakawa S, Samarajiwa SA, Nakashima T, Yoshida S, et al. Spatial coupling of mTOR and autophagy augments secretory phenotypes. Science. 2011;332:966–70.
Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell. 2005;120:513–22.
Passos JF, Nelson G, Wang C, Richter T, Simillion C, Proctor CJ, et al. Feedback between p21 and reactive oxygen production is necessary for cell senescence. Mol Syst Biol. 2010;6:347.
Stein GH, Drullinger LF, Soulard A, Dulić V. Differential roles for cyclin-dependent kinase inhibitors p21 and p16 in the mechanisms of senescence and differentiation in human fibroblasts. Mol Cell Biol. 1999;19:2109–17.
Rodier F, Muñoz DP, Teachenor R, Chu V, Le O, Bhaumik D, et al. DNA-SCARS: distinct nuclear structures that sustain damage-induced senescence growth arrest and inflammatory cytokine secretion. J Cell Sci. 2011;124:68–81.
Narita M, Nũnez S, Heard E, Narita M, Lin AW, Hearn SA, et al. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell. 2003;113:703–16.
Ivanov A, Pawlikowski J, Manoharan I, van Tuyn J, Nelson DM, Rai TS, et al. Lysosome-mediated processing of chromatin in senescence. J Cell Biol. 2013;202:129–43.
Miwa S, Kashyap S, Chini E, von Zglinicki T. Mitochondrial dysfunction in cell senescence and aging. J Clin Invest. 2022;132.
Coppé JP, Patil CK, Rodier F, Sun Y, Muñoz DP, Goldstein J, et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853–68.
Kurz DJ, Decary S, Hong Y, Erusalimsky JD. Senescence-associated (beta)-galactosidase reflects an increase in lysosomal mass during replicative ageing of human endothelial cells. J Cell Sci. 2000;113:3613–22.
Oguma Y, Alessio N, Aprile D, Dezawa M, Peluso G, Di Bernardo G, et al. Meta-analysis of senescent cell secretomes to identify common and specific features of the different senescent phenotypes: a tool for developing new senotherapeutics. Cell Commun Signal. 2023;21:262.
Ovadya Y, Landsberger T, Leins H, Vadai E, Gal H, Biran A, et al. Impaired immune surveillance accelerates accumulation of senescent cells and aging. Nat Commun. 2018;9:5435.
Pereira BI, Devine OP, Vukmanovic-Stejic M, Chambers ES, Subramanian P, Patel N, et al. Senescent cells evade immune clearance via HLA-E-mediated NK and CD8+ T cell inhibition. Nat Commun. 2019;10:2387.
Nelson G, Wordsworth J, Wang C, Jurk D, Lawless C, Martin-Ruiz C, et al. A senescent cell bystander effect: senescence-induced senescence. Aging Cell. 2012;11:345–9.
D’Agnillo F, Walters K-A, Xiao Y, Sheng Z-M, Scherler K, Park J, et al. Lung epithelial and endothelial damage, loss of tissue repair, inhibition of fibrinolysis, and cellular senescence in fatal COVID-19. Sci Transl Med. 2021;13:eabj7790.
Urata R, Ikeda K, Yamazaki E, Ueno D, Katayama A, Shin-Ya M, et al. Senescent endothelial cells are predisposed to SARS-CoV-2 infection and subsequent endothelial dysfunction. Sci Rep. 2022;12:11855.
Gioia U, Tavella S, Martínez-Orellana P, Cicio G, Colliva A, Ceccon M, et al. SARS-CoV-2 infection induces DNA damage, through CHK1 degradation and impaired 53BP1 recruitment, and cellular senescence. Nat Cell Biol. 2023;25:550–64.
Lee S, Yu Y, Trimpert J, Benthani F, Mairhofer M, Richter-Pechanska P, et al. Virus-induced senescence is a driver and therapeutic target in COVID-19. Nature. 2021;599:283–9.
Camell CD, Yousefzadeh MJ, Zhu Y, Prata L, Huggins MA, Pierson M, et al. Senolytics reduce coronavirus-related mortality in old mice. Science 2021;373.
Pastor-Fernández A, Bertos AR, Sierra-Ramírez A, del Moral-Salmoral J, Merino J, de Ávila AI, et al. Treatment with the senolytics dasatinib/quercetin reduces SARS-CoV-2-related mortality in mice. Aging Cell. 2023;22:e13771.
Bordoni V, Mariotti D, Matusali G, Colavita F, Cimini E, Ippolito G, et al. SARS-CoV-2 infection of airway epithelium triggers pulmonary endothelial cell activation and senescence associated with type I IFN production. Cells. 2022;11:2912.
Meyer K, Patra T, Vijayamahantesh, Ray R. SARS-CoV-2 spike protein induces paracrine senescence and leukocyte adhesion in endothelial cells. J Virol. 2021;95: https://doi.org/10.1128/jvi.00794-21.
Lipskaia L, Maisonnasse P, Fouillade C, Sencio V, Pascal Q, Flaman J-M, et al. Evidence that SARS-CoV-2 induces lung cell senescence: potential impact on COVID-19 lung disease. Am J Respir Cell Mol Biol. 2022;66:107–11.
Lekva T, Ueland T, Halvorsen B, Murphy SL, Dyrhol-Riise AM, Tveita A, et al. Markers of cellular senescence is associated with persistent pulmonary pathology after COVID-19 infection. Infect Dis (Lond). 2022;54:918–23.
Berentschot JC, Drexhage HA, Aynekulu Mersha DG, Wijkhuijs AJM, GeurtsvanKessel CH, Koopmans MPG, et al. Immunological profiling in long COVID: overall low grade inflammation and T-lymphocyte senescence and increased monocyte activation correlating with increasing fatigue severity. Front Immunol. 2023;14:1254899.
Lord JM, Veenith T, Sullivan J, Sharma-Oates A, Richter AG, Greening NJ, et al. Accelerated immune ageing is associated with COVID-19 disease severity. Immun Ageing. 2024;21:6.
Abdul Y, Karakaya E, Chandran R, Jamil S, Ergul A. Endothelin A receptors contribute to senescence of brain microvascular endothelial cells. Can J Physiol Pharm. 2022;100:1087–96.
El Habhab A, Altamimy R, Abbas M, Kassem M, Amoura L, Qureshi AW, et al. Significance of neutrophil microparticles in ischaemia-reperfusion: pro-inflammatory effectors of endothelial senescence and vascular dysfunction. J Cell Mol Med. 2020;24:7266–81.
Park S-H, Belcastro E, Hasan H, Matsushita K, Marchandot B, Abbas M, et al. Angiotensin II-induced upregulation of SGLT1 and 2 contributes to human microparticle-stimulated endothelial senescence and dysfunction: protective effect of gliflozins. Cardiovasc Diabetol. 2021;20:1–17.
Shang D, Liu H, Tu Z. Pro-inflammatory cytokines mediating senescence of vascular endothelial cells in atherosclerosis. Fundam Clin Pharm. 2023;37:928–36.
Sfera A, Osorio C, Jafri N, Diaz EL, Campo Maldonado JE. Intoxication with endogenous angiotensin II: a COVID-19 hypothesis. Front Immunol. 2020;11:1472.
Bloom SI, Islam MT, Lesniewski LA, Donato AJ. Mechanisms and consequences of endothelial cell senescence. Nat Rev Cardiol. 2023;20:38–51.
Wang P, Konja D, Singh S, Zhang B, Wang Y. Endothelial senescence: from macro- to micro-vasculature and its implications on cardiovascular health. Int J Mol Sci. 2024;25:1978.
Sabbatinelli J, Prattichizzo F, Olivieri F, Procopio AD, Rippo MR, Giuliani A. Where metabolism meets senescence: focus on endothelial cells. Front Physiol. 2019;10:1523.
Hasan H, Park S-H, Auger C, Belcastro E, Matsushita K, Marchandot B, et al. Thrombin induces angiotensin II-mediated senescence in atrial endothelial cells: impact on pro-remodeling patterns. J Clin Med. 2019;8:1570.
Cohen C, Le Goff O, Soysouvanh F, Vasseur F, Tanou M, Nguyen C, et al. Glomerular endothelial cell senescence drives age-related kidney disease through PAI-1. EMBO Mol Med. 2021;13:e14146.
Grillari J, Hohenwarter O, Grabherr RM, Katinger H. Subtractive hybridization of mRNA from early passage and senescent endothelial cells. Exp Gerontol. 2000;35:187–97.
Alavi P, Yousef Abdualla R, Brown D, Mojiri A, Nagendran J, Lewis J, et al. Aging is associated with organ-specific alterations in the level and expression pattern of von Willebrand factor. Arterioscler Thromb Vasc Biol. 2023;43:2183–96.
Matsushita H, Chang E, Glassford AJ, Cooke JP, Chiu C-P, Tsao PS. eNOS activity is reduced in senescent human endothelial cells. Circ Res. 2001;89:793–8.
Demaria M, Ohtani N, Youssef SA, Rodier F, Toussaint W, Mitchell JR, et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31:722–33.
Prattichizzo F, Giuliani A, Recchioni R, Bonafè M, Marcheselli F, De Carolis S, et al. Anti-TNF-α treatment modulates SASP and SASP-related microRNAs in endothelial cells and in circulating angiogenic cells. Oncotarget. 2016;7:11945–58.
Liao Y-L, Fang Y-F, Sun J-X, Dou G-R. Senescent endothelial cells: a potential target for diabetic retinopathy. Angiogenesis. 2024;27:663–79.
Zerón-Rugerio MF, Zaragozá MC, Domingo JC, Sanmartín-Sentañes R, Alegre-Martin J, Castro-Marrero J, et al. Sleep and circadian rhythm alterations in myalgic encephalomyelitis/chronic fatigue syndrome and post-COVID fatigue syndrome and its association with cardiovascular risk factors: a prospective cohort study. Chronobiol Int. 2024;41:1104–15.
Svitailo VS, Chemych MD. Indicators of blood coagulation function, concentration of endothelin-1 and Long-COVID. Bull Probl Biol Medi. 2024;3:180−6.
Philippe A, Günther S, Rancic J, Cavagna P, Renaud B, Gendron N, et al. VEGF-A plasma levels are associated with impaired DLCO and radiological sequelae in long COVID patients. Angiogenesis. 2024;27:51–66.
Kavyani B, Ahn SB, Missailidis D, Annesley SJ, Fisher PR, Schloeffel R, et al. Dysregulation of the kynurenine pathway, cytokine expression pattern, and proteomics profile link to symptomology in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Mol Neurobiol. 2024;61:3771–87.
Abraham GR, Kuc RE, Althage M, Greasley PJ, Ambery P, Maguire JJ, et al. Endothelin-1 is increased in the plasma of patients hospitalised with Covid-19. J Mol Cell Cardiol. 2022;167:92–6.
Turgunova L, Mekhantseva I, Laryushina Y, Alina A, Bacheva I, Zhumadilova Z, et al. The association of endothelin-1 with early and long-term mortality in COVID-19. J Pers Med. 2023;13:1558.
Farhangrazi ZS, Moghimi SM. Elevated circulating endothelin-1 as a potential biomarker for high-risk COVID-19 severity. Precis Nanomed. 2020;3:622–8.
Yokoi T, Fukuo K, Yasuda O, Hotta M, Miyazaki J, Takemura Y, et al. Apoptosis signal-regulating kinase 1 mediates cellular senescence induced by high glucose in endothelial cells. Diabetes. 2006;55:1660–5.
Wirth KJ, Scheibenbogen C, Paul F. An attempt to explain the neurological symptoms of myalgic encephalomyelitis/chronic fatigue syndrome. J Transl Med. 2021;19:471.
Wang Y, Yang Z, Zheng X, Liang X, Wu L, Wu C, et al. Cerebral blood flow alterations and host genetic association in individuals with long COVID: a transcriptomic-neuroimaging study. J Cereb Blood Flow Metab. 2025;45:431–42.
Kell DB, Laubscher GJ, Pretorius E. A central role for amyloid fibrin microclots in long COVID/PASC: origins and therapeutic implications. Biochem J. 2022;479:537–59.
Kim SY, Cheon J. Senescence-associated microvascular endothelial dysfunction: a focus on the blood-brain and blood-retinal barriers. Ageing Res Rev. 2024;100:102446.
Chala N, Moimas S, Giampietro C, Zhang X, Zambelli T, Exarchos V, et al. Mechanical fingerprint of senescence in endothelial cells. Nano Lett. 2021;21:4911–20.
Yamazaki Y, Baker DJ, Tachibana M, Liu C-C, van Deursen JM, Brott TG, et al. Vascular cell senescence contributes to blood–brain barrier breakdown. Stroke. 2016;47:1068–77.
Najari Beidokhti M, Villalba N, Ma Y, Reynolds A, Villamil JH, Yuan SY. Lung endothelial cell senescence impairs barrier function and promotes neutrophil adhesion and migration. Geroscience 2025;47:2655−71.
Donato AJ, Morgan RG, Walker AE, Lesniewski LA. Cellular and molecular biology of aging endothelial cells. J Mol Cell Cardiol. 2015;89:122–35.
Nunes JM, Kell DB, Pretorius E. Cardiovascular and haematological pathology in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a role for viruses. Blood Rev. 2023;60:101075.
Hira R, Baker JR, Siddiqui T, Patel A, Valani FGA, Lloyd MG, et al. Attenuated cardiac autonomic function in patients with long-COVID with impaired orthostatic hemodynamics. Clin Auton Res. 2025;35:301−14.
Ohno Y, Hashiguchi, Maenosono R T, Yamashita H, Taira Y, Minowa K, et al. The diagnostic value of endothelial function as a potential sensor of fatigue in health. Vasc Health Risk Manag. 2010;6:135–44.
Honda S, Ikeda K, Urata R, Yamazaki E, Emoto N, Matoba S. Cellular senescence promotes endothelial activation through epigenetic alteration, and consequently accelerates atherosclerosis. Sci Rep. 2021;11:14608.
Pantsulaia I, Ciszewski WM, Niewiarowska J. Senescent endothelial cells: Potential modulators of immunosenescence and ageing. Ageing Res Rev. 2016;29:13–25.
Müller L, Di Benedetto S. Inflammaging, immunosenescence, and cardiovascular aging: insights into long COVID implications. Front Cardiovasc Med. 2024;11:1384996.
Schmitt CA, Tchkonia T, Niedernhofer LJ, Robbins PD, Kirkland JL, Lee S. COVID-19 and cellular senescence. Nat Rev Immunol. 2023;23:251–63.
Pedroso RB, Torres L, Ventura LA, Camatta GC, Mota C, Mendes AC, et al. Rapid progression of CD8 and CD4 T cells to cellular exhaustion and senescence during SARS-CoV2 infection. J Leukoc Biol. 2024;116:1385–97.
De Biasi S, Meschiari M, Gibellini L, Bellinazzi C, Borella R, Fidanza L, et al. Marked T cell activation, senescence, exhaustion and skewing towards TH17 in patients with COVID-19 pneumonia. Nat Commun. 2020;11:3434.
Wiech M, Chroscicki P, Swatler J, Stepnik D, De Biasi S, Hampel M, et al. Remodeling of t cell dynamics during long COVID is dependent on severity of SARS-CoV-2 infection. Front Immunol. 2022;13:886431.
Van Campenhout J, Buntinx Y, Xiong H-Y, Wyns A, Polli A, Nijs J, et al. Unravelling the connection between energy metabolism and immune senescence/exhaustion in patients with myalgic encephalomyelitis/chronic fatigue syndrome. Biomolecules. 2025;15:357.
Arora S, Thompson PJ, Wang Y, Bhattacharyya A, Apostolopoulou H, Hatano R, et al. Invariant natural killer and T-cells coordinate removal of senescent cells. Med. 2021;2:938–50.e8.
Kale A, Sharma A, Stolzing A, Desprez P-Y, Campisi J. Role of immune cells in the removal of deleterious senescent cells. Immun Ageing. 2020;17:16.
Eaton-Fitch N, du Preez S, Cabanas H, Staines D, Marshall-Gradisnik S. A systematic review of natural killer cells profile and cytotoxic function in myalgic encephalomyelitis/chronic fatigue syndrome. Syst Rev. 2019;8:279.
Baraniuk JN, Eaton-Fitch N, Marshall-Gradisnik S. Meta-analysis of natural killer cell cytotoxicity in myalgic encephalomyelitis/chronic fatigue syndrome. Front Immunol. 2024;15:1440643.
Thierry AR. NETosis creates a link between diabetes and Long COVID. Physiol Rev. 2024;104:651–4.
Shafqat A, Khan S, Omer MH, Niaz M, Albalkhi I, AlKattan K, et al. Cellular senescence in brain aging and cognitive decline. Front Aging Neurosci. 2023;15:1281581.
Binet F, Cagnone G, Crespo-Garcia S, Hata M, Neault M, Dejda A, et al. Neutrophil extracellular traps target senescent vasculature for tissue remodeling in retinopathy. Science. 2020;369:eaay5356.
Thierry AR, Usher T, Sanchez C, Turner S, Venter C, Pastor B, et al. Circulating microclots are structurally associated with neutrophil extracellular traps and their amounts are elevated in long COVID patients. J Med Virol. 2025;97:e70613.
Curriu M, Carrillo J, Massanella M, Rigau J, Alegre J, Puig J, et al. Screening NK-, B- and T-cell phenotype and function in patients suffering from Chronic Fatigue Syndrome. J Transl Med. 2013;11:68.
Maya J. Surveying the metabolic and dysfunctional profiles of T cells and NK cells in myalgic encephalomyelitis/chronic fatigue syndrome. Int J Mol Sci. 2023;24:11937.
Arese P, Turrini F, Schwarzer E. Band 3/complement-mediated recognition and removal of normally senescent and pathological human erythrocytes. Cell Physiol Biochem. 2005;16:133–46.
Lutz L, Rohrhofer J, Zehetmayer S, Stingl M, Untersmayr E. Evaluation of immune dysregulation in an Austrian patient cohort suffering from myalgic encephalomyelitis/chronic fatigue syndrome. Biomolecules. 2021;11:1359.
Baillie K, Davies HE, Keat SBK, Ladell K, Miners KL, Jones SA, et al. Complement dysregulation is a prevalent and therapeutically amenable feature of long COVID. Med. 2024;5:239–53.e5.
Yu S, Cheng Y, Li B, Xue J, Yin Y, Gao J, et al. M1 macrophages accelerate renal glomerular endothelial cell senescence through reactive oxygen species accumulation in streptozotocin-induced diabetic mice. Int Immunopharmacol. 2020;81:106294.
Gao C, Xie R, Li W, Zhou J, Liu S, Cao F, et al. Endothelial cell phagocytosis of senescent neutrophils decreases procoagulant activity. Thromb Haemost. 2013;109:1079–90.
Altmann DM, Whettlock EM, Liu S, Arachchillage DJ, Boyton RJ. The immunology of long COVID. Nat Rev Immunol. 2023;23:618–34.
Riederer I, Sievert W, Eissner G, Molls M, Multhoff G. Irradiation-induced up-regulation of HLA-E on macrovascular endothelial cells confers protection against killing by activated natural killer cells. PLoS ONE. 2010;5:e15339.
Knopp RC, Erickson MA, Rhea EM, Reed MJ, Banks WA. Cellular senescence and the blood-brain barrier: implications for aging and age-related diseases. Exp Biol Med (Maywood). 2023;248:399–411.
Sakamuri SSVP, Sure VN, Kolli L, Liu N, Evans WR, Sperling JA, et al. Glycolytic and oxidative phosphorylation defects precede the development of senescence in primary human brain microvascular endothelial cells. GeroScience. 2022;44:1975–94.
Phoenix A, Chandran R, Ergul A. Cerebral microvascular senescence and inflammation in diabetes. Front Physiol. 2022;13:864758.
Cantuti-Castelvetri L, Ojha R, Pedro LD, Djannatian M, Franz J, Kuivanen S, et al. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science. 2020;370:856–60.
Stein SR, Ramelli SC, Grazioli A, Chung J-Y, Singh M, Yinda CK, et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature. 2022;612:758–63.
Casiraghi C, Dorovini-Zis K, Horwitz MS. Epstein-Barr virus infection of human brain microvessel endothelial cells: a novel role in multiple sclerosis. J Neuroimmunol. 2011;230:173–7.
Kasimir F, Toomey D, Liu Z, Kaiping AC, Ariza ME, Prusty BK. Tissue specific signature of HHV-6 infection in ME/CFS. Front Mol Biosci. 2022;9:1044964.
Volle R, Archimbaud C, Couraud P-O, Romero IA, Weksler B, Mirand A, et al. Differential permissivity of human cerebrovascular endothelial cells to enterovirus infection and specificities of serotype EV-A71 in crossing an in vitro model of the human blood–brain barrier. J Gen Virol. 2015;96:1682–95.
Zhu Y, Wang X, He Z, Zhao P, Ren H, Qi Z. Enterovirus 71 enters human brain microvascular endothelial cells through an ARF6-mediated endocytic pathway. J Med Virol. 2023;95:e28915.
Luo W, Zhong J, Zhao W, Liu J, Zhang R, Peng L, et al. Proteomic analysis of human brain microvascular endothelial cells reveals differential protein expression in response to enterovirus 71 infection. BioMed Res Int. 2015;2015:864169.
Higazy D, Lin X, Xie T, Wang K, Gao X, Cui M. Altered gene expression in human brain microvascular endothelial cells in response to the infection of influenza H1N1 virus. Anim Dis. 2022;2:25.
Lei Y, Sun Y, Wu W, Liu H, Wang X, Shu Y, et al. Influenza H7N9 virus disrupts the monolayer human brain microvascular endothelial cells barrier in vitro. Virol J. 2023;20:219.
Budamagunta V, Kumar A, Rani A, Bean L, Manohar-Sindhu S, Yang Y, et al. Effect of peripheral cellular senescence on brain aging and cognitive decline. Aging Cell. 2023;22:e13817.
Pushpam M, Talukdar A, Anilkumar S, Maurya SK, Issac TG, Diwakar L. Recurrent endothelin-1 mediated vascular insult leads to cognitive impairment protected by trophic factor pleiotrophin. Exp Neurol. 2024;381:114938.
Greene C, Connolly R, Brennan D, Laffan A, O’Keeffe E, Zaporojan L, et al. Blood–brain barrier disruption and sustained systemic inflammation in individuals with long COVID-associated cognitive impairment. Nat Neurosci. 2024;27:421–32.
Chaganti J, Poudel G, Cysique LA, Dore GJ, Kelleher A, Matthews G, et al. Blood brain barrier disruption and glutamatergic excitotoxicity in post-acute sequelae of SARS COV-2 infection cognitive impairment: potential biomarkers and a window into pathogenesis. Front Neurol. 2024;15:1350848.
Renz-Polster H, Tremblay ME, Bienzle D, Fischer JE. The Pathobiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: The Case for Neuroglial Failure. Front Cell Neurosci. 2022;16:888232.
Shan ZY, Barnden LR, Kwiatek RA, Bhuta S, Hermens DF, Lagopoulos J. Neuroimaging characteristics of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a systematic review. J Transl Med. 2020;18:335.
Ashktorab H, Challa SR, Singh G, Nanduri S, Ibrahim M, Martirosyan Z, et al. Gastrointestinal manifestations and their association with neurologic and sleep problems in long COVID-19 minority patients: a prospective follow-up study. Digest Dis Sci. 2024;69:562–9.
Steinsvik EK, Hausken T, Fluge Ø, Mella O, Gilja OH. Gastric dysmotility and gastrointestinal symptoms in myalgic encephalomyelitis/chronic fatigue syndrome. Scand J Gastroenterol. 2023;58:718–25.
Guo C, Che X, Briese T, Ranjan A, Allicock O, Yates RA, et al. Deficient butyrate-producing capacity in the gut microbiome is associated with bacterial network disturbances and fatigue symptoms in ME/CFS. Cell Host Microbe. 2023;31:288–304.e8.
Su Q, Lau RI, Liu Q, Li MKT, Yan Mak JW, Lu W, et al. The gut microbiome associates with phenotypic manifestations of post-acute COVID-19 syndrome. Cell Host Microbe. 2024;32:651–60.e4.
Martín F, Blanco-Suárez M, Zambrano P, Cáceres O, Almirall M, Alegre-Martín J, et al. Increased gut permeability and bacterial translocation are associated with fibromyalgia and myalgic encephalomyelitis/chronic fatigue syndrome: implications for disease-related biomarker discovery. Front Immunol. 2023;14:1253121.
Mouchati C, Durieux JC, Zisis SN, Labbato D, Rodgers MA, Ailstock K, et al. Increase in gut permeability and oxidized ldl is associated with post-acute sequelae of SARS-CoV-2. Front Immunol. 2023;14:1182544.
Stallmach A, Quickert S, Puta C, Reuken PA. The gastrointestinal microbiota in the development of ME/CFS: a critical view and potential perspectives. Front Immunol. 2024;15:1352744.
Zollner A, Meyer M, Jukic A, Adolph T, Tilg H. The intestine in acute and long COVID: pathophysiological insights and key lessons. Yale J Biol Med. 2024;97:447–62.
Ke Y, Li D, Zhao M, Liu C, Liu J, Zeng A, et al. Gut flora-dependent metabolite Trimethylamine-N-oxide accelerates endothelial cell senescence and vascular aging through oxidative stress. Free Radic Biol Med. 2018;116:88–100.
Saeedi Saravi SS, Pugin B, Constancias F, Shabanian K, Spalinger M, Thomas A, et al. Gut microbiota-dependent increase in phenylacetic acid induces endothelial cell senescence during aging. Nat Aging. 2025;5:1025−45.
Poole DC, Behnke BJ, Musch TI. The role of vascular function on exercise capacity in health and disease. J Physiol. 2021;599:889–910.
Korthuis RJ. Integrated Systems Physiology: from Molecule to Function to Disease. Skeletal Muscle Circulation. 2011. Morgan & Claypool Life Sciences
Joseph P, Arevalo C, Oliveira RKF, Faria-Urbina M, Felsenstein D, Oaklander AL, et al. Insights from invasive cardiopulmonary exercise testing of patients with myalgic encephalomyelitis/chronic fatigue syndrome. CHEST. 2021;160:642–51.
Singh I, Joseph P, Heerdt PM, Cullinan M, Lutchmansingh DD, Gulati M, et al. Persistent exertional intolerance after COVID-19: insights from invasive cardiopulmonary exercise testing. Chest. 2022;161:54–63.
Bertinat R, Villalobos-Labra R, Hofmann L, Blauensteiner J, Sepúlveda N, Westermeier F. Decreased NO production in endothelial cells exposed to plasma from ME/CFS patients. Vasc Pharm. 2022;143:106953.
Barrett-O’Keefe Z, Ives SJ, Trinity JD, Morgan G, Rossman MJ, Donato AJ, et al. Taming the “sleeping giant”: the role of endothelin-1 in the regulation of skeletal muscle blood flow and arterial blood pressure during exercise. Am J Physiol Heart Circ Physiol. 2013;304:H162–9.
Rapoport RM, Merkus D. Endothelin-1 regulation of exercise-induced changes in flow: dynamic regulation of vascular tone. Front Pharmacol. 2017;8:517.
Wray DW, Nishiyama SK, Donato AJ, Sander M, Wagner PD, Richardson RS. Endothelin-1-mediated vasoconstriction at rest and during dynamic exercise in healthy humans. Am J Physiol-Heart Circ Physiol. 2007;293:H2550–H6.
Bevan GH, Jenkins T, Josephson R, Rajagopalan S, Al-Kindi SG. Endothelin-1 and peak oxygen consumption in patients with heart failure with preserved ejection fraction. Heart Lung. 2021;50:442–6.
Liu S-Y, Chen L-K, Jhong Y-T, Chen C-W, Hsiao L-E, Ku H-C, et al. Endothelin-1 impairs skeletal muscle myogenesis and development via ETB receptors and p38 MAPK signaling pathway. Clin Sci. 2024;138:711–23.
Tsai CH, Huang PJ, Lee IT, Chen CM, Wu MH. Endothelin-1-mediated miR-let-7g-5p triggers interlukin-6 and TNF-α to cause myopathy and chronic adipose inflammation in elderly patients with diabetes mellitus. Aging (Albany NY). 2022;14:3633–51.
Horinouchi T, Hoshi A, Harada T, Higa T, Karki S, Terada K, et al. Endothelin-1 suppresses insulin-stimulated Akt phosphorylation and glucose uptake via GPCR kinase 2 in skeletal muscle cells. Br J Pharm. 2016;173:1018–32.
Alcalde-Estévez E, Asenjo-Bueno A, Sosa P, Olmos G, Plaza P, Caballero-Mora M, et al. Endothelin-1 induces cellular senescence and fibrosis in cultured myoblasts. A potential mechanism of aging-related sarcopenia. Aging (Albany NY). 2020;12:11200–23.
Liu Z, Hollmann C, Kalanidhi S, Grothey A, Keating S, Mena-Palomo I, et al. Increased circulating fibronectin, depletion of natural IgM and heightened EBV, HSV-1 reactivation in ME/CFS and long COVID. medRxiv [Preprint]. 2023.
Aschman T, Wyler E, Baum O, Hentschel A, Rust R, Legler F, et al. Post-COVID exercise intolerance is associated with capillary alterations and immune dysregulations in skeletal muscles. Acta Neuropathol Commun. 2023;11:193.
Hejbøl EK, Harbo T, Agergaard J, Madsen LB, Pedersen TH, Østergaard LJ, et al. Myopathy as a cause of fatigue in long-term post-COVID-19 symptoms: Evidence of skeletal muscle histopathology. Eur J Neurol. 2022;29:2832–41.
Rahman FA, Krause MP. PAI-1, the plasminogen system, and skeletal muscle. Int J Mol Sci. 2020;21:7066.
Young LV, Morrison W, Campbell C, Moore EC, Arsenault MG, Dial AG, et al. Loss of dystrophin expression in skeletal muscle is associated with senescence of macrophages and endothelial cells. Am J Physiol-Cell Physiol. 2021;321:C94–C103.
Sugihara H, Teramoto N, Nakamura K, Shiga T, Shirakawa T, Matsuo M, et al. Cellular senescence-mediated exacerbation of Duchenne muscular dystrophy. Sci Rep. 2020;10:16385.
Berenjabad NJ, Nejati V, Rezaie J. Angiogenic ability of human endothelial cells was decreased following senescence induction with hydrogen peroxide: possible role of vegfr-2/akt-1 signaling pathway. BMC Mol Cell Biol. 2022;23:31.
Dinulovic I, Furrer R, Handschin C. Plasticity of the muscle stem cell microenvironment. Adv Exp Med Biol. 2017;1041:141–69.
Mierzejewski B, Ciemerych MA, Streminska W, Janczyk-Ilach K, Brzoska E. miRNA-126a plays important role in myoblast and endothelial cell interaction. Sci Rep. 2023;13:15046.
Keller B, Receno CN, Franconi CJ, Harenberg S, Stevens J, Mao X, et al. Cardiopulmonary and metabolic responses during a 2-day CPET in myalgic encephalomyelitis/chronic fatigue syndrome: translating reduced oxygen consumption to impairment status to treatment considerations. J Transl Med. 2024;22:627.
van Campen CMC, Visser FC. Comparing idiopathic chronic fatigue and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) in males: response to two-day cardiopulmonary exercise testing protocol. Healthcare. 2021;9:683.
Pretorius E, Mbotwe S, Bester J, Robinson CJ, Kell DB. Acute induction of anomalous and amyloidogenic blood clotting by molecular amplification of highly substoichiometric levels of bacterial lipopolysaccharide. J R Soc Interface 2016;13.
Pretorius E, Page MJ, Hendricks L, Nkosi NB, Benson SR, Kell DB. Both lipopolysaccharide and lipoteichoic acids potently induce anomalous fibrin amyloid formation: assessment with novel Amytracker™; stains†. J R Soc Interface. 2018;15:20170941.
Kell DB, Pretorius E. Proteins behaving badly. Substoichiometric molecular control and amplification of the initiation and nature of amyloid fibril formation: lessons from and for blood clotting. Prog Biophys Mol Biol. 2017;123:16–41.
Ryu JK, Sozmen EG, Dixit K, Montano M, Matsui Y, Liu Y, et al. SARS-CoV-2 spike protein induces abnormal inflammatory blood clots neutralized by fibrin immunotherapy. biorxiv:2021.10.12.464152 [Preprint]. 2021.
Bonilla H, Hampton D, Marques de Menezes EG, Montoya JG, Norris PJ. Comparative analysis of extracellular vesicles in patients with severe and mild myalgic encephalomyelitis/chronic fatigue syndrome. Front Immunol. 2022;13:841910.
Jahanbani F, Maynard RD, Sing JC, Jahanbani S, Perrino JJ, Spacek DV, et al. Phenotypic characteristics of peripheral immune cells of Myalgic encephalomyelitis/chronic fatigue syndrome via transmission electron microscopy: a pilot study. PLoS ONE. 2022;17:e0272703.
Bochenek ML, Bauer T, Gogiraju R, Nadir Y, Mann A, Schönfelder T, et al. The endothelial tumor suppressor p53 is essential for venous thrombus formation in aged mice. Blood Adv. 2018;2:1300–14.
Valenzuela CA, Quintanilla R, Olate-Briones A, Venturini W, Mancilla D, Cayo A, et al. SASP-dependent interactions between senescent cells and platelets modulate migration and invasion of cancer cells. Int J Mol Sci. 2019;20.
Venturini W, Olate-Briones A, Valenzuela C, Méndez D, Fuentes E, Cayo A, et al. Platelet activation is triggered by factors secreted by senescent endothelial hmec-1 cells in vitro. Int J Mol Sci. 2020;21.
Silva GC, Abbas M, Khemais-Benkhiat S, Burban M, Ribeiro TP, Toti F, et al. Replicative senescence promotes prothrombotic responses in endothelial cells: role of NADPH oxidase- and cyclooxygenase-derived oxidative stress. Exp Gerontol. 2017;93:7–15.
Abbas M, Jesel L, Auger C, Amoura L, Messas N, Manin G, et al. Endothelial microparticles from acute coronary syndrome patients induce premature coronary artery endothelial cell aging and thrombogenicity. Circulation. 2017;135:280–96.
Bochenek ML, Schütz E, Schäfer K. Endothelial cell senescence and thrombosis: ageing clots. Thromb Res. 2016;147:36–45.
Mas-Bargues C, Borrás C, Alique M. The contribution of extracellular vesicles from senescent endothelial and vascular smooth muscle cells to vascular calcification. Front Cardiovasc Med. 2022;9:854726.
Kruger A, Vlok M, Turner S, Venter C, Laubscher GJ, Kell DB, et al. Proteomics of fibrin amyloid microclots in long COVID/post-acute sequelae of COVID-19 (PASC) shows many entrapped pro-inflammatory molecules that may also contribute to a failed fibrinolytic system. Cardiovasc Diabetol. 2022;21:190.
Rubin O, Crettaz D, Tissot JD, Lion N. Microparticles in stored red blood cells: submicron clotting bombs?. Blood Transfus. 2010;8:s31–8.
Schuermans S, Kestens C, Marques PE. Systemic mechanisms of necrotic cell debris clearance. Cell Death Dis. 2024;15:557.
Walton CC, Begelman D, Nguyen W, Andersen JK. Senescence as an amyloid cascade: the amyloid senescence hypothesis. Front Cell Neurosci. 2020;14:129.
Li R, Li Y, Zuo H, Pei G, Huang S, Hou Y. Alzheimer’s amyloid-β accelerates cell senescence and suppresses SIRT1 in human neural stem cells. Biomolecules. 2024;14:189.
Grobbelaar LM, Venter C, Vlok M, Ngoepe M, Laubscher GJ, Lourens PJ, et al. SARS-CoV-2 spike protein S1 induces fibrin(ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19. Biosci Rep. 2021;41.
Lopez-Vilchez I, Escolar G, Diaz-Ricart M, Fuste B, Galan AM, White JG. Tissue factor-enriched vesicles are taken up by platelets and induce platelet aggregation in the presence of factor VIIa. Thromb Haemost. 2007;97:202–11.
Pricoco R, Meidel P, Hofberger T, Zietemann H, Mueller Y, Wiehler K, et al. One-year follow-up of young people with ME/CFS following infectious mononucleosis by Epstein-Barr virus. Front Pediatr. 2023;11:1266738.
Tabachnikova A, Klein J, Wood J, Jaycox J, Dhodapkar R, Lu P, et al. The role of Epstein-Barr virus in long COVID. J Immunol. 2024;212:1121_6087–1121_6087.
Vojdani A, Almulla AF, Zhou B, Al-Hakeim HK, Maes M. Reactivation of herpesvirus type 6 and IgA/IgM-mediated responses to activin-A underpin long COVID, including affective symptoms and chronic fatigue syndrome. Acta Neuropsychiatr. 2024;36:172–84.
Chen Y-L, Chen Y-J, Tsai W-H, Ko Y-C, Chen J-Y, Lin S-F. The Epstein-Barr virus replication and transcription activator, Rta/BRLF1, induces cellular senescence in epithelial cells. Cell Cycle. 2009;8:58–65.
García-Fleitas J, García-Fernández A, Martí-Centelles V, Sancenón F, Bernardos A, Martínez-Máñez R. Chemical strategies for the detection and elimination of senescent cells. Acc Chem Res. 2024;57:1238–53.
Veroutis D, Argyropoulou OD, Goules AV, Kambas K, Palamidas DA, Evangelou K, et al. Senescent cells in giant cell arteritis display an inflammatory phenotype participating in tissue injury via IL-6-dependent pathways. Ann Rheum Dis. 2024;83:342–50.
Lagoumtzi SM, Chondrogianni N. Senolytics and senomorphics: natural and synthetic therapeutics in the treatment of aging and chronic diseases. Free Radic Biol Med. 2021;171:169–90.
Aguado J, Amarilla AA, Taherian Fard A, Albornoz EA, Tyshkovskiy A, Schwabenland M, et al. Senolytic therapy alleviates physiological human brain aging and COVID-19 neuropathology. Nat Aging. 2023;3:1561–75.
Villacampa A, Shamoon L, Valencia I, Morales C, Figueiras S, de la Cuesta F, et al. SARS-CoV-2 S protein reduces cytoprotective defenses and promotes human endothelial cell senescence. Aging Dis. 2024;16:1626–38.
Tripathi U, Nchioua R, Prata L, Zhu Y, Gerdes EOW, Giorgadze N, et al. SARS-CoV-2 causes senescence in human cells and exacerbates the senescence-associated secretory phenotype through TLR-3. Aging (Albany NY). 2021;13:21838–54.
Choi J-Y, Park JH, Jo C, Kim K-C, Koh YH. SARS-CoV-2 spike S1 subunit protein-mediated increase of beta-secretase 1 (BACE1) impairs human brain vessel cells. Biochem Biophys Res Commun. 2022;626:66–71.
Liu F, Han K, Blair R, Kenst K, Qin Z, Upcin B, et al. SARS-CoV-2 infects endothelial cells in vivo and in vitro. Front Cell Infect Microbiol. 2021;11:701278.
Yang R-C, Huang K, Zhang H-P, Li L, Zhang Y-F, Tan C, et al. SARS-CoV-2 productively infects human brain microvascular endothelial cells. J Neuroinflammation. 2022;19:149.
Caccuri F, Bugatti A, Zani A, De Palma A, Di Silvestre D, Manocha E, et al. SARS-CoV-2 infection remodels the phenotype and promotes angiogenesis of primary human lung endothelial cells. Microorganisms. 2021;9:1438.
Yamada S, Hashita T, Yanagida S, Sato H, Yasuhiko Y, Okabe K, et al. SARS-CoV-2 causes dysfunction in human iPSC-derived brain microvascular endothelial cells potentially by modulating the Wnt signaling pathway. Fluids Barriers CNS. 2024;21:32.
Hatch CJ, Piombo SD, Fang JS, Gach JS, Ewald ML, Van Trigt WK, et al. SARS-CoV-2 infection of endothelial cells, dependent on flow-induced ACE2 expression, drives hypercytokinemia in a vascularized microphysiological system. Front Cardiovasc Med. 2024;11:1360364.
Tsumita T, Takeda R, Maishi N, Hida Y, Sasaki M, Orba Y, et al. Viral uptake and pathophysiology of the lung endothelial cells in age-associated severe SARS-CoV-2 infection models. Aging Cell. 2024;23:e14050.
Jones K, Rivera C, Sgadari C, Franklin J, Max EE, Bhatia K, et al. Infection of human endothelial cells with Epstein-Barr virus. J Exp Med. 1995;182:1213–21.
Farina A, Rosato E, York M, Gewurz BE, Trojanowska M, Farina GA. Innate immune modulation induced by EBV lytic infection promotes endothelial cell inflammation and vascular injury in scleroderma. Front Immunol. 2021;12:651013.
Xiong A, Clarke-Katzenberg RH, Valenzuela G, Izumi KM, Millan MT. Epstein-Barr virus latent membrane protein 1 activates nuclear factor-kappa B in human endothelial cells and inhibits apoptosis. Transplantation. 2004;78:41–9.
Ban S, Goto Y, Kamada K, Takahama M, Watanabe H, Iwahori T, et al. Systemic granulomatous arteritis associated with Epstein-Barr virus infection. Virchows Arch. 1999;434:249–54.
Chen X-l, Huang Z-h, Huang X-h, Yao X, Pang K-l, He X-l, et al. Epstein-Barr virus (EBV) infection of endothelial cells via endocytosis is associated with a poor prognosis in nasopharyngeal carcinoma. Microbiol Spectr. 2025;0:e03427-24.
Caruso A, Rotola A, Comar M, Favilli F, Galvan M, Tosetti M, et al. HHV-6 infects human aortic and heart microvascular endothelial cells, increasing their ability to secrete proinflammatory chemokines. J Med Virol. 2002;67:528–33.
Caruso A, Favilli F, Rotola A, Comar M, Horejsh D, Alessandri G, et al. Human herpesvirus-6 modulates RANTES production in primary human endothelial cell cultures. J Med Virol. 2003;70:451–8.
Wu CA, Shanley JD. Chronic infection of human umbilical vein endothelial cells by human herpesvirus-6. J Gen Virol. 1998;79:1247–56.
Shioda S, Kasai F, Ozawa M, Hirayama N, Satoh M, Kameoka Y, et al. The human vascular endothelial cell line HUV-EC-C harbors the integrated HHV-6B genome which remains stable in long term culture. Cytotechnology. 2018;70:141–52.
Rotola A, Di Luca D, Cassai E, Ricotta D, Giulio A, Turano A, et al. Human herpesvirus 6 infects and replicates in aortic endothelium. J Clin Microbiol. 2000;38:3135–6.
Caruso A, Caselli E, Fiorentini S, Rotola A, Prandini A, Garrafa E, et al. U94 of human herpesvirus 6 inhibits in vitro angiogenesis and lymphangiogenesis. Proc Natl Acad Sci USA. 2009;106:20446–51.
Hiyoshi M, Indalao IL, Yano M, Yamane K, Takahashi E, Kido H. Influenza A virus infection of vascular endothelial cells induces GSK-3β-mediated β-catenin degradation in adherens junctions, with a resultant increase in membrane permeability. Arch Virol. 2015;160:225–34.
Bauer L, Rijsbergen LC, Leijten L, Benavides FF, Noack D, Lamers MM, et al. The pro-inflammatory response to influenza A virus infection is fueled by endothelial cells. Life Sci Alliance. 2023;6:e202201837.
Armstrong SM, Wang C, Tigdi J, Si X, Dumpit C, Charles S, et al. Influenza infects lung microvascular endothelium leading to microvascular leak: role of apoptosis and claudin-5. PLoS ONE. 2012;7:e47323.
Suo J, Zhao L, Wang J, Zhu Z, Zhang H, Gao R. Influenza virus aggravates the ox-LDL-induced apoptosis of human endothelial cells via promoting p53 signaling. J Med Virol. 2015;87:1113–23.
Sumikoshi M, Hashimoto K, Kawasaki Y, Sakuma H, Suzutani T, Suzuki H, et al. Human influenza virus infection and apoptosis induction in human vascular endothelial cells. J Med Virol. 2008;80:1072–8.
Han T, Lai Y, Jiang Y, Liu X, Li D. Influenza A virus infects pulmonary microvascular endothelial cells leading to microvascular leakage and release of pro-inflammatory cytokines. PeerJ. 2021;9:e11892.
Xia B, Chen H, Taleb SJ, Xi X, Shaheen N, Baoyinna B, et al. FBXL19 in endothelial cells protects the heart from influenza A infection by enhancing antiviral immunity and reducing cellular senescence programs. Am J Physiol-Heart Circ Physiol. 2024;327:H937–H46.
Lipskaia L, Delval L, Sencio V, Born E, Heumel S, Houssaini A, et al. Virus-induced cellular senescence causes pulmonary sequelae post-influenza infection. Aging Cell. 2025;n/a:e70140.
Saijets S, Ylipaasto P, Vaarala O, Hovi T, Roivainen M. Enterovirus infection and activation of human umbilical vein endothelial cells. J Med Virol. 2003;70:430–9.
Liang C-C, Sun M-J, Lei H-Y, Chen S-H, Yu C-K, Liu C-C, et al. Human endothelial cell activation and apoptosis induced by enterovirus 71 infection. J Med Virol. 2004;74:597–603.
Wen J, Huang C. Coxsackieviruses B3 infection of myocardial microvascular endothelial cells activates fractalkine via the ERK1/2 signaling pathway. Mol Med Rep. 2017;16:7548–52.
von Kietzell K, Pozzuto T, Heilbronn R, Grössl T, Fechner H, Weger S. Antibody-mediated enhancement of parvovirus B19 uptake into endothelial cells mediated by a receptor for complement factor C1q. J Virol. 2014;88:8102–15.
Pasquinelli G, Bonvicini F, Foroni L, Salfi N, Gallinella G. Placental endothelial cells can be productively infected by Parvovirus B19. J Clin Virol. 2009;44:33–8.