Infection during pregnancy causes almost immediate changes to placental cells called spongiotrophoblasts, according to a new study in mice. That change compromises maternal immune tolerance of the fetus and in turn sets off a cascade of inflammation that ultimately leads to behavioral differences in some male pups, the researchers found.
“The biggest surprise is just how stark the sex difference is,” says Elisa Zhang, assistant professor and Howard Hughes Medical Institute Freeman Hrabowski Scholar at the University of California, Davis, who was not involved in the study.
In people, infection during pregnancy increases a child’s likelihood of autism about threefold. The mechanisms are not completely understood but could involve the immune reaction to the infectious agent.
The new findings offer insight into how an immune response to infection affects development almost immediately.
“There’s a constellation of changes that occur at the maternal-fetal interface of male embryos that’s missing from females, and that’s likely giving rise to developmental deficits that are sex-specific,” says study investigator Lucas Cheadle, associate professor at Cold Spring Harbor Laboratory.
Cheadle and his colleagues injected pregnant mice with a compound called poly I:C, which mimics a viral infection and triggers an immune response, on embryonic day 12.5. Some but not all of the male pups, upon reaching adulthood, displayed behaviors associated with autism, such as decreased social preferences, increased repetitive behaviors and motor deficits. The female pups did not display these behaviors.
Administering poly I:C on embryonic day 14.5, when the placental barrier is more mature, did not lead to notable behavioral differences in any pups. That result suggests the placenta could hold clues as to what might be going awry.
“Usually, people in the field of neurodevelopmental disorders check what happens immediately post-birth. But the new aspect here is to look at what happens in the fetal stage, really during the process of development in utero,” says study investigator Irene Sanchez-Martin, a postdoctoral fellow in Cheadle’s lab. The team posted the study on bioRxiv earlier this month.
W
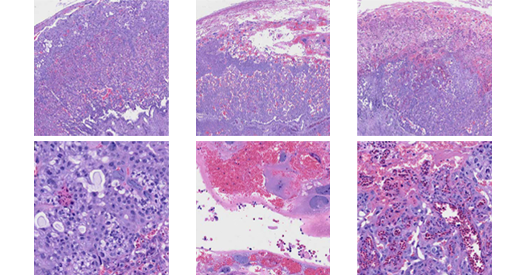
ithin 24 hours after immune activation, spongiotrophoblasts, which form the barrier between the maternal and fetal parts of the placenta, show a strong upregulation of inflammatory genes in males that go on to exhibit behavioral differences, the researchers found. “The findings were really striking,” Cheadle says. “If there was an immune conflict in the placenta, you might expect it to happen [in that barrier].”
They’re really nailing down a very specific structural phenomenon in a specific subset of placental cells, which I think is a level of resolution that’s remarkable.
—
—
Serena Banu Gumusoglu
The activation of inflammatory genes suggests that there is a loss of immunosuppression at the maternal-fetal interface, which normally protects the fetus from the maternal immune system, Cheadle says. The mice with behavioral differences had more spongiotrophoblasts, and these cells were not structurally organized in the right way, Cheadle adds.
“We’ve understood for a long time that maternal immune activation leads to sex-specific, male-specific vulnerabilities,” says Serena Banu Gumusoglu, assistant professor of obstetrics and gynecology and psychiatry at the University of Iowa, who did not contribute to the research. But the mechanistic understanding of how this effect occurs is new, Gumusoglu says. “They’re really nailing down a very specific structural phenomenon in a specific subset of placental cells, which I think is a level of resolution that’s remarkable.”
Affected male mice also showed rapid accumulation of immune cells and cytokines—particularly IL-6, which is associated with autism-related behaviors in mice—in the amniotic fluid, which bathes the developing fetus. None of the cytokines were increased in the amniotic fluid of females.
One likely reason for the sex difference in the risk of placental immune conflict is that the male fetus has antigens—for example, Y-chromosome-encoded antigens—that the female fetus does not, Cheadle says. Having more antigens that the mother’s immune system recognizes as foreign, in combination with the loss of immunosuppression at the maternal-fetal interface, might make the males more susceptible to the impact of maternal immune activation, the researchers suggest.
Not all male mice were equally affected, but those that were tended to be ones that, in the embryonic stage, were neighbored by other male embryos rather than female embryos, the researchers observed. Thus, a pup’s likelihood of behavioral differences was related not only to its sex but also that of neighboring fetuses in the womb.
“If we really want to understand autism and neurodevelopmental disorders on a comprehensive level, we need to make room for studies that are focused not only on brain-intrinsic drivers of autism spectrum disorder, but on other drivers as well,” Cheadle says. “Understanding autism from a variety of perspectives, whether it’s neurological, immunological, or even ‘placentological’—I think those are all important pieces of the puzzle.”