Scientists have discovered how tuberculosis bacteria stiffen immune cell membranes to survive inside the body, a breakthrough that could lead to the development of new TB treatments.

Researchers have discovered a sophisticated biophysical trick that allows tuberculosis-causing bacteria to survive inside human cells, which could lead to new treatments for the disease.
Tuberculosis, which kills more than a million people each year, continues to pose a major public health challenge, particularly in Asia, Africa and Latin America. The disease is caused by mycobacteria, microscopic organisms that have evolved strategies to hijack human immune cells and avoid destruction.
“Tuberculosis is rampant in India,” said Ayush Panda, formerly a graduate student in the laboratory of Mohammed Saleem at the National Institute of Science Education and Research, India. “I grew up in a state where tuberculosis outbreaks are a major problem, and I was always curious about how these diseases spread. That’s what drew me to this research.”
Vesicles that stiffen immune cells
The new study shows that mycobacteria release tiny packages known as extracellular vesicles. These vesicles fuse with the membranes of immune cells and contain specialised lipids.
Ordinarily, immune cells trap harmful bacteria in compartments called phagosomes, which then fuse with lysosomes.
Ordinarily, immune cells trap harmful bacteria in compartments called phagosomes, which then fuse with lysosomes. The researchers found that by stiffening the phagosome membrane, mycobacteria prevent this fusion, effectively building a protective bunker inside human cells.
“If the membrane becomes more rigid, it becomes much harder for the phagosome to fuse with the lysosome,” Panda said. “It’s an elegant biophysical mechanism: the bacteria remodel the membrane architecture to escape the very process that would have killed them.” The team also discovered that the vesicles are not confined to infected cells, they can also weaken nearby immune cells even before they encounter the bacteria.
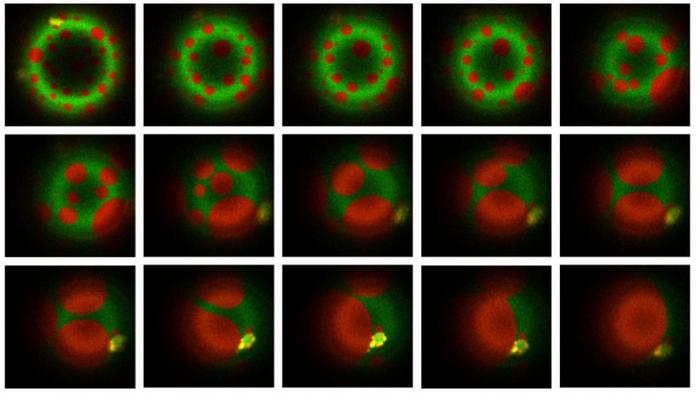

Mycobacteria have evolved sophisticated ways to hijack human immune cells and avoid being destroyed. Specifically, they stiffen the internal membrane to prevent the digestive enzymes within lysosomes from destroying the bacteria.
A lipid-centric approach
This discovery represents a new way of understanding mycobacterial survival. While previous research focused mainly on the proteins bacteria disrupt, this study highlights the crucial role of lipids. Simply introducing bacterial lipids into host cell membranes is enough to trigger immune dysfunction.
“The most surprising finding was when we introduced mycobacterial lipids into membranes that mimic the host phagosome, we saw remarkable physical changes – the membrane properties were completely altered,” Panda said.
Implications beyond tuberculosis
The researchers also observed similar membrane-stiffening effects mediated by extracellular vesicles in other pathogens, including Klebsiella pneumoniae and Staphylococcus aureus, suggesting this could be an evolutionarily conserved strategy.
Now that we understand how the bacteria protect themselves, we can start looking for ways to stop them.
The findings could have big implications for potential treatments, as scientists may be able to target the proteins involved in vesicle production or counteract the membrane-stiffening effects, allowing immune cells to function normally.
“Now that we understand how the bacteria protect themselves, we can start looking for ways to stop them,” Panda said. “If we can block the bacteria from stiffening those membranes, our immune cells might be able to do their job and stop the infection.”