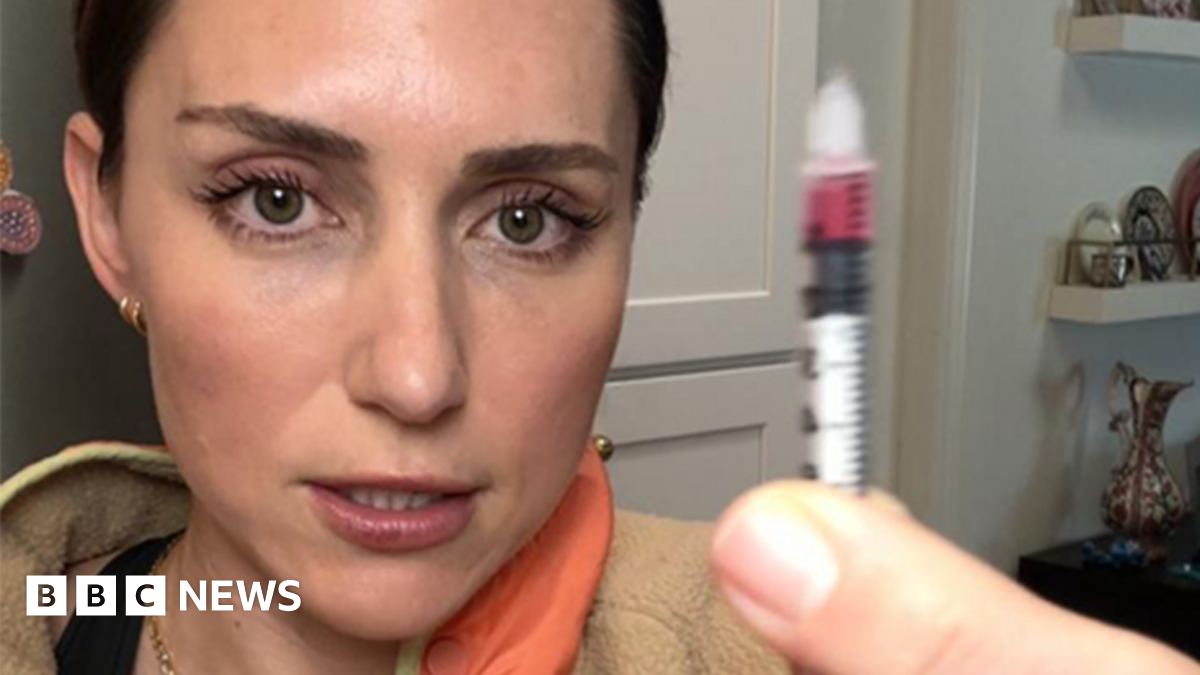
At this stage, using unregulated peptides isn’t “bio-hacking”, it’s a biological gamble, Dr Mrozinski warns. “If this ‘guinea pig’ culture spreads, we risk a public health crisis of ‘mystery’ chronic conditions caused by these unregulated peptides that the traditional medical system isn’t yet equipped to reverse.”
At the same time as millions of posts about peptides pop up across socials, a growing number of clinics are offering peptide therapy.
Dr Syed Omar Babar, an A&E consultant and the director of a private clinic in Leicester, offers peptide therapy – using unregulated peptides like BPC-157 and TB-500, among others. He believes this is a “golden era” for peptides and that they will play a huge part in the future of healthcare.
But why, I ask, are there no gold-standard trials on humans? If these peptides are so safe and effective, why aren’t they licensed medicines?
He says it’s down to funding – to get a product from animal studies to human trials and to a fully licensed medicine takes years and billions of dollars. It’s not in the interests of big pharmaceuticals, he explains, to fund this process.
“Many of the peptides we are talking about are completely natural – our bodies make them – therefore, making them difficult to patent.
“They have to be markedly different to their natural form – which is tricky with peptides.”
Without a patent, he says, companies risk pouring money into a product with little financial protection.
Dr Babar says the peptide therapies his clinic offers are overseen by a GMC-registered doctor but, because these products are not licensed and there is “no textbook” on how to use them, it comes down to experience and clinicians like himself learning “from each other”.
As a doctor, he says he can’t suggest these unregulated peptides are able to treat a certain condition. However, he says he can highlight to patients which products are available and, with their full consent and understanding that these peptides are not for human consumption, he is able to recommend peptide therapy.
In response to the explosion of the unregulated peptides market, the MHRA, the body responsible for licensing and overseeing the use of medicines in the UK, says that it does not “accept attempts to evade medicines regulation” by selling peptides for “research purposes” where evidence indicates they are being used by people.
It says it will remove products from the market if they claim to treat or change how the body works if they are not licensed under the Human Medicines Regulations Act 2012, and that it strongly advises against purchasing and using unauthorised medicinal products – “particularly those promoted on social media”.