Gaj, T., Sirk, S. J. & Barbas, C. F. Expanding the scope of site-specific recombinases for genetic and metabolic engineering. Biotechnol. Bioeng. 111, 1–15 (2014).
Olorunniji, F. J., Rosser, S. J. & Stark, W. M. Site-specific recombinases: molecular machines for the genetic revolution. Biochem. J. 473, 673–684 (2016).
Meinke, G., Bohm, A., Hauber, J., Pisabarro, M. T. & Buchholz, F. Cre recombinase and other tyrosine recombinases. Chem. Rev. 116, 12785–12820 (2016).
Jelicic, M. et al. Discovery and characterization of novel Cre-type tyrosine site-specific recombinases for advanced genome engineering. Nucleic Acids Res. 51, 5285–5297 (2023).
Smith, M. C. M., Brown, W. R. A., McEwan, A. R. & Rowley, P. A. Site-specific recombination by φC31 integrase and other large serine recombinases. Biochem. Soc. Trans. 38, 388–394 (2010).
Xu, Z. et al. Accuracy and efficiency define Bxb1 integrase as the best of fifteen candidate serine recombinases for the integration of DNA into the human genome. BMC Biotechnol. 13, 87 (2013).
Yarnall, M. T. N. et al. Drag-and-drop genome insertion of large sequences without double-strand DNA cleavage using CRISPR-directed integrases. Nat. Biotechnol. 41, 500–512 (2023).
Durrant, M. G. et al. Systematic discovery of recombinases for efficient integration of large DNA sequences into the human genome. Nat. Biotechnol. 41, 488–499 (2023).
Wu, S. C.-Y. et al. piggyBac is a flexible and highly active transposon as compared to Sleeping Beauty, Tol2, and Mos1 in mammalian cells. Proc. Natl Acad. Sci. USA 103, 15008–15013 (2006).
Yusa, K., Zhou, L., Li, M. A., Bradley, A. & Craig, N. L. A hyperactive piggyBac transposase for mammalian applications. Proc. Natl Acad. Sci. USA 108, 1531–1536 (2011).
Klompe, S. E., Vo, P. L. H., Halpin-Healy, T. S. & Sternberg, S. H. Transposon-encoded CRISPR–Cas systems direct RNA-guided DNA integration. Nature 571, 219–225 (2019).
Strecker, J. et al. RNA-guided DNA insertion with CRISPR-associated transposases. Science 365, 48–53 (2019).
Durrant, M. G. et al. Bridge RNAs direct programmable recombination of target and donor DNA. Nature 630, 984–993 (2024).
Siddiquee, R., Pong, C. H., Hall, R. M. & Ataide, S. F. A programmable seekRNA guides target selection by IS1111 and IS110 type insertion sequences. Nat. Commun. 15, 5235 (2024).
Mukhametzyanova, L. et al. Activation of recombinases at specific DNA loci by zinc-finger domain insertions. Nat. Biotechnol. https://doi.org/10.1038/s41587-023-02121-y (2024).
Fauser, F. et al. Systematic development of reprogrammed modular integrases enables precise genomic integration of large DNA Sequences. Preprint at bioRxiv https://doi.org/10.1101/2024.05.09.593242 (2024).
Merrick, C. A., Zhao, J. & Rosser, S. J. Serine integrases: advancing synthetic biology. ACS Synth. Biol. 7, 299–310 (2018).
Hornung, V. & Latz, E. Intracellular DNA recognition. Nat. Rev. Immunol. 10, 123–130 (2010).
Motwani, M., Pesiridis, S. & Fitzgerald, K. A. DNA sensing by the cGAS–STING pathway in health and disease. Nat. Rev. Genet. 20, 657–674 (2019).
Carpenter, S. & O’Neill, L. A. J. From periphery to center stage: 50 years of advancements in innate immunity. Cell 187, 2030–2051 (2024).
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z. J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013).
Wu, J. et al. Cyclic GMP-AMP is an endogenous second messenger in innate immune signaling by cytosolic DNA. Science 339, 826–830 (2013).
Neves, C., Escriou, V., Byk, G., Scherman, D. & Wils, P. Intracellular fate and nuclear targeting of plasmid DNA. Cell Biol. Toxicol. 15, 193–202 (1999).
Zabner, J., Fasbender, A. J., Moninger, T., Poellinger, K. A. & Welsh, M. J. Cellular and molecular barriers to gene transfer by a cationic lipid. J. Biol. Chem. 270, 18997–19007 (1995).
Wang, D., Tai, P. W. L. & Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat. Rev. Drug Discov. 18, 358–378 (2019).
Hanlon, K. S. et al. High levels of AAV vector integration into CRISPR-induced DNA breaks. Nat. Commun. 10, 4439 (2019).
Greig, J. A. et al. Integrated vector genomes may contribute to long-term expression in primate liver after AAV administration. Nat. Biotechnol. 42, 1232–1242 (2024).
Simpson, B. P., Yrigollen, C. M., Izda, A. & Davidson, B. L. Targeted long-read sequencing captures CRISPR editing and AAV integration outcomes in brain. Mol. Ther. 31, 760–773 (2023).
Ertl, H. C. J. Immunogenicity and toxicity of AAV gene therapy. Front. Immunol. 13, 975803 (2022).
Hinderer, C. et al. Severe toxicity in nonhuman primates and piglets following high-dose intravenous administration of an adeno-associated virus vector expressing human SMN. Hum. Gene Ther. 29, 285–298 (2018).
Mays, L. E. & Wilson, J. M. The complex and evolving story of T cell activation to AAV vector-encoded transgene products. Mol. Ther. 19, 16–27 (2011).
Kotterman, M. A., Chalberg, T. W. & Schaffer, D. V. Viral vectors for gene therapy: translational and clinical outlook. Annu. Rev. Biomed. Eng. 17, 63–89 (2015).
Moreno, A. M. et al. Immune-orthogonal orthologues of AAV capsids and of Cas9 circumvent the immune response to the administration of gene therapy. Nat. Biomed. Eng. 3, 806–816 (2019).
Wu, Z., Yang, H. & Colosi, P. Effect of genome size on AAV vector packaging. Mol. Ther. 18, 80–86 (2010).
Manno, C. S. et al. Successful transduction of liver in hemophilia by AAV-factor IX and limitations imposed by the host immune response. Nat. Med. 12, 342–347 (2006).
Chirmule, N. et al. Humoral immunity to adeno-associated virus type 2 vectors following administration to murine and nonhuman primate muscle. J. Virol. 74, 2420–2425 (2000).
Das, A. et al. Epigenetic silencing of recombinant adeno-associated virus genomes by NP220 and the HUSH complex. J. Virol. 96, e02039-21 (2022).
Cabrera, A. et al. The sound of silence: transgene silencing in mammalian cell engineering. Cell Syst. 13, 950–973 (2022).
Wang, J.-H., Gessler, D. J., Zhan, W., Gallagher, T. L. & Gao, G. Adeno-associated virus as a delivery vector for gene therapy of human diseases. Signal Transduct. Target. Ther. 9, 78 (2024).
Decout, A., Katz, J. D., Venkatraman, S. & Ablasser, A. The cGAS–STING pathway as a therapeutic target in inflammatory diseases. Nat. Rev. Immunol. 21, 548–569 (2021).
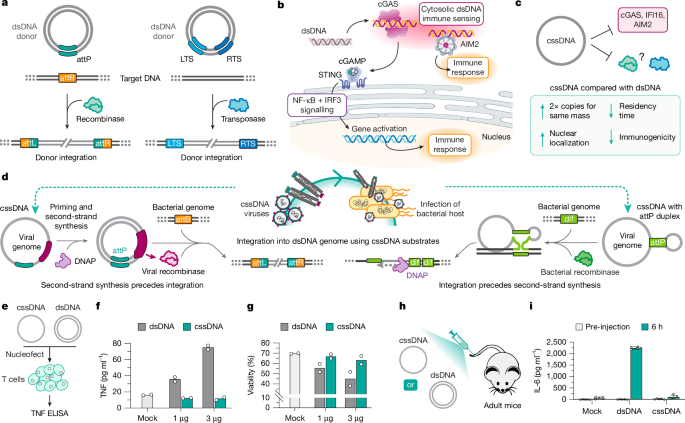
Xie, K. et al. Efficient non-viral immune cell engineering using circular single-stranded DNA-mediated genomic integration. Nat. Biotechnol. https://doi.org/10.1038/s41587-024-02504-9 (2024).
Cao, X., Tang, L. & Song, J. Circular single-stranded DNA: discovery, biological effects, and applications. ACS Synth. Biol. 13, 1038–1058 (2024).
Tang, L. et al. Circular single-stranded DNA as switchable vector for gene expression in mammalian cells. Nat. Commun. 14, 6665 (2023).
Kranzusch, P. J., Lee, A. S.-Y., Berger, J. M. & Doudna, J. A. Structure of human cGAS reveals a conserved family of second-messenger enzymes in innate immunity. Cell Rep. 3, 1362–1368 (2013).
Kumari, P., Russo, A. J., Shivcharan, S. & Rathinam, V. A. AIM2 in health and disease: Inflammasome and beyond. Immunol. Rev. 297, 83–95 (2020).
Roberts, T. L. et al. HIN-200 proteins regulate caspase activation in response to foreign cytoplasmic DNA. Science 323, 1057–1060 (2009).
Unterholzner, L. et al. IFI16 is an innate immune sensor for intracellular DNA. Nat. Immunol. 11, 997–1004 (2010).
Garg, A., Stallings, C. M. & Sohn, J. Filament assembly underpins the double-stranded DNA specificity of AIM2-like receptors. Nucleic Acids Res. 51, 2574–2585 (2023).
Krupovic, M. & Forterre, P. Single-stranded DNA viruses employ a variety of mechanisms for integration into host genomes. Ann. N. Y. Acad. Sci. 1341, 41–53 (2015).
Roth, T. L. et al. Reprogramming human T cell function and specificity with non-viral genome targeting. Nature 559, 405–409 (2018).
Shy, B. R. et al. High-yield genome engineering in primary cells using a hybrid ssDNA repair template and small-molecule cocktails. Nat. Biotechnol. 41, 521–531 (2023).
Iyer, S. et al. Efficient homology-directed repair with circular single-stranded DNA donors. CRISPR J. https://doi.org/10.1089/crispr.2022.0058 (2022).
Yeh, C. D., Richardson, C. D. & Corn, J. E. Advances in genome editing through control of DNA repair pathways. Nat. Cell Biol. 21, 1468–1478 (2019).
Tou, C. J. & Kleinstiver, B. P. Recent advances in double-strand break-free kilobase-scale genome editing technologies. Biochemistry https://doi.org/10.1021/acs.biochem.2c00311 (2022).
Shepherd, T. R., Du, R. R., Huang, H., Wamhoff, E.-C. & Bathe, M. Bioproduction of pure, kilobase-scale single-stranded DNA. Sci. Rep. 9, 6121 (2019).
Lucas, C. R. et al. DNA origami nanostructures elicit dose-dependent immunogenicity and are nontoxic up to high doses in vivo. Small 18, 2108063 (2022).
Ranawakage, D. C. et al. Efficient CRISPR-Cas9-mediated knock-in of composite tags in zebrafish using long ssDNA as a donor. Front. Cell Dev. Biol. 8, 598634 (2021).
Lin-Shiao, E. et al. CRISPR–Cas9-mediated nuclear transport and genomic integration of nanostructured genes in human primary cells. Nucleic Acids Res. 50, 1256–1268 (2022).
Andreeva, L. et al. cGAS senses long and HMGB/TFAM-bound U-turn DNA by forming protein–DNA ladders. Nature 549, 394–398 (2017).
Luecke, S. et al. cGAS is activated by DNA in a length-dependent manner. EMBO Rep. 18, 1707–1715 (2017).
Zhou, W. et al. Structure of the human cGAS–DNA complex reveals enhanced control of immune surveillance. Cell 174, 300–311 (2018).
Reus, J. B., Trivino-Soto, G. S., Wu, L. I., Kokott, K. & Lim, E. S. SV40 large T antigen is not responsible for the loss of STING in 293T cells but can inhibit cGAS-STING interferon induction. Viruses 12, 137 (2020).
Bonnamy, M., Blanc, S. & Michalakis, Y. Replication mechanisms of circular ssDNA plant viruses and their potential implication in viral gene expression regulation. mBio 14, e01692-23 (2023).
Geider, K. & Kornberg, A. Conversion of the M13 viral single strand to the double-stranded replicative forms by purified proteins. J. Biol. Chem. 249, 3999–4005 (1974).
Fanton, A. et al. Site-specific DNA insertion into the human genome with engineered recombinases. Nat. Biotechnol. https://doi.org/10.1038/s41587-025-02895-3 (2025).
Tobiason, D. M., Lenich, A. G. & Glasgow, A. C. Multiple DNA binding activities of the novel site-specific recombinase, Piv, from Moraxella lacunata. J. Biol. Chem. 274, 9698–9706 (1999).
Marrs, C. F., Rozsa, F. W., Hackel, M., Stevens, S. P. & Glasgow, A. C. Identification, cloning, and sequencing of piv, a new gene involved in inverting the pilin genes of Moraxella lacunata. J. Bacteriol. 172, 4370–4377 (1990).
Lau, L., Gray, E. E., Brunette, R. L. & Stetson, D. B. DNA tumor virus oncogenes antagonize the cGAS-STING DNA-sensing pathway. Science 350, 568–571 (2015).
Takaki, T., Millar, R., Hiley, C. T. & Boulton, S. J. Micronuclei induced by radiation, replication stress, or chromosome segregation errors do not activate cGAS-STING. Mol. Cell 84, 2203–2213 (2024).
Al Khatib, I. et al. Activation of the cGAS-STING innate immune response in cells with deficient mitochondrial topoisomerase TOP1MT. Hum. Mol. Genet. 32, 2422–2440 (2023).
Chen, G.-Y. et al. Defective antiviral responses of induced pluripotent stem cells to baculoviral vector transduction. J. Virol. 86, 8041–8049 (2012).
Anzalone, A. V. et al. Programmable deletion, replacement, integration and inversion of large DNA sequences with twin prime editing. Nat. Biotechnol. 40, 731–740 (2022).
Anzalone, A. V. et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576, 149–157 (2019).
Ferreira Da Silva, J. et al. Click editing enables programmable genome writing using DNA polymerases and HUH endonucleases. Nat. Biotechnol. https://doi.org/10.1038/s41587-024-02324-x (2024).
Liu, B. et al. Targeted genome editing with a DNA-dependent DNA polymerase and exogenous DNA-containing templates. Nat. Biotechnol. https://doi.org/10.1038/s41587-023-01947-w (2023).
Pandey, S. et al. Efficient site-specific integration of large genes in mammalian cells via continuously evolved recombinases and prime editing. Nat. Biomed. Eng. https://doi.org/10.1038/s41551-024-01227-1 (2024).
Karasu, M. E. et al. Removal of TREX1 activity enhances CRISPR–Cas9-mediated homologous recombination. Nat. Biotechnol. https://doi.org/10.1038/s41587-024-02356-3 (2024).
Yan, J. et al. Improving prime editing with an endogenous small RNA-binding protein. Nature 628, 639–647 (2024).
Hazelbaker, D. Z. et al. Large serine integrase off-target discovery and validation for therapeutic genome editing. Preprint at bioRxiv https://doi.org/10.1101/2024.08.23.609471 (2024).
Giannoukos, G. et al. UDiTaSTM, a genome editing detection method for indels and genome rearrangements. BMC Genom. 19, 212 (2018).
Adams, D. et al. Patisiran, an RNAi therapeutic, for hereditary transthyretin amyloidosis. N. Engl. J. Med. 379, 11–21 (2018).
Low, B. E., Hosur, V., Lesbirel, S. & Wiles, M. V. Efficient targeted transgenesis of large donor DNA into multiple mouse genetic backgrounds using bacteriophage Bxb1 integrase. Sci. Rep. 12, 5424 (2022).
Rose, J. et al. Engineered Bxb1 variants improve integrase activity and fidelity. Preprint at bioRxiv https://doi.org/10.1101/2024.10.21.619419 (2024).
Dong, M. & Fitzgerald, K. A. DNA-sensing pathways in health, autoinflammatory and autoimmune diseases. Nat. Immunol. 25, 2001–2014 (2024).
Patel, M. N. et al. Safer non-viral DNA delivery using lipid nanoparticles loaded with endogenous anti-inflammatory lipids. Nat. Biotechnol. https://doi.org/10.1038/s41587-025-02556-5 (2025).
Roberts, T. C., Langer, R. & Wood, M. J. A. Advances in oligonucleotide drug delivery. Nat. Rev. Drug Discov. 19, 673–694 (2020).
Yin, H. et al. Structure-guided chemical modification of guide RNA enables potent non-viral in vivo genome editing. Nat. Biotechnol. 35, 1179–1187 (2017).
Finn, J. D. et al. A single administration of CRISPR/Cas9 lipid nanoparticles achieves robust and persistent in vivo genome editing. Cell Rep. 22, 2227–2235 (2018).
Hew, B. E. et al. Directed evolution of hyperactive integrases for site specific insertion of transgenes. Nucleic Acids Res. 52, e64 (2024).
Huhtinen, O., Prince, S., Lamminmäki, U., Salbo, R. & Kulmala, A. Increased stable integration efficiency in CHO cells through enhanced nuclear localization of Bxb1 serine integrase. BMC Biotechnol 24, 44 (2024).
Karikó, K., Buckstein, M., Ni, H. & Weissman, D. Suppression of RNA recognition by toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity 23, 165–175 (2005).
Cha, T.-G. et al. Genetic control of aerogel and nanofoam properties, applied to Ni–MnO cathode design. Adv. Funct. Mater. 31, 2010867 (2021).
Wrenbeck, E. E. et al. Plasmid-based one-pot saturation mutagenesis. Nat. Methods 13, 928–930 (2016).
Kim, M. J. & Ahituv, N. in Pharmacogenomics: Methods and Protocols (eds Innocenti, F. & van Schaik, R. H. N.) 279–289 (Humana Press, 2013).
Rohland, N. & Reich, D. Cost-effective, high-throughput DNA sequencing libraries for multiplexed target capture. Genome Res. 22, 939–946 (2012).
Kleinstiver, B. P. et al. Engineered CRISPR–Cas12a variants with increased activities and improved targeting ranges for gene, epigenetic and base editing. Nat. Biotechnol. 37, 276–282 (2019).
Chen, P. J. et al. Enhanced prime editing systems by manipulating cellular determinants of editing outcomes. Cell 184, 5635–5652.e29 (2021).
Ge, S. X., Jung, D. & Yao, R. ShinyGO: a graphical gene-set enrichment tool for animals and plants. Bioinformatics 36, 2628–2629 (2020).
Escudero, J. A. et al. Unmasking the ancestral activity of integron integrases reveals a smooth evolutionary transition during functional innovation. Nat. Commun. 7, 10937 (2016).
Francia, M. V., Zabala, J. C., De La Cruz, F. & García Lobo, J. M. The IntI1 integron integrase preferentially binds single-stranded DNA of the attC site. J. Bacteriol. 181, 6844–6849 (1999).
Grabundzija, I., Hickman, A. B. & Dyda, F. Helraiser intermediates provide insight into the mechanism of eukaryotic replicative transposition. Nat. Commun. 9, 1278 (2018).
Del Pilar Garcillán-Barcia, M., Bernales, I., Mendiola, M. V. & De La Cruz, F. Single-stranded DNA intermediates in IS91 rolling-circle transposition. Mol. Microbiol. 39, 494–502 (2001).
Barabas, O. et al. Mechanism of IS200/IS605 family DNA transposases: activation and transposon-directed target site selection. Cell 132, 208–220 (2008).
Xie, K. et al. Efficient non-viral immune cell engineering using circular single-stranded DNA-mediated genomic integration. Nat. Biotechnol. 43, 1821–1832 (2025).
Hew, B. E. et al. Directed evolution of hyperactive integrases for site specific insertion of transgenes. Nucleic Acids Res. https://doi.org/10.1093/nar/gkae534 (2024).
Wu, J., Corbett, A. H. & Berland, K. M. The intracellular mobility of nuclear import receptors and NLS cargoes. Biophys. J. 96, 3840–3849 (2009).
Suzuki, K. et al. In vivo genome editing via CRISPR/Cas9 mediated homology-independent targeted integration. Nature 540, 144–149 (2016).
Witte, I. P. et al. Programmable gene insertion in human cells with a laboratory-evolved CRISPR-associated transposase. Science 388, eadt5199 (2025).