Despite the advances in herd management, animal care, stables, animal feeding, cattle industry and timely use of biopharmaceuticals, neonatal calf diarrhea remains a major cause of economic loss in the cattle industry worldwide32. Among the most significant etiological agents of diarrhea in newborns of numerous animal species worldwide is BVDV33. In addition to being crucial for monitoring the epidemiological state of a protected area, obtaining and characterizing BVDV isolates from the field is also essential for verifying the field protection provided by vaccines and immunizations. The viral strain lacks a particular genetic or structural characteristic (a “notch” or marker) that would enable researchers to readily change or differentiate it when developing vaccine strains. In order to control disease and avoid financial losses, it is critical to identify the disease factor in newborn calves in herds.
There is an important distinction between infectious and nutritional causes of diarrhea in buffalo calves. Although BVDV was detected, its presence does not exclude concurrent nutritional factors. In this study, cases were classified using clinical, management, and laboratory criteria to distinguish between infectious and nutritional diarrhea. This approach minimizes misclassification and clarifies BVDV’s role: detection in febrile, systemically affected calves supports an infectious origin, whereas its presence in calves with recent dietary changes but no systemic signs may indicate an incidental or contributing factor. Future work should apply multiplex diagnostics and standardized feeding assessments to better elucidate mixed etiologies.
In the present investigation, the diarrheal calves had variable degree of diarrhea, dehydration, pale mucous membranes, weakness, emaciation, dullness, depression, and off-food. Additionally, their body temperature, pulse rate, and respiration rate significantly increased in comparison to the healthy calves. The clinical results we obtained were comparable to those reported by34,35,36,37,38,39,40. Dehydration and metabolic acidosis contribute to increased rectal temperatures due to reduced heat elimination efficiency, followed by a decrease in body temperature. Metabolic acidosis stimulates the sympathetic nervous system, leading to elevated heart rates (tachycardia) and respiratory rates (hyperpnea)41,42. According to Radostits et al.43, muscle weakness brought on by intracellular potassium escaping, hyperkalemia, and hypoglycemia may be the cause of the observed anorexia, depression, and dullness. However, the production of pyrogenic substances such prostaglandins, hypothalamic thermoregulatory center motivation, infection and inflammation may be responsible for the symptoms of pain and hyperthermia44.
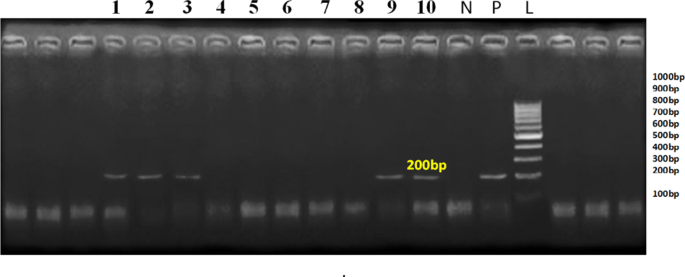
Many techniques, such as electron microscopy, antigen-capture ELISA, lateral flow assays, and molecular approaches, can be used to identify the virus because acute infection causes it to be excreted in enormous amounts in the feces (up to 10¹¹ particles/g). Because PCR is such a sensitive molecular technique, it was used to analyze all of the samples gathered for this investigation. Although diarrhea in buffalo calves typically results from mixed infections involving viral, bacterial, and protozoal agents, only BVDV was detected in the present study. This may be attributed to the specific molecular focus on BVDV, while other enteropathogens such as BRV, BCV, ETEC, and protozoa were not included in the screening. In addition, species-specific differences in susceptibility, sampling timing, and regional epidemiological factors could have influenced detection outcomes. The findings suggest that BVDV may play a significant role in enteric disorders of buffalo calves under certain field conditions. However, future investigations using multiplex or metagenomic approaches are recommended to reveal the full spectrum of enteric pathogens and their interactions. The molecular results of our investigation showed that 20% of the calves analyzed in South Sinai Governorate, Egypt, had BVD, which is about the same as the results of the earlier study (21.43% in DAMIETTA, Egypt) by45. As reported by Mahranet al.46,47,48,49,50,51, Gabr et al.48, Soltan et al.49, Mokhtar et al.50, Ahmed et al.51, the prevalence of BVD infection in the animals under examination in Fayoum, Alexandria, Qaluobia, Ismailia, and Assiut in Egypt, was 6.7%, 8.4%, 10.4%, 3.4%, and 15%, respectively. According to Chang et al.52,53, the molecular positive rates for BVDV in Holstein were 7.2 and 1.42%, respectively. Our rates were lower than that of54 (22.64%) in Switzerland, and55 in Chinese dairy cattle, all of whom had rates of 27.1%. Differences in sample collecting times, sample sizes, sanitary conditions, environmental conditions, and the use of various diagnostic techniques could all be responsible for the discrepancies.
The nucleotide identity of the 5’UTR gene varied from 84.1% to 100%, according to a sequence comparison for BVD strains. The Polish dairy cattle BVDV-1 strain (MK381368) had the lowest homology (83.1%) when compared to representative BVD strains in GenBank, while the BVDV-1 strains isolated from dairy cattle and buffalo in Ismailia, Egypt (KP127973) had the highest homology (100%). Additional strains exhibiting 100% homology include those from Chinese naturally fermented dairy products (KP029825) and American calf (MW713362).
We investigated the alterations in the immunological and antioxidant states of calves with diarrhea in comparison to healthy calves by assessing the mRNA levels of immune and antioxidant genes. The expression of the genes SLC11A1, CD14, PTX3, IRF3 and ST1P1 was much higher in the affected calves than in the healthy ones. However, PRDX2, PRDX6, and GPX levels were decreased.
This study used a PCR-DNA sequencing technique to characterize the immunological (SLC11A1, CD14, PTX3, and IRF3) and antioxidant (PRDX2, PRDX6, GPX, ST1P1) genes in calves with diarrhea and healthy calves. The results demonstrate that there are differences in the SNPs involving the two categories. It is crucial to stress that, in contrast to the similar datasets obtained from GenBank, the polymorphisms discovered and made available in this context offer extra information for the evaluated indicators. No research has looked at the relationship between buffalo calves’ risk of diarrhea and SNPs alongside expression changes in the immune and antioxidant genes. This link is initially shown by the Bubalus bubalis gene sequences used in our study, which were published in PubMed.
Three of the thirteen SNPs found by sequencing were directly linked to calf diarrhea among the genes under investigation. Two of these were non-synonymous changes that were expected to alter the structure and function of proteins. Non-synonymous mutations frequently change the structure or function of proteins, which may affect immunological and metabolic processes56,57. Conversely, while some synonymous changes can affect gene regulation, they are generally thought to have less of an effect56. It was hypothesized in this study that substitutions like PTX3 (A22G) and PRDX2 (A51S) would decrease immunological and antioxidant activities. Additional functional confirmation of these loci is necessary58.
Newborn animals with diarrhea were monitored for health using the candidate gene approach. For example, CXCR1 SNPs were not significantly linked to clinical intestinal disorders in dairy calves, according to Hosseini Moghaddam et al.59, has demonstrated a connection between a genetic variant in the swine leukocyte antigen-DRA gene and piglet diarrhea. Additionally, Cheng et al.60, revealed the polymorphism of the Nramp1 gene and its correlation with pig diarrhea. Nucleotide sequence differences between healthy and afflicted kids were found by PCR-DNA sequencing of goats for the TMED1, CALR, FBXW9, HS6ST3, SMURF1, KPNA7, FBXL2, PIN1, S1PR5, ICAM1, EDN1, MAPK11, CSF1R, LRRK1, and CFH markers61. The immunological (SELL, JAK2, SLC11A1, IL10, FEZF1, NCF4, LITAF, SBD2, NFKB, TNF-α, IL1B, IL6, LGALS, and CATH1) and antioxidant (SOD1, CAT, GPX1, GST, Nrf2, Keap1, HMOX1, and NQO1) indicators linked to bacterial diarrhea susceptibility in Barki lambs were examined by62.
Transcriptome study of goat peripheral blood mononuclear cells (PBMCs) infected with the bovine viral diarrhea virus-2 revealed differential expression of immune-related genes60, according to the ruminant gene expression profile63. It was further explained that the TLR4 and downstream signalling pathways of newborn goats with diarrhea and healthy goats shared characteristics. Additionally, it was determined that the gene expression profile of diarrheal goats showed considerably higher levels of TMED1, CALR, FBXW9, HS6ST3, SMURF1, KPNA7, FBXL2, PIN1, S1PR5, ICAM1, EDN1, MAPK11, CSF1R, and LRRK1 than resistant goats61. Keap1, HMOX1, CMPK2, ASPG, FPGT, TNNI3 K, and LPCAT1 genes were much more highly expressed in diarrheal calves compared to resistant ones, according to Ateya et al.64. However, a distinct pattern was generated by the Nrf2, PRDX2, and PRDX6 genes.
The examined genes’ nucleotide sequences differed between the diarrheal and healthy calves. Gene expression analysis revealed that PTX3 was significantly up-regulated (p < 0.05) in pneumonic ewes than healthy ones65. Single nucleotide polymorphisms and GPX marker gene expression were linked to bacterial pneumonia in Barki sheep, according to Sayed et al.66.
Transmembrane protein solute carrier family 11 member 1 (SLC11A1) has been identified as one of the most well-known putative candidate genes that support innate immunity against many intracellular infections67. When68 sequenced and examined the SLC11A1 gene in Holstein and Brown, they discovered SNPs linked to mastitis susceptibility or tolerance. Compared to tolerant Holstein and Brown Swiss dairy cows, mastitic Holsteins have substantially higher levels of SLC11A1 gene expression.
CD14 has a vital role in innate immunity. According to Wright et al.69, the anti-bacterial peptide CD14 is one of the most important molecules that binds and neutralises bacterial endotoxins. CD14 and PRDX2 nucleotide sequence variation and expression were correlated with mastitis resistance/susceptibility in goat70.
In response to primary inflammatory stimuli, such as those mediated by TNFα, IL-1β, and TLR agonists, several cell types produce the glycoprotein pentraxin 3 (PTX3)71. The PTX3 gene is principally in charge of controlling inflammatory reactions and inherent resistance to infections, claims72. Additionally, it has antibacterial properties that may help shield the mammary gland from subclinical and chronic infections. PTX3, which is significantly up-regulated in the goats’ udder, is the first line of defence against S. aureus infection, according to multiple studies73. Single nucleotide polymorphisms (SNPs) and alterations in the gene expression profile associated with mastitis resistance/susceptibility were discovered in the PTX3 gene in the Holstein and Montbéliarde dairy cows under investigation74.
IRF3 belongs to the family of interferon regulatory transcription factors (IRF)75. Response to microbial infection by the innate immune system is significantly influenced by IRF376. Individual genetic loci and MRNA levels of the IRF3 biomarker linked to pneumonia susceptibility in Baladi goats were discovered by Ateya et al.77.
Hydrogen peroxide can be catalyzed by the peroxiredoxin (PRDX) family of antioxidant enzyme oxidoreductase proteins due to a conserved ionised thiol. Thiol-specific peroxidase detoxifies peroxides, including sulphur, and radicals, assisting cells in defending against oxidative stress. Additionally, according to Wadley et al.78, it acts as a sensor for signaling events brought on by hydrogen peroxide. The PRDX6 gene was examined by Elsayed et al.79 as an antioxidant linked to dromedary camels’ vulnerability to trypanosomiasis.
Free radicals can be scavenged or detoxified by antioxidants, which can also stop them from generating or sequester the transition metals that cause them80. Glutathione peroxidase (GPX) is an example of enzymatic and nonenzymatic mechanisms that make up the body’s natural antioxidant defenses81.
The functions of HSP70 and HSP90 in protein folding are regulated and coordinated by the adaptor protein stress-induced phosphoprotein (STIP1)82. Furthermore, STIP1 is expressed in response to cellular physiological stress caused by elevated temperatures or.
additional elements83. In order to select for and enhance mastitis resistance in Dromedary camels84, integrated the nucleotide sequence variations and transcript levels of the STIP1marker.
The significant change in the pattern of immunological, and antioxidant, marker expression in diarrheal calves may be explained by the fact that damaged tissues react to free radicals more frequently than healthy ones85. Additionally, the intestine, which was the main site for a variety of microbes, nutrients, and immune cell interactions, was extremely susceptible to degradation86. The rationale may be that invasions by gastrointestinal pathogens are potent oxidizing triggers that set off immunological responses that mobilize macrophages and neutrophils to fight off invasions. This results in an excess of ROS generation and buildup, which eventually leads to oxidative stress87. It is clear that during bacterial infection, TLR4 and the signaling pathways it interacts with downstream are essential for inducing the production of inflammatory cytokines. Significant alterations in the mechanism controlling the gut barrier’s function are associated with diarrhea, which may increase the intestinal permeability to harmful microorganisms88. Additionally, ROS takes involvement in competition between microorganisms89. We therefore hypothesise that the diarrhea seen in these calves is mostly an infectious one. Furthermore, there is solid evidence from our real-time PCR data indicating the diarrheal calves were exhibiting a marked inflammatory response.
The diarrheal calves had significantly lower amounts of serum glucose, total protein, albumin, globulin than the healthy ones. Our findings matched with those published by40,90,91. Hypoglycemia in diarrheal calves may be caused by decreased glucose absorption from the damaged intestine and glucose excretion in the intestinal lumen during diarrhea, according to the available evidence92,93. Damage to the intestinal mucosa and increased vascular permeability may result in a protein-losing enteropathy that causes hypoproteinemia, hypoalbuminemia, and hypoglobulinemia in the diarrheal calves94.
When compared to control calves, the serum concentrations of triglyceride, creatinine, urea, and the activities of AST, ALT, GGT and LDH were significantly higher in the diarrheal calves. Our findings aligned with those of 40,95. Serum triglyceride levels have significantly increased, which may be related to poor liver function and fatty tissue lipolysis with impaired fatty acid production during infections and inflammation96. According to Singh et al.97, a decrease in renal function, a drop in glomerular filtration rate, and a decrease in urine production as a result of hypovolemia, systemic arterial hypotension, and vasopressin release may account for the elevated levels of creatinine and urea in calves with diarrhea. It may also be the consequence of the body’s proteins being catabolized under adverse conditions, which leads to an excess of urea98. The hemoconcentration or the spread of BVDV into extra-intestinal tissues, lamina propria, Peyer’s patches, mesenteric lymph nodes, lung, liver, kidney, and bile duct may be the cause of the markedly higher average ALT, AST, GGT, LDH concentrations in the serum of BVDV-infected calves compared to healthy calves99.
Since the creatine phosphokinase (CPK) enzyme is essential for maintaining tissue cells’ energy homeostasis and ensuring a steady level of ATP in the cells, elevated plasma concentrations of CPK are linked to tissue damage, poor muscle tissue reperfusion, hypoxia, exhaustion, and increased muscle membrane permeability after stress and abrupt metabolic changes. It is therefore helpful in the assessment of conditions including injury to the central nervous system, skeletal muscle, and myocardium. According to Huang et al.100, an increase in diarrheal calves is a sign of exhaustion, metabolic diseases, and skeletal muscle degenerations.
In order to monitor health and vitality, C-reactive protein (CRP), a diagnostic inflammatory biomarker that rises quickly in cases of inflammation or tissue destruction, binds with metabolites released from cellular degeneration to reenter the host metabolic processes so that the pathogen cannot use them. CRP elevation in diarrheal calves thus signifies pathogenic infection and tissue injury101.
Serum cardiac troponin I (cTn-I) levels were considerably higher in the diarrheal calves than in the healthy ones. Our findings agreed with those of95. Since pericarditis causes the release of cTn proteins into the bloodstream, which are normally present in blood at very low concentrations or below the limit in detection of most assays, the significant increase in cTn-I concentration in the affected group suggests myocardial cell damage102.
Serum cortisol levels were considerably higher in the diarrheal calves than in the healthy ones. Our results were consistent with those of95. When stress occurs, the primary hormone secreted to restore physiological conditions and homeostasis is cortisol. In the present investigation, diarrheal calves’ cortisol levels rose in an attempt to alleviate the stress brought on by the clinical and biochemical disruptions brought on by diarrhea103.
The diarrheic calves had significantly higher blood K levels and significantly lower amounts of serum Na and Cl levels than the healthy calves. Our results were consistent with those of40,104,105. The observed variations in serum Na, Cl, and K levels may be caused by hypovolemia, which is linked to a decrease in glomerular filtration rate, which is a major factor in the pathophysiology of hyperkalemia, as well as excessive water loss with feces, which results in dehydration and impaired cell membrane permeability92.
Diarrheic calves infected with BVDV also had significantly lower levels of Ca, P, Cu, Zn and Fe. The results of the present investigation were in line with those of earlier studies105,106. Persistent diarrhea and dehydration caused by the loss of calcium in stools may be the cause of the drop in calcium levels98. According to EL-dessouky et al.107, the decline in P was caused by more electrolyte loss than water loss. According to Palomares108, the notable decline in Cu, Zn, and Fe levels may be caused by a reduction in the intestinal absorption of dietary nutrients and fecal losses.
Acute phase proteins constitute an extra component of the innate immune response. These non-specific hepatic proteins are produced in reaction to cytokines that promote inflammation109. Diarrheic calves in this study exhibited significantly higher serum concentrations of APPs (HP, SAA and Cp). These findings were similar to those that were published by104,110,111. They are crucial in preserving homeostasis and biological harmony as well as regulating microbial growth until a specific immunity is established. Additionally, they possess strong anti-inflammatory, antibacterial, and antioxidant properties112. Moreover, APPs may enable treatment choices and serve as helpful prognostic instruments113.
The close correlation between gene expression and serum biochemical profiles highlights the interplay between immune activation, oxidative stress, and metabolic disturbance during diarrhea in buffalo calves. The strong association of SLC11A1 with proteins and minerals supports its role in macrophage activation and metal ion transport, while elevated correlations with cortisol and cTnI indicate systemic stress and secondary cardiac involvement114,115. The association of CD14 with Hp and AST indicates activation of innate immune signaling via LPS recognition, stimulating hepatic APP synthesis, while its negative correlation with Ca reflects electrolyte loss during diarrhea116. PTX3, IRF3, and PRDX2 showed strong correlations with CRP, GGT, LDH, and cortisol, highlighting their roles in acute-phase and oxidative defense responses117. The linkage of PRDX2 with CPK suggests protection against oxidative damage in muscle and liver tissues117. The relationship between GPX and Zn supports the trace element dependency of glutathione peroxidase activity, and the correlation of NDUFS6 with Cl implies mitochondrial adaptation to ionic imbalance during dehydration and acidosis118.
Although this study provides valuable insights into the molecular detection, genetic characterization, and host immune response to BVDV infection in diarrheic buffalo calves, several limitations should be acknowledged. The restricted sample size and geographic coverage may not reflect the national epidemiological status of BVDV in Egypt. The focus on BVDV alone, without screening for other enteric pathogens, limits understanding of its role in mixed infections. Future multi-regional studies with broader pathogen screening and whole-genome sequencing are needed to clarify virulence factors and enhance vaccine matching. Because only fecal samples from diarrheic calves were analyzed and paired blood samples were unavailable, the true prevalence and systemic infection status could not be fully assessed. Including both healthy and diarrheic animals in longitudinal studies integrating molecular, immunological, and biochemical analyses would enable functional validation and improve understanding of BVDV-associated diarrhea in buffalo calves.