You have full access to this article via your institution.
Mechanical cues at intracellular organelles are emerging as a parallel layer of immune regulation alongside biochemical signaling. A recent study published in Cell Research shows that programmable magneto-mechanical stimulation of lysosomes can integrate intracellular forces over time to durably reprogram macrophage polarization and antitumor immunity.
Tumor-associated macrophages are among the most abundant immune cells in solid tumors, yet in many cancers they predominantly adopt an M2-like polarization state. These M2-like macrophages support tumor growth by promoting immune suppression, angiogenesis, tissue remodeling, and resistance to therapy.1,2,3 In contrast, M1-like macrophages exhibit pro-inflammatory activity, enhance antigen presentation, and support cytotoxic immune responses. Consequently, shifting macrophages from an M2-like to an M1-like state has become a major therapeutic objective in cancer immunotherapy. However, despite strong preclinical evidence, achieving stable and durable macrophage reprogramming in vivo remains challenging, and many strategies are limited by toxicity, transient effects, or rapid re-education by the tumor microenvironment.2,3
Most current macrophage-targeting therapies rely on biochemical signaling pathways, including CSF1–CSF1R blockade, CD40 and Toll-like receptor agonists, cytokine-based treatments, STAT3 inhibitors, TFEB activation strategies, and engineered delivery systems. In many cases, these approaches show modest activity as monotherapies and require combination with immune checkpoint inhibitors to achieve meaningful antitumor effects.2,3,4,5 A recurring limitation is that macrophage repolarization is often transient or dose-limited by on-target and systemic effects. In this context, the study by Cheng and colleagues introduces a fundamentally different strategy that exploits subcellular magneto-mechanical stimulation to control macrophage fate.6
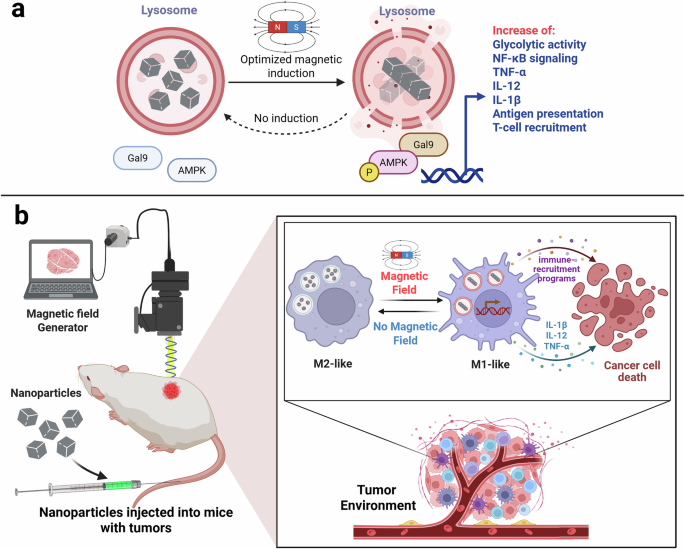
While most magnetogenetic approaches rely on magnetic field-responsive ion channels to regulate ionic fluxes in cells, including immune cells,7 the present study adopts a distinct strategy. Cheng et al. employ magnetic nanomotors that are internalized by macrophages and accumulate within lysosomes through the endolysosomal pathway.6 Upon exposure to a rotating magnetic field, these nanomotors self-assemble and generate localized fluid motion inside the lysosomal lumen. Importantly, the magnitude of the resulting mechanical force can be tuned by adjusting the frequency of the magnetic field, enabling precise control over intracellular mechanical stimulation and lysosomal membrane permeabilization (LMP) (Fig. 1a).
Fig. 1: Magneto-mechanical lysosomal control of macrophage immunity.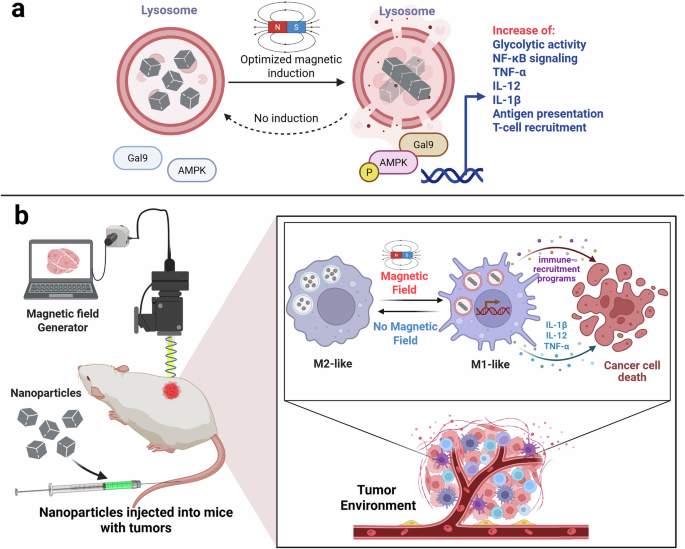
a Magnetic nanomotors accumulate in macrophage lysosomes and, upon rotating magnetic fields, induce reversible LMP, triggering Gal9-AMPK-NF-κB signaling. b Lysosomal mechanostimulation drives an M1-like macrophage state with increased glycolysis, inflammatory cytokines (TNF-α, IL-12, IL-1β), antigen presentation, and T cell recruitment, promoting antitumor immunity in nanoparticle-treated tumor-bearing mice.
Although LMP has traditionally been studied using chemical agents and peptides for diverse applications, the controlled magneto-mechanical LMP (MagLMP) described here introduces a fundamentally different paradigm.6 Rather than representing an inherently pathological form of lysosomal damage, moderate and cyclic mechanical perturbation produces a repairable membrane state that functions as a signaling event rather than a trigger for cell death. This reversibility is essential, as it enables repeated stimulation without compromising macrophage viability, thereby permitting sustained immune modulation.
Beyond its technical novelty, this work introduces a broader conceptual shift by establishing intracellular mechanical forces as a signaling modality in their own right. By tuning the frequency and repetition of magneto-mechanical stimulation, the study demonstrates that force itself can encode biological information alongside established biochemical and metabolic signaling pathways, consistent with emerging views of lysosomes as mechanosensitive immune signaling hubs.8
Mechanistically, the authors identify galectin-9 (Gal9) as a key sensor of mechanically induced lysosomal stress.6 Gal9 is recruited to lysosomes following MagLMP, where it engages AMP-activated protein kinase (AMPK) independently of classical energy stress. In this context, AMPK functions as a transducer of mechanically induced lysosomal signals rather than a conventional metabolic sensor, consistent with established lysosomal damage signaling pathways that connect galectin recruitment to AMPK activation.9 Activation of AMPK leads to downstream activation of the NF-κB pathway, resulting in transcriptional and metabolic reprogramming toward an inflammatory, M1-like macrophage phenotype.6
A particularly striking feature of the study is the durability of the immune response. Repeated, sublethal lysosomal perturbations do not merely induce transient activation but instead stabilize macrophage identity over time, even within immunosuppressive tumor microenvironments.1 This behavior suggests that lysosomes act as intracellular force-integrating platforms capable of translating repeated mechanical inputs into a persistent immune state, enabling immune programming that outlasts individual stimulation events.
In vivo, magneto-mechanical stimulation of lysosomes induces robust antitumor effects across multiple syngeneic mouse tumor models.6 Treatment results in reduced tumor growth, prolonged survival, and remodeling of the tumor immune microenvironment, accompanied by macrophage repolarization toward an M1-like state, increased cytotoxic T cell infiltration, and enhanced inflammatory signaling (Fig. 1b). Notably, these immune effects persist beyond the stimulation period, supporting the induction of a durable immune state rather than a transient activation.
Despite its promise, several translational challenges remain. The long-term safety, biodistribution, and clearance of magnetic nanomotors must be evaluated, particularly their accumulation in liver and spleen macrophages. In addition, the requirement for localized magnetic field application in disseminated or metastatic lesions with poorly defined anatomical locations may limit the range of treatable tumors. Precise control of stimulation parameters will also be required to avoid excessive lysosomal damage or unintended effects on non-target cells. These considerations define key areas for future development.
It is also informative to compare this strategy with pharmacological approaches that target lysosomes. Cationic amphiphilic drugs (CADs), such as chloroquine and some antihistamines, accumulate in lysosomes and can induce lysosomal stress, LMP, and downstream signaling programs, including STAT3 inhibition and TFEB activation, that promote macrophage repolarization toward an M1-like phenotype.4,5 CADs have shown enhanced antitumor efficacy when combined with immune checkpoint inhibitors.10 However, these agents act systemically and lack spatial specificity. In contrast, magneto-mechanical lysosomal stimulation offers a non-chemical and more controllable approach with spatial and temporal precision.
Together, the study by Cheng and colleagues positions intracellular mechanics as a programmable signaling language and identifies lysosomes as central hubs for integrating mechanical information into durable immune decisions. By extending immune regulation beyond chemical and genetic mechanisms to include mechanical force, this work opens new conceptual and therapeutic directions for macrophage-targeted cancer immunotherapy.