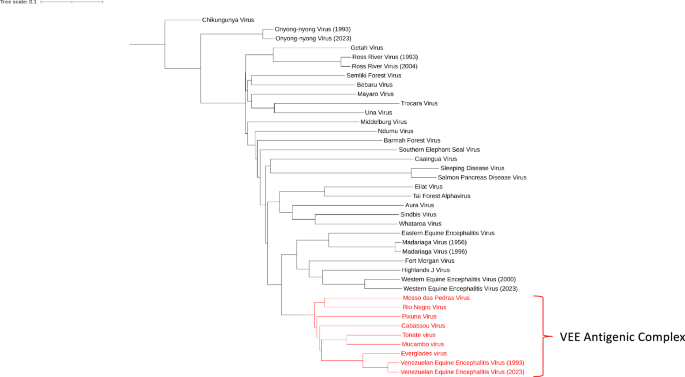
Forrester, N. L. et al. Evolution and spread of Venezuelan equine encephalitis complex alphavirus in the Americas. PLoS Negl. Trop. Dis. 11, e0005693 (2017).
Estrada-Franco, J. G. et al. Venezuelan equine encephalitis virus, southern Mexico. Emerg. Infect. Dis. 10, 2113–2121 (2004).
Kubes, V. & Rios, F. A. The causative agent of infectious equine encephalomyelitis in Venezuela. Science 90, 20–21 (1939).
Oberste, M. S. et al. Association of Venezuelan equine encephalitis virus subtype IE with two equine epizootics in Mexico. Am. J. Trop. Med. Hyg. 59, 100–107 (1998).
Barrera, R. et al. Contrasting sylvatic foci of Venezuelan equine encephalitis virus in northern South America. Am. J. Trop. Med. Hyg. 67, 324–334 (2002).
Brault, A. C., Powers, A. M. & Weaver, S. C. Vector infection determinants of Venezuelan equine encephalitis virus reside within the E2 envelope glycoprotein. J. Virol. 76, 6387–6392 (2002).
Ferro, C. et al. Natural enzootic vectors of Venezuelan equine encephalitis virus, Magdalena Valley, Colombia. Emerg. Infect. Dis. 9, 49–54 (2003).
Kramer, L. D. & Scherer, W. F. Vector competence of mosquitoes as a marker to distinguish Central American and Mexican epizootic from enzootic strains of Venezuelan encephalitis virus. Am. J. Trop. Med. Hyg. 25, 336–346 (1976).
Navarro, J. C. & Weaver, S. C. Molecular phylogeny of the Vomerifer and Pedroi Groups in the Spissipes Section of the subgenus Culex (Melanoconion). J. Med. Entomol. 41, 575–581 (2004).
Rivas, F. et al. Epidemic Venezuelan equine encephalitis in La Guajira, Colombia, 1995. J. Infect. Dis. 175, 828–832 (1997).
Franz, D. R. et al. Clinical recognition and management of patients exposed to biological warfare agents. Clin. Lab Med. 21, 435–473 (2001).
Hanson, R. P. et al. Arbovirus infections of laboratory workers. Extent of problem emphasizes the need for more effective measures to reduce hazards. Science 158, 1283–1286 (1967).
Phelps, A. L. et al. Susceptibility and lethality of western equine encephalitis virus in Balb/c mice when infected by the aerosol route. Viruses 9, https://doi.org/10.3390/v9070163 (2017).
Reed, D. S. et al. Aerosol exposure to western equine encephalitis virus causes fever and encephalitis in cynomolgus macaques. J. Infect. Dis. 192, 1173–1182 (2005).
Roy, C. J. et al. Pathogenesis of aerosolized Eastern Equine Encephalitis virus infection in guinea pigs. Virol. J. 6, 170 (2009).
Williams, J. A. et al. Eastern equine encephalitis virus rapidly infects and disseminates in the brain and spinal cord of cynomolgus macaques following aerosol challenge. PLoS Negl. Trop. Dis. 16, e0010081 (2022).
Pittman, P. R. et al. Long-term duration of detectable neutralizing antibodies after administration of live-attenuated VEE vaccine and following booster vaccination with inactivated VEE vaccine. Vaccine 14, 337–343 (1996).
Alevizatos, A. C., McKinney, R. W. & Feigin, R. D. Live, attenuated Venezuelan equine encephalomyelitis virus vaccine. Clin. Eff. Man. Am. J. Trop. Med. Hyg. 16, 762–768 (1967).
Casamassima, A. C., Hess, L. W. & Marty, A. TC-83 Venezuelan equine encephalitis vaccine exposure during pregnancy. Teratology 36, 287–289 (1987).
Guzman-Teran, C., Calderon-Rangel, A., Rodriguez-Morales, A. & Mattar, S. Venezuelan equine encephalitis virus: the problem is not over for tropical America. Ann. Clin. Microbiol. Antimicrob. 19, 19 (2020).
Luethy, D. Eastern, Western, and Venezuelan Equine Encephalitis and West Nile viruses: clinical and public health considerations. Vet. Clin. North Am. Equine Pr. 39, 99–113 (2023).
Weaver, S. C., Bellew, L. A. & Rico-Hesse, R. Phylogenetic analysis of alphaviruses in the Venezuelan equine encephalitis complex and identification of the source of epizootic viruses. Virology 191, 282–290 (1992).
Anishchenko, M. et al. Venezuelan encephalitis emergence mediated by a phylogenetically predicted viral mutation. Proc. Natl. Acad. Sci. USA 103, 4994–4999 (2006).
Moncayo, A. C. et al. Vector competence of eastern and western forms of Psorophora columbiae (Diptera: Culicidae) mosquitoes for enzootic and epizootic Venezuelan equine encephalitis virus. Am. J. Trop. Med. Hyg. 78, 413–421 (2008).
Ortiz, D. I., Anishchenko, M. & Weaver, S. C. Susceptibility of Psorophora confinnis (Diptera: Culicidae) to infection with epizootic (subtype IC) and enzootic (subtype ID) Venezuelan Equine encephalitis viruses. J. Med. Entomol. 42, 857–863 (2005).
Sudia, W. D. et al. Epidemic Venezuelan equine encephalitis in North America in 1971: vector studies. Am. J. Epidemiol. 101, 17–35 (1975).
Kafai, N. M. et al. Entry receptor LDLRAD3 is required for Venezuelan equine encephalitis virus peripheral infection and neurotropism leading to pathogenesis in mice. Cell Rep. 42, 112946 (2023).
Ma, B., Huang, C., Ma, J., Xiang, Y. & Zhang, X. Structure of Venezuelan equine encephalitis virus with its receptor LDLRAD3. Nature 598, 677–681 (2021).
Ma, H. et al. LDLRAD3 is a receptor for Venezuelan equine encephalitis virus. Nature 588, 308–314 (2020).
Adeyinka, O. S. et al. nsP2 protease inhibitor blocks the replication of New World alphaviruses and offer protection in mice. ACS Infect. Dis. 11, 181–196 (2025).
de Groot, R. J., Hardy, W. R., Shirako, Y. & Strauss, J. H. Cleavage-site preferences of Sindbis virus polyproteins containing the non-structural proteinase. Evidence for temporal regulation of polyprotein processing in vivo. EMBO J. 9, 2631–2638 (1990).
Lark, T., Keck, F. & Narayanan, A. Interactions of alphavirus nsP3 protein with host proteins. Front. Microbiol. 8, 2652 (2017).
Shirako, Y. & Strauss, J. H. Regulation of Sindbis virus RNA replication: uncleaved P123 and nsP4 function in minus-strand RNA synthesis, whereas cleaved products from P123 are required for efficient plus-strand RNA synthesis. J. Virol. 68, 1874–1885 (1994).
Li, C. et al. mRNA capping by Venezuelan equine encephalitis virus nsP1: functional characterization and implications for antiviral research. J. Virol. 89, 8292–8303 (2015).
Hoffka, G. et al. Self-inhibited state of Venezuelan equine encephalitis virus (VEEV) nsP2 cysteine protease: a crystallographic and molecular dynamics analysis. J. Mol. Biol. 435, 168012 (2023).
Kim, D. Y., Atasheva, S., Frolova, E. I. & Frolov, I. Venezuelan equine encephalitis virus nsP2 protein regulates packaging of the viral genome into infectious virions. J. Virol. 87, 4202–4213 (2013).
Foy, N. J., Akhrymuk, M., Shustov, A. V., Frolova, E. I. & Frolov, I. Hypervariable domain of nonstructural protein nsP3 of Venezuelan equine encephalitis virus determines cell-specific mode of virus replication. J. Virol. 87, 7569–7584 (2013).
Kril, V. et al. Alphavirus nsP3 organizes into tubular scaffolds essential for infection and the cytoplasmic granule architecture. Nat. Commun. 15, 8106 (2024).
Tan, Y. B. et al. Crystal structures of alphavirus nonstructural protein 4 (nsP4) reveal an intrinsically dynamic RNA-dependent RNA polymerase fold. Nucleic Acids Res. 50, 1000–1016 (2022).
Kielian, M., Chanel-Vos, C. & Liao, M. Alphavirus entry and membrane fusion. Viruses 2, 796–825 (2010).
Leung, J. Y., Ng, M. M. & Chu, J. J. Replication of alphaviruses: a review on the entry process of alphaviruses into cells. Adv. Virol. 2011, 249640 (2011).
Mukhopadhyay, S. et al. Mapping the structure and function of the E1 and E2 glycoproteins in alphaviruses. Structure 14, 63–73 (2006).
Mayne, J. T., Rice, C. M., Strauss, E. G., Hunkapiller, M. W. & Strauss, J. H. Biochemical studies of the maturation of the small Sindbis virus glycoprotein E3. Virology 134, 338–357 (1984).
Sjoberg, M., Lindqvist, B. & Garoff, H. Activation of the alphavirus spike protein is suppressed by bound E3. J. Virol. 85, 5644–5650 (2011).
Liljestrom, P. & Garoff, H. Internally located cleavable signal sequences direct the formation of Semliki Forest virus membrane proteins from a polyprotein precursor. J. Virol. 65, 147–154 (1991).
Welch, W. J. & Sefton, B. M. Two small virus-specific polypeptides are produced during infection with Sindbis virus. J. Virol. 29, 1186–1195 (1979).
Zhang, R. et al. 4.4 A cryo-EM structure of an enveloped alphavirus Venezuelan equine encephalitis virus. EMBO J. 30, 3854–3863 (2011).
Parker, M. D. et al. Antibody to the E3 glycoprotein protects mice against lethal venezuelan equine encephalitis virus infection. J. Virol. 84, 12683–12690 (2010).
Atasheva, S., Garmashova, N., Frolov, I. & Frolova, E. Venezuelan equine encephalitis virus capsid protein inhibits nuclear import in Mammalian but not in mosquito cells. J. Virol. 82, 4028–4041 (2008).
Negi, V., Miller, A. S. & Kuhn, R. J. Advances in viroporin function and structure: a comparative analysis of alphavirus 6k with well-characterized viroporins. Viruses 17, 868 (2025).
Gardner, C. L. et al. Eastern and Venezuelan equine encephalitis viruses differ in their ability to infect dendritic cells and macrophages: impact of altered cell tropism on pathogenesis. J. Virol. 82, 10634–10646 (2008).
Trabalza, A. et al. Venezuelan equine encephalitis virus glycoprotein pseudotyping confers neurotropism to lentiviral vectors. Gene Ther. 20, 723–732 (2013).
Hollidge, B. S. et al. Toll-like receptor 4 mediates blood-brain barrier permeability and disease in C3H mice during Venezuelan equine encephalitis virus infection. Virulence 12, 430–443 (2021).
Salimi, H. et al. Encephalitic alphaviruses exploit caveola-mediated transcytosis at the blood-brain barrier for central nervous system entry. mBio 11, https://doi.org/10.1128/mBio.02731-19 (2020).
Charles, P. C., Walters, E., Margolis, F. & Johnston, R. E. Mechanism of neuroinvasion of Venezuelan equine encephalitis virus in the mouse. Virology 208, 662–671 (1995).
Weaver, S. C. et al. Re-emergence of epidemic Venezuelan equine encephalomyelitis in South America. VEE Study Group. Lancet 348, 436–440 (1996).
Aguilar, P. V. et al. Endemic Venezuelan equine encephalitis in the Americas: hidden under the dengue umbrella. Future Virol. 6, 721–740 (2011).
Steele, K. E. & Twenhafel, N. A. REVIEW PAPER: pathology of animal models of alphavirus encephalitis. Vet. Pathol. 47, 790–805 (2010).
Taylor, K., Kolokoltsova, O., Ronca, S. E., Estes, M. & Paessler, S. Live, attenuated Venezuelan equine encephalitis virus vaccine (TC83) causes persistent brain infection in mice with non-functional alphabeta T-Cells. Front. Microbiol. 8, 81 (2017).
Henderson, B. E., Chappell, W. A., Johnston, J. G. Jr & Sudia, W. D. Experimental infection of horses with three strains of Venezuelan equine encephalomyelitis virus. I. Clinical and virological studies. Am. J. Epidemiol. 93, 194–205 (1971).
Mackenzie, R. M., de Siger, J. & Parra, D. Venezuelan equine encephalitis virus: comparison of infectivity and virulence of strains V-38 and P676 in donkeys. Am. J. Trop. Med. Hyg. 25, 494–499 (1976).
Greene, I. P. et al. Envelope glycoprotein mutations mediate equine amplification and virulence of epizootic Venezuelan equine encephalitis virus. J. Virol. 79, 9128–9133 (2005).
George, J. et al. Prior exposure to Zika virus significantly enhances peak dengue-2 viremia in rhesus macaques. Sci. Rep. 7, 10498 (2017).
Valiant, W. G. et al. Zika convalescent macaques display delayed induction of anamnestic cross-neutralizing antibody responses after dengue infection. Emerg. Microbes Infect. 7, 130 (2018).
Valiant, W. G. et al. Human serum with high neutralizing antibody titres against both Zika and dengue virus shows delayed in vitro antibody dependent enhancement of dengue virus infection. Open Forum Infect. Dis. 5, ofy151 (2018).
Valiant, W. G. et al. Simultaneous coinfection of macaques with Zika and dengue viruses does not enhance acute plasma viremia but leads to activation of monocyte subsets and biphasic release of pro-inflammatory cytokines. Sci. Rep. 9, 7877 (2019).
Burke, C. W. et al. Therapeutic monoclonal antibody treatment protects nonhuman primates from severe Venezuelan equine encephalitis virus disease after aerosol exposure. PLoS Pathog. 15, e1008157 (2019).
Paessler, S. et al. Alpha-beta T cells provide protection against lethal encephalitis in the murine model of VEEV infection. Virology 367, 307–323 (2007).
Yun, N. E. et al. CD4 + T cells provide protection against acute lethal encephalitis caused by Venezuelan equine encephalitis virus. Vaccine 27, 4064–4073 (2009).
Kafai, N. M. et al. Neutralizing antibodies protect mice against Venezuelan equine encephalitis virus aerosol challenge. J. Exp. Med. 219, https://doi.org/10.1084/jem.20212532 (2022).
Tretyakova, I. et al. Venezuelan equine encephalitis vaccine with rearranged genome resists reversion and protects non-human primates from viremia after aerosol challenge. Vaccine 38, 3378–3386 (2020).
Elvin, S. J., Bennett, A. M. & Phillpotts, R. J. Role for mucosal immune responses and cell-mediated immune functions in protection from airborne challenge with Venezuelan equine encephalitis virus. J. Med. Virol. 67, 384–393 (2002).
Berge, T. O. B., I. S. & Tigertt, W. D. Attenuation of Venezuelan equine encephalomyelitis virus by in vitro cultivation in guinea-pig heart cells. Am. J. Epidemiol. 73, 209–218 (1961).
Kinney, R. M. et al. Attenuation of Venezuelan equine encephalitis virus strain TC-83 is encoded by the 5’-noncoding region and the E2 envelope glycoprotein. J. Virol. 67, 1269–1277 (1993).
Kinney, R. M., Johnson, B. J., Welch, J. B., Tsuchiya, K. R. & Trent, D. W. The full-length nucleotide sequences of the virulent Trinidad donkey strain of Venezuelan equine encephalitis virus and its attenuated vaccine derivative, strain TC-83. Virology 170, 19–30 (1989).
White, L. J., Wang, J. G., Davis, N. L. & Johnston, R. E. Role of alpha/beta interferon in Venezuelan equine encephalitis virus pathogenesis: effect of an attenuating mutation in the 5’ untranslated region. J. Virol. 75, 3706–3718 (2001).
Mecham, J. O. & Trent, D. W. A biochemical comparison of the in vitro replication of a virulent and an avirulent strain of Venezuelan encephalitis virus. J. Gen. Virol. 64, 1111–1119 (1983).
Hyde, J. L. et al. A viral RNA structural element alters host recognition of nonself RNA. Science 343, 783–787 (2014).
Burke, D. S., Ramsburg, H. H. & Edelman, R. Persistence in humans of antibody to subtypes of Venezuelan equine encephalomyelitis (VEE) virus after immunization with attenuated (TC-83) VEE virus vaccine. J. Infect. Dis. 136, 354–359 (1977).
Ferguson, J. A., Reeves, W. C., Milby, M. M. & Hardy, J. L. Study of homologous and heterologous antibody response in California horses vaccinated with attenuated Venezuelan equine encephalomyelitis vaccine (strain TC-83). Am. J. Vet. Res. 39, 371–376 (1978).
Fillis, C. A. & Calisher, C. H. Neutralizing antibody responses of humans and mice to vaccination with Venezuelan encephalitis (TC-83) virus. J. Clin. Microbiol. 10, 544–549 (1979).
Jahrling, P. B. & Stephenson, E. H. Protective efficacies of live attenuated and formaldehyde-inactivated Venezuelan equine encephalitis virus vaccines against aerosol challenge in hamsters. J. Clin. Microbiol. 19, 429–431 (1984).
Hart, M. K., Pratt, W., Panelo, F., Tammariello, R. & Dertzbaugh, M. Venezuelan equine encephalitis virus vaccines induce mucosal IgA responses and protection from airborne infection in BALB/c, but not C3H/HeN mice. Vaccine 15, 363–369 (1997).
Phillpotts, R. J. & Wright, A. J. TC-83 vaccine protects against airborne or subcutaneous challenge with heterologous mouse-virulent strains of Venezuelan equine encephalitis virus. Vaccine 17, 982–988 (1999).
Bennett, A. M., Elvin, S. J., Wright, A. J., Jones, S. M. & Phillpotts, R. J. An immunological profile of Balb/c mice protected from airborne challenge following vaccination with a live attenuated Venezuelan equine encephalitis virus vaccine. Vaccine 19, 337–347 (2000).
Marker, S. C. & Ascher, M. S. Specific in vitro lymphocyte transformation with Venezuelan equine encephalitis virus. Cell Immunol. 23, 32–38 (1976).
Jones, L. D., Bennett, A. M., Moss, S. R., Gould, E. A. & Phillpotts, R. J. Cytotoxic T-cell activity is not detectable in Venezuelan equine encephalitis virus-infected mice. Virus Res. 91, 255–259 (2003).
Mathews, J. H., Kinney, R. M., Roehrig, J. T., Barrett, A. D. & Trent, D. W. Murine T-helper cell immune response to recombinant vaccinia-Venezuelan equine encephalitis virus. Vaccine 12, 620–624 (1994).
Pedersen, C. E. Jr, Robinson, D. M. & Cole, F. E. Jr. Isolation of the vaccine strain of Venezuelan equine encephalomyelitis virus from mosquitoes in Louisiana. Am. J. Epidemiol. 95, 490–496 (1972).
Turell, M. J., Ludwig, G. V., Kondig, J. & Smith, J. F. Limited potential for mosquito transmission of genetically engineered, live-attenuated Venezuelan equine encephalitis virus vaccine candidates. Am. J. Trop. Med. Hyg. 60, 1041–1044 (1999).
Tretyakova, I., Tomai, M., Vasilakos, J. & Pushko, P. Live-attenuated VEEV vaccine delivered by iDNA using microneedles is immunogenic in rabbits. Front. Trop. Dis. 3, https://doi.org/10.3389/fitd.2022.813671 (2022).
Centers, A. et al. V4020 Venezuelan equine encephalitis vaccine: mitigating neuroinvasion and reversion through rational design. Viruses 17, https://doi.org/10.3390/v17081136 (2025).
Johnson, D. M. et al. Advanced safety and genetic stability in mice of a novel DNA-launched Venezuelan equine encephalitis virus vaccine with rearranged structural genes. Vaccines 8, https://doi.org/10.3390/vaccines8010114 (2020).
Rossi, S. L. et al. IRES-based Venezuelan equine encephalitis vaccine candidate elicits protective immunity in mice. Virology 437, 81–88 (2013).
Rossi, S. L. et al. IRES-containing VEEV vaccine protects cynomolgus macaques from IE Venezuelan equine encephalitis virus aerosol challenge. PLoS Negl. Trop. Dis. 9, e0003797 (2015).
Reed, D. S. et al. Combined alphavirus replicon particle vaccine induces durable and cross-protective immune responses against equine encephalitis viruses. J. Virol. 88, 12077–12086 (2014).
Hart, M. K. et al. Improved mucosal protection against Venezuelan equine encephalitis virus is induced by the molecularly defined, live-attenuated V3526 vaccine candidate. Vaccine 18, 3067–3075 (2000).
Pratt, W. D., Davis, N. L., Johnston, R. E. & Smith, J. F. Genetically engineered, live attenuated vaccines for Venezuelan equine encephalitis: testing in animal models. Vaccine 21, 3854–3862 (2003).
Reed, D. S. et al. Genetically engineered, live, attenuated vaccines protect nonhuman primates against aerosol challenge with a virulent IE strain of Venezuelan equine encephalitis virus. Vaccine 23, 3139–3147 (2005).
Turell, M. J. & Parker, M. D. Protection of hamsters by Venezuelan equine encephalitis virus candidate. Am. J. Trop. Med. Hyg. 78, 328–332 (2008).
Rao, V., Hinz, M. E., Roberts, B. A. & Fine, D. Environmental hazard assessment of Venezuelan equine encephalitis virus vaccine candidate strain V3526. Vaccine 22, 2667–2673 (2004).
Paessler, S. et al. Replication and clearance of Venezuelan equine encephalitis virus from the brains of animals vaccinated with chimeric SIN/VEE viruses. J. Virol. 80, 2784–2796 (2006).
Coates, E. E. et al. Safety and immunogenicity of a trivalent virus-like particle vaccine against western, eastern, and Venezuelan equine encephalitis viruses: a phase 1, open-label, dose-escalation, randomised clinical trial. Lancet Infect. Dis. 22, 1210–1220 (2022).
Dupuy, L. C. et al. A DNA vaccine for Venezuelan equine encephalitis virus delivered by intramuscular electroporation elicits high levels of neutralizing antibodies in multiple animal models and provides protective immunity to mice and nonhuman primates. Clin. Vaccin. Immunol. 18, 707–716 (2011).
Hannaman, D., Dupuy, L. C., Ellefsen, B. & Schmaljohn, C. S. A Phase 1 clinical trial of a DNA vaccine for Venezuelan equine encephalitis delivered by intramuscular or intradermal electroporation. Vaccine 34, 3607–3612 (2016).
Edelman, R. et al. Evaluation in humans of a new, inactivated vaccine for Venezuelan equine encephalitis virus (C-84). J. Infect. Dis. 140, 708–715 (1979).
Tretyakova, I. et al. Novel DNA-launched Venezuelan equine encephalitis virus vaccine with rearranged genome. Vaccine 37, 3317–3325 (2019).
Paessler, S. & Weaver, S. C. Vaccines for Venezuelan equine encephalitis. Vaccine 27, D80–D85 (2009).
Kendra, J. A. et al. Ablation of programmed -1 ribosomal frameshifting in Venezuelan equine encephalitis virus results in attenuated neuropathogenicity. J. Virol. 91, https://doi.org/10.1128/JVI.01766-16 (2017).