Nine laboratories are proposing cruelty-free alternatives to animal-based testing for protein digestibility in food labeling. Nutrition Insight speaks with the scientist whose team carried out tests using new methods to support this transition, as current US and Canadian regulations rely on using animals for specific food ingredient tests. The new methods come amid a rise in alternative protein innovations.
Supporting the study, the Institute for the Advancement of Food and Nutrition Sciences points out that companies creating new food and ingredients cannot back them with existing data that do not cover these new items. Thus, animal-based digestibility tests become a costly and ethical hurdle.
The researchers suggest that their new cost-effective methods, which follow the Uniform Methods Committee standards, help food formulators make ingredient decisions and processing choices when considering protein digestibility. The Canadian and the US-based laboratories in the study analyzed the same 12 plant and animal protein ingredients and used two non-animal tests to test their reliability.
The study in the Journal of Food Composition & Analysis used pH-drop and pH-stat, which confirm repeatability and stable reproducibility. The pH-drop method determines changes in pH when digestive enzymes break down proteins. The pH-stat method keeps a solution’s pH at a constant level during digestion.
Compared to other animal-testing alternatives
Erin Goldberg, Ph.D., study director for the collaborative trial and research associate at the University of Manitoba, Canada, tells us how the new methods compare to other non-animal tests, what regulators need to see before approving these methods, and how soon companies can start using them.
“pH-drop and pH-stat sit in the practical, near-term lane of the animal-free transition. They are much simpler than organ-on-a-chip systems, but that is exactly their strength. While organ-on-a-chip systems aim to replicate more complex aspects of human physiology, including absorption and cellular responses, they remain technically demanding, costly, and difficult to standardize across laboratories.”
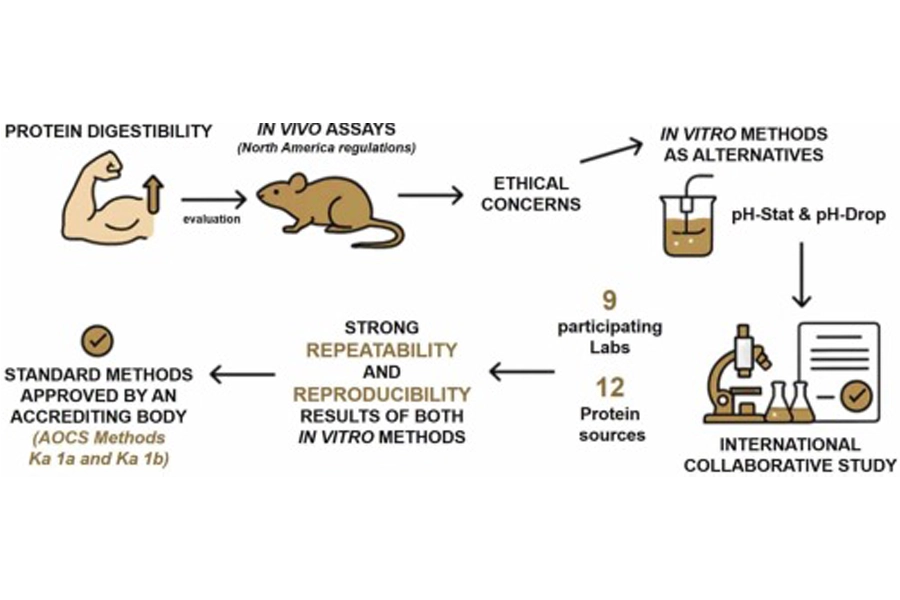 Nine labs prove pH-drop/pH-stat methods replace animal tests for protein digestibility in novel ingredients.“In contrast, pH-drop and pH-stat are simple, fast, and affordable, with demonstrated reproducibility across nine laboratories and formal recognition as Official Methods by the American Oil Chemists’ Society. This positions them as scalable tools for routine digestibility assessment. They align well with established in vivo benchmarks for true fecal protein digestibility and are well-suited for ingredient screening and potential regulatory use,” she explains.
Nine labs prove pH-drop/pH-stat methods replace animal tests for protein digestibility in novel ingredients.“In contrast, pH-drop and pH-stat are simple, fast, and affordable, with demonstrated reproducibility across nine laboratories and formal recognition as Official Methods by the American Oil Chemists’ Society. This positions them as scalable tools for routine digestibility assessment. They align well with established in vivo benchmarks for true fecal protein digestibility and are well-suited for ingredient screening and potential regulatory use,” she explains.
Choosing the lab option
Goldberg notes that more precise methods, such as the Digestible Indispensable Amino Acid Score (DIAAS), which measures ileal amino acid digestibility, are rising. However, these methods endorsed by the UN Food and Agriculture Organization rely on pig models. Even though such methods are more advanced, they are not aligned with the push to eliminate animal testing.
“This highlights an important opportunity to build from validated in vitro methods toward fully animal-free approaches that can support future protein quality frameworks. We don’t need a perfect system (as no method can be perfect). The food industry needs options, and pH drop, pH stat, and InfoGest offer those options,” she says.
“We need a reliable, validated, and implementable step forward as we continue to refine in vitro methods.”
EU labeling requirements on top
Protein labeling has different regulatory requirements across regions, notes Goldberg. In the EU, front-of-pack protein claims are based on protein quantity relative to energy. For instance, 12% of protein can carry a “source of protein” label, while 20% is “high in protein.” However, she says, “there is no requirement to directly measure protein digestibility or amino acid availability.”
“In contrast, Canada and the US incorporate protein quality into labeling, requiring methods such as Protein Digestibility Corrected Amino Acid Score (PDCAAS) to adjust protein content based on digestibility and amino acid composition before claims like ‘source’ or percent daily value can be made.”
“This creates a clear divide between a simpler, quantity-based system in Europe and a more scientifically complex, quality-adjusted framework in North America that reflects how well the body can actually use the protein,” explains Goldberg.
According to an animal testing perspective, she says that the European rules on protein claims are inherently animal-free. This is because front-of-pack claims are solely based on protein quantity relative to energy and do not rely on PDCAAS or DIAAS, which historically have used animal studies.
“This means companies can substantiate protein claims without conducting or relying on rat or pig feeding trials, removing a major ethical and logistical barrier. It also lowers the cost and complexity of compliance, making it easier for plant-based and novel protein innovators to bring products to market without engaging in animal testing,” says Goldberg.
 Animal methods, such as the DIAAS, rely on pig models.“In effect, the EU framework aligns regulatory practice with the broader push to reduce animal use in science, while still maintaining clear and standardized criteria for consumer-facing claims.”
Animal methods, such as the DIAAS, rely on pig models.“In effect, the EU framework aligns regulatory practice with the broader push to reduce animal use in science, while still maintaining clear and standardized criteria for consumer-facing claims.”
Gaining regulatory approval
Goldberg explains that for a regulator to approve in vitro methods for protein labeling, there are five major points to consider.
“First, the method must achieve formal recognition as an accredited standard with a clearly defined protocol, including scope, procedures, and performance criteria. We achieved this in 2025 when our methods were adopted as Official Methods by the American Oil Chemists Society.”
“Second, there must be strong evidence of repeatability and reproducibility across multiple laboratories, which this international collaborative trial demonstrates. Third, regulators need evidence that the method is fit for purpose, meaning that in vitro digestibility values align sufficiently with accepted in vivo benchmarks for relevant food ingredients.”
Moreover, the methods must be proven to be reliable across various food matrices and processing conditions, Goldberg adds. This is especially relevant for plant-based foods, where digestibility varies due to antinutritional factors and processing.
“Finally, a formal policy decision is required to incorporate the method into existing regulatory frameworks, which in North America are still tied to PDCAAS-based approaches for protein claims,” she says.
 Goldberg welcomes food companies to begin using these methods immediately in internal R&D.“We don’t need a perfect system immediately. Regulators and industry need validated, implementable options that can be adopted within existing frameworks while the science continues to evolve.”
Goldberg welcomes food companies to begin using these methods immediately in internal R&D.“We don’t need a perfect system immediately. Regulators and industry need validated, implementable options that can be adopted within existing frameworks while the science continues to evolve.”
The methods worked reliably
Goldberg says that her team’s message is not about which one ingredient performed best, but that the methods worked consistently across 12 proteins — including dairy, egg, soy, pea, cereal flours, and pulses.
“This demonstrates that the methods are robust across a wide range of protein sources rather than being optimized for a narrow subset. The pH-stat method is particularly useful for strongly buffering proteins such as casein and egg white, where maintaining constant pH provides a more controlled measurement.”
“More refined or processed proteins are generally easier to assess due to simpler matrices and stronger reference data. The main challenges arise from matrix effects. Less processed plant materials can contain structural barriers and antinutritional compounds such as protease inhibitors and tannins, and digestibility can change significantly with processing methods like cooking or extrusion,” she details.
“These factors make complex, matrix-bound proteins and highly variable ingredients the most challenging cases for current in vitro methods.”
Goldberg notes that complex food matrices are challenging for in vivo measures of digestibility, as they require the protein to be added into species-specific animal feed. However, this can affect the digestibility of the test protein.
Innovation pathway
Goldberg welcomes food companies to begin using these methods immediately in internal R&D.
“Their low cost, speed, and reproducibility make them well-suited for early-stage ingredient screening, formulation design, and evaluating how processing affects digestibility,” she adds. “However, their use for front-of-pack protein claims depends on regulatory acceptance.”
She notes that current regulations in Canada and the US still rely on PDCAAS-based frameworks, meaning that in vitro methods still cannot be used alone to substantiate labeling claims. “It’s important to note that digestibility is only one component of protein quality.”
“Claims related to ‘complete protein’ require both digestibility data and a complete essential amino acid profile to demonstrate that all indispensable amino acids are present in adequate proportions.”
Therefore, she concludes that these new methods can already be useful for innovation and screening, but future regulatory adoption will determine their use for formal labeling.








