Your support helps us to tell the story
From reproductive rights to climate change to Big Tech, The Independent is on the ground when the story is developing. Whether it’s investigating the financials of Elon Musk’s pro-Trump PAC or producing our latest documentary, ‘The A Word’, which shines a light on the American women fighting for reproductive rights, we know how important it is to parse out the facts from the messaging.
At such a critical moment in US history, we need reporters on the ground. Your donation allows us to keep sending journalists to speak to both sides of the story.
The Independent is trusted by Americans across the entire political spectrum. And unlike many other quality news outlets, we choose not to lock Americans out of our reporting and analysis with paywalls. We believe quality journalism should be available to everyone, paid for by those who can afford it.
Your support makes all the difference.Read more
Five advertisements for health supplements, which made unsubstantiated claims about treating symptoms of menopause, polycystic ovarian syndrome (PCOS), and other women’s hormonal conditions, have been banned.
Products from 222 Balance Me, Lunera, Minerva, and Nova Menopause Vitality were among those prohibited for claiming to prevent, treat, or cure menopause symptoms. Separately, PolyBiotics’ ad and website suggested its supplements could prevent, cure, or treat PCOS.
The Advertising Standards Authority (ASA) confirmed the bans, stating it pays close attention to adverts that could exploit people’s health worries, emotional concerns, or financial pressures.
The most recent rulings followed an AI-powered sweep of health claims in online ads by the watchdog, which it said had revealed emerging and ongoing issues around misleading claims.
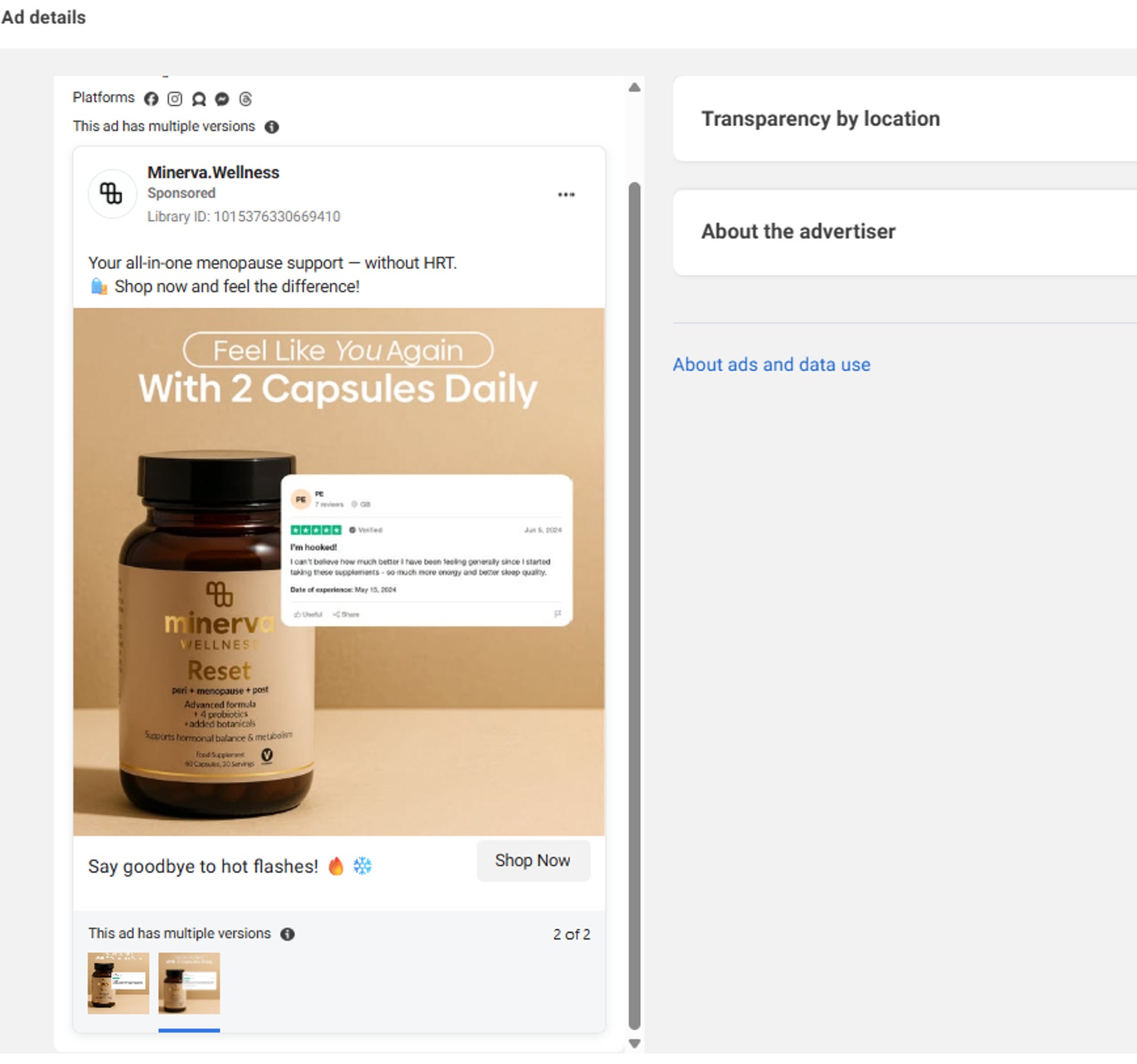 A photo issued by the Advertising Standards Authority (ASA) of a Minerva Wellness Meta advert s (ASA/PA Wire)
A photo issued by the Advertising Standards Authority (ASA) of a Minerva Wellness Meta advert s (ASA/PA Wire)
The ASA said “many” of the claims in the ads were “unacceptable” and had not only broken a number of the authority’s rules but risked misleading vulnerable people, or steering those who needed it away from appropriate medical advice.
222 Collective told the ASA it was a new, founder-run small business and still learning about the requirements of advertising regulations.
The firm acknowledged that wording in the ads may have “inadvertently implied that the product could treat or relieve symptoms such as PMS, menopause-related symptoms, anxiety, bloating, heavy bleeding, or mood disorders”.
They had since been working with Trading Standards to ensure they did not make explicit or implied disease or symptom treatment claims.
Lunera said it accepted that its claims would be understood by consumers to attribute a medicinal property to a food supplement and should not have appeared.
 A photo issued by the Advertising Standards Authority (ASA) of a Nova Relief Meta advert (ASA)
A photo issued by the Advertising Standards Authority (ASA) of a Nova Relief Meta advert (ASA)
PolyBiotics told the ASA it accepted that references to PCOS, ovulation, fertility, cycle regulation, insulin resistance and related symptoms constituted disease treatment or symptom-management claims, which were not permitted for food supplements.
Minerva and Nova did not respond to the ASA’s enquiries.
ASA investigations manager Catherine Drewett said: “When it comes to women’s health, people deserve clear and accurate information.
“Ads making misleading claims about treating symptoms of the menopause, PCOS and other hormonal conditions can cause real harm and today’s rulings hold advertisers to account.
“We’ll continue to monitor this sector closely and we encourage anyone with concerns about an ad they’ve seen to get in touch.”







