Scientists have discovered a potential cure for type 1 diabetes in mice, which could one day transform the treatment of this chronic and often debilitating disease. The study, led by Dr. Judith Shizuru, a professor at Stanford University, demonstrates how scientists created a “blended immune system” in mice that successfully reprogrammed the immune response to prevent the destruction of insulin-producing cells in the pancreas. This discovery could signal a new era in diabetes research and treatment, offering hope to millions affected by this autoimmune condition.
A Step Forward in Diabetes Research
Type 1 diabetes is an autoimmune disease in which the body’s immune system mistakenly attacks and destroys the islet cells in the pancreas that produce insulin. Insulin is vital for regulating blood sugar levels, and without it, people with type 1 diabetes must rely on insulin injections to survive. Over the years, scientists have experimented with various treatments, including islet cell transplants. However, these have been limited by the need for lifelong immunosuppressant drugs to prevent the body from rejecting the transplanted cells.
The latest breakthrough, which involves using stem cells from a donor and transplanting them alongside the islet cells, eliminates the need for these immunosuppressants, offering a more sustainable and promising solution. The researchers believe this technique could eventually lead to a cure, replacing the need for insulin therapy altogether.
“This is potentially a way to cure diabetes,” Dr. John DiPersio, an oncologist at Washington University in St. Louis, told Live Science. “It does represent, in theory, a big step forward.”
While it’s still early days, the potential impact on patients who rely on insulin injections could be transformative.
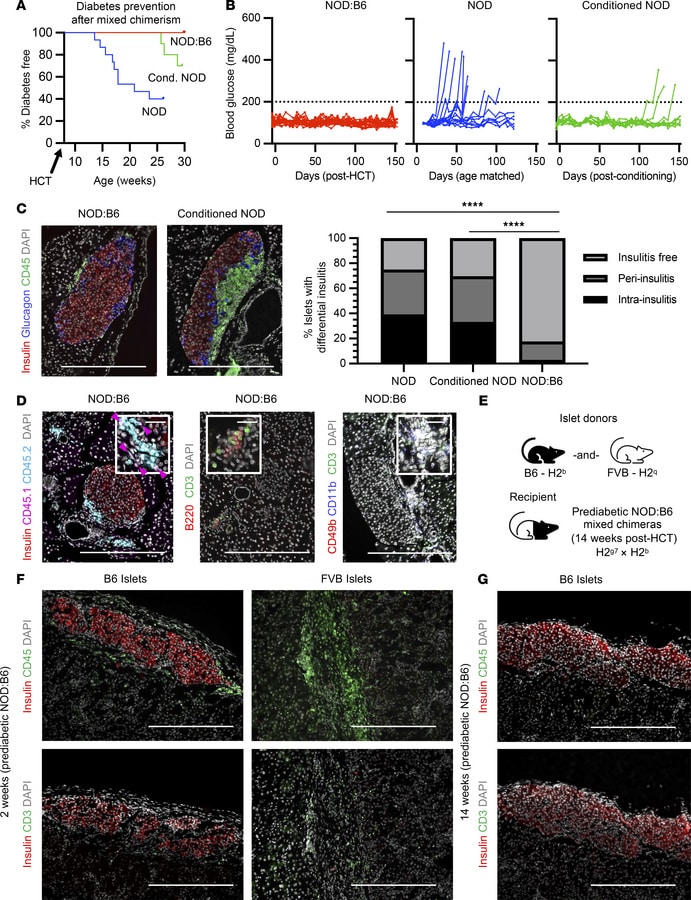 Mixed chimerism prevents autoimmune diabetes and establishes donor-specific islet tolerance.
Mixed chimerism prevents autoimmune diabetes and establishes donor-specific islet tolerance.
(A) Autoimmune diabetes development curves of prediabetic NOD:B6 mixed chimeric mice (n = 19 from 4 independent experiments), conditioned controls (n = 10), and naive NOD mice (n = 15). P < 0.0001 for chimeras vs. naive NOD mice, P < 0.01 for chimeras vs. conditioned controls using log-rank (Mantel-Cox) test. P < 0.0001 for chimeras vs. naive NOD, P < 0.05 for chimeras vs. conditioned controls using Fisher’s exact test. P < 0.05 and no significance for conditioned controls vs. naive NOD mice using Mantel-Cox and Fisher’s exact test, respectively. (B) Nonfasting blood glucose of NOD:B6 chimeras (red) compared with naive NOD mice (blue) and conditioned controls (green). Dotted line indicates normoglycemic threshold (200 mg/dL). (C) Percentage of different stages of insulitis in individual islets and representative histology of pancreas from NOD:B6 mice 20 weeks after HCT (n = 14) compared with naive NOD mice (n = 5) and conditioned controls (n = 10). ****P < 0.0001. Statistical analyses done using χ2 test. (D) Representative pancreatic histology of NOD:B6 chimeras at 20 weeks after HCT stained for insulin, CD45.1 (host), and CD45.2 (donor) or B220 and CD3 or CD49b, CD11b, and CD3 (n = 3-6). Pink arrowheads indicate CD45.1+ immune cells. (E) Experimental transplantation schematic. Prediabetic NOD:B6 mixed chimeras received B6 islets in the left kidney and FVB islets in the opposite kidney at 14 weeks after HCT. (F) FVB and B6 islet grafts 2 weeks after islet transplantation in mixed chimeras stained for insulin and CD3 or CD45 (n = 3). (G) B6 islet grafts 14 weeks after islet transplantation in mixed chimeras stained for insulin and CD3 or CD45 (n = 4). (C–G) Scale bars = 200 μm (C, D, F, and G); 20 μm (D, insets). HCT, hematopoietic cell transplantation.
How the Blended Immune System Works
The key to this new treatment lies in a process called immune “conditioning.” The research team used a combination of antibodies, low-dose radiation, and a drug called baricitinib to reprogram the immune system of the recipient mice. The aim was to create space in the bone marrow for donor stem cells without fully wiping out the host’s immune system. This is crucial because, in previous approaches, eliminating the host’s immune cells entirely left the body vulnerable to infections.
Dr. Shizuru, who led the study, described the process as “musical chairs,” where the donor stem cells must be carefully introduced into the recipient’s immune system. “If you don’t get the recipient stem cells out of the niche, you can’t get the donor cells in,” she explained.
The transplanted stem cells gradually reprogrammed the immune system, allowing it to recognize the transplanted islet cells as part of the body and preventing the immune system from attacking them. The procedure successfully prevented the rejection of the donor cells, allowing the mice to produce insulin for weeks after the transplant.
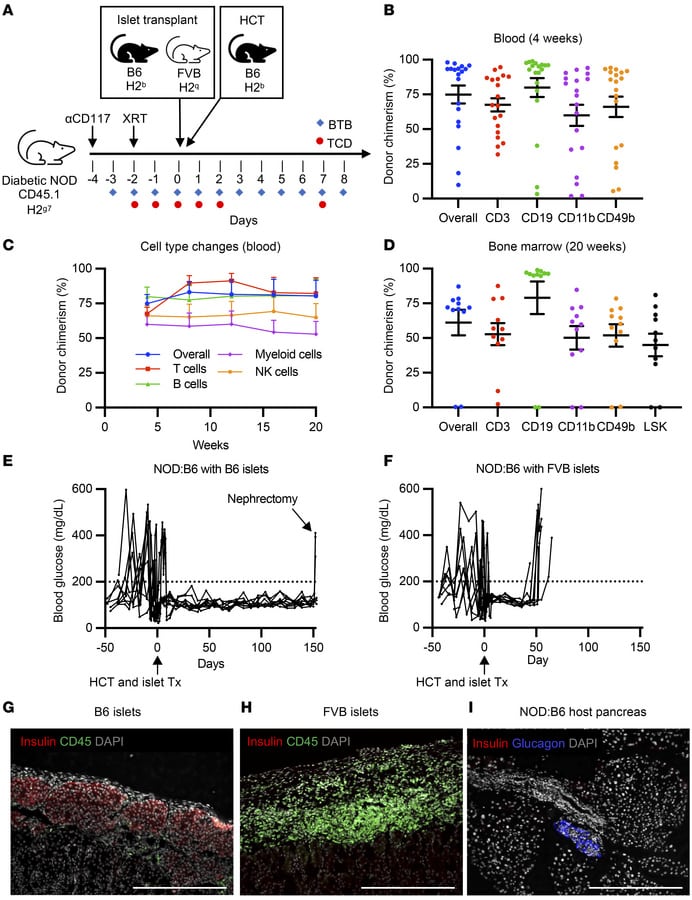 Curing autoimmune diabetes with allogeneic hematopoietic cell and islet transplantation.
Curing autoimmune diabetes with allogeneic hematopoietic cell and islet transplantation.
(A) Experimental conditioning and transplantation timeline. (B) Multilineage chimerism analysis 4 weeks after HCT (n = 19, 6 independent experiments). (C) Longitudinal chimerism analysis of peripheral blood over a 20-week period after HCT (n = 11). (D) Chimerism levels of immune cell subtypes in the bone marrow of mixed chimeras at 20 weeks after HCT (n = 11). (E) Nonfasting blood glucose of mice that received B6 islets and developed mixed chimerism (n = 9). Nephrectomy was conducted at experimental endpoint (n = 3). (F) Nonfasting blood glucose of mixed chimeric mice that received FVB islets (n = 8). (E and F) Dotted line (200 mg/dL) indicates normoglycemia threshold. (G) B6 islet graft 20 weeks after transplantation in NOD:B6 mixed chimera, stained for insulin and CD45 (n = 9). (H) FVB islet graft 8 weeks after transplantation in NOD:B6 mixed chimera, stained for insulin and CD45 (n = 8). (I) NOD:B6 host pancreas 20 weeks after transplant stained for insulin and glucagon (n = 17). (B–E) Data are represented as mean ± SEM. Scale bar: 200 μm. XRT, radiation therapy; HCT, hematopoietic cell transplant; BTB, baricitinib; TCD, T cell depletion.
Ongoing Challenges and Future Hurdles
While this development offers immense promise, there are still many hurdles to overcome before this technique can be applied to humans. One of the key challenges is that the antibodies used in the experiment have not yet been approved for human use. Additionally, finding matching donor cells for both the bone marrow and islet cells remains a significant logistical challenge, as islet cells are already scarce for transplants.
Moreover, maintaining the delicate balance of the “blended” immune system is a complex task. Dr. DiPersio expressed concerns about the long-term viability of this method in humans:
“It’s hard to do that over a long period of time,” he noted. “If the balance shifted, the islets could gradually die or you could get a dangerous tissue rejection reaction.”
Researchers will need to closely monitor patients for signs of rejection or other complications in the long term.
What This Means for the Future of Type 1 Diabetes Treatment
Despite these challenges, this breakthrough offers new hope for people with type 1 diabetes. The possibility of curing the disease, rather than simply managing it with insulin injections, could be a game changer for millions of people worldwide. The technique could one day make it possible for patients to live without the constant need for insulin therapy and avoid the long-term complications that often accompany the disease, such as heart disease, kidney failure, and blindness.
While it will likely take years of further research and clinical trials before this approach is ready for human patients, this discovery represents a significant step toward a cure. If successful, it could alter the landscape of diabetes treatment forever.





