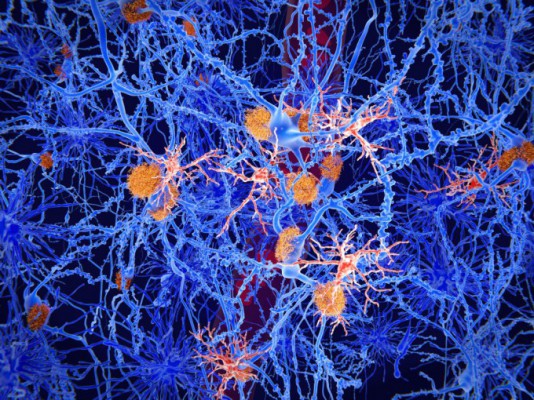
A research team led by brain science professor Um Ji-won at Daegu Gyeongbuk Institute of Science and Technology (DGIST) has identified for the first time in the world how a biological switch in brain immune cells can ease Alzheimer’s disease, offering a potential pathway to repurpose existing drugs.

Daegu Gyeongbuk Institute of Science and Technology (DGIST) professor Um Ji-won / Courtesy of DGIST
The study revealed that the brain’s somatostatin, a neuropeptide that dampens neural activity and directly regulates microglia, which are resident immune cells in the brain, shifting them into a protective state that reduces disease pathology.
When microglia were treated with somatostatin, their ability to clear toxic waste surged while inflammatory signals sharply declined, suggesting that the peptide effectively puts brain immunity back in balance.
“This study is the first to show that the brain neurotransmitter somatostatin can directly reset immune cell states to ease dementia pathology and even improve memory performance,” the professor said.
Alzheimer’s disease is marked by the accumulation of sticky protein clumps building up in the brain, known as amyloid‑beta plaques, and chronic neuroinflammation driven by overactivated microglia. While these cells initially help clear toxic proteins, they can later become hyperactive, releasing inflammatory factors that damage synapses and accelerate cognitive decline.
Um’s team found that somatostatin acts as a key modulator of this process. In experiments using Alzheimer’s disease model mice, boosting somatostatin levels suppressed neuroinflammation and significantly reduced amyloid‑beta plaques.
The treated mice also showed a statistically significant improvement in spatial memory, demonstrating that the effect goes beyond the cellular level to real cognitive recovery.
“So far, research on neurodegenerative diseases has focused mainly on neurons, but this study shows that a neuropeptide can directly reset the state of microglia, allowing integrated control of both neuroinflammation and amyloid pathology,” the research team said.
“Building on this, it should be possible to design next‑generation brain disease and neuroinflammation therapies that precisely target specific state changes in microglia.”
It noted that somatostatin receptor agonists used in the experiments are already approved by the U.S. Food and Drug Administration for other conditions, such as acromegaly. This raises the possibility of accelerating Alzheimer’s treatment development through repurposing existing compounds and extending into new treatment strategies.
The team said its ultimate goal is to build a new framework for therapy that treats synapses and their immune environment as a single, integrated system. By decoding how microglia and other external regulators reshape synaptic function, they hope to pinpoint drug targets that can actually halt the progression of hard‑to‑treat neurodegenerative and psychiatric diseases.