Geng, L. N. et al. 2024 update of the RECOVER-Adult long COVID research index. JAMA 333, 694–700 (2025).
Kuri-Cervantes, L. et al. Comprehensive mapping of immune perturbations associated with severe COVID-19. Sci. Immunol. 5, eabd7114 (2020).
Rahimi, R. A. et al. Advancing lung immunology research: an official American Thoracic Society workshop report. Am. J. Respir. Cell Mol. Biol. 67, e1–e18 (2022).
Talbot, S., Foster, S. L. & Woolf, C. J. Neuroimmunity: physiology and pathology. Annu. Rev. Immunol. 34, 421–447 (2016).
Almanzar, N. et al. Vagal TRPV1+ sensory neurons protect against influenza virus infection by regulating lung myeloid cell dynamics. Sci. Immunol. 10, eads6243 (2025). This paper shows that vagal TRPV1-positive sensory neurons protect the host from lung injury during influenza A infection by skewing pulmonary myeloid cell responses, including those of neutrophils and macrophages, towards less tissue-damaging states, revealing a key neuroimmune mechanism that regulates disease severity during viral respiratory infection.
Hiroki, C. H. et al. Nociceptor neurons suppress alveolar macrophage-induced Siglec-F+ neutrophil-mediated inflammation to protect against pulmonary fibrosis. Immunity 58, 2054–2068.e6 (2025). This paper shows that TRPV1-positive nociceptor sensory neurons protect the host against pulmonary fibrosis by restraining alveolar macrophage-driven VIP/TGFβ signalling and the subsequent recruitment of NET-producing Siglec-F-positive neutrophils, revealing reveal a neuroimmune circuit that limits pathological lung inflammation and fibrotic progression.
McGovern, A. E., Davis-Poynter, N., Farrell, M. J. & Mazzone, S. B. Transneuronal tracing of airways-related sensory circuitry using herpes simplex virus 1, strain H129. Neuroscience 207, 148–166 (2012).
Su, Y. et al. Identification of lung innervating sensory neurons and their target specificity. Am. J. Physiol. Lung Cell. Mol. Physiology 322, L50–L63 (2022).
Dinh, Q. T. et al. Substance P expression in TRPV1 and trkA-positive dorsal root ganglion neurons innervating the mouse lung. Respir. Physiol. Neurobiol. 144, 15–24 (2004).
Kim, S.-H. et al. Mapping of the sensory innervation of the mouse lung by specific vagal and dorsal root ganglion neuronal subsets. eNeuro 9, ENEURO.0026-22.2022 (2022).
Boers, J. E., den Brok, J. L., Koudstaal, J., Arends, J. W. & Thunnissen, F. B. Number and proliferation of neuroendocrine cells in normal human airway epithelium. Am. J. Respir. Crit. Care Med. 154, 758–763 (1996).
Chang, R. B., Strochlic, D. E., Williams, E. K., Umans, B. D. & Liberles, S. D. Vagal sensory neuron subtypes that differentially control breathing. Cell 161, 622–633 (2015). This paper shows that the vagus nerve contains distinct neuronal populations that define labelled lines with different physiological roles: NPY2R-expressing, slow-conducting C-fibres drive rapid, shallow breathing, whereas P2RY1-expressing, fast-conducting A-fibres suppress respiration.
Patil, M. J. et al. Anatomical and functional mapping of vagal nociceptive sensory nerve subsets innervating the mouse lower airways by intersectional genetics. J. Physiol. https://doi.org/10.1113/JP289279 (2025).
Dutschmann, M., Bautista, T. G., Mörschel, M. & Dick, T. E. Learning to breathe: habituation of Hering–Breuer inflation reflex emerges with postnatal brainstem maturation. Respiratory Physiol. Neurobiol. 195, 44–49 (2014).
Nonomura, K. et al. Piezo2 senses airway stretch and mediates lung inflation-induced apnoea. Nature 541, 176–181 (2017).
Schappe, M. S. et al. A vagal reflex evoked by airway closure. Nature 627, 830–838 (2024).
Dubin, A. E. et al. Inflammatory signals enhance Piezo2-mediated mechanosensitive currents. Cell Rep. 2, 511–517 (2012).
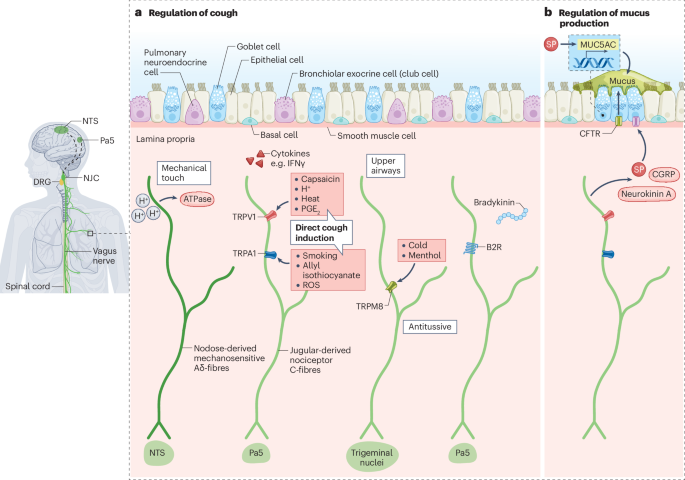
Canning, B. J. & Mori, N. An essential component to brainstem cough gating identified in anesthetized guinea pigs. FASEB J. 24, 3916–3926 (2010).
Chou, Y.-L., Scarupa, M. D., Mori, N. & Canning, B. J. Differential effects of airway afferent nerve subtypes on cough and respiration in anesthetized guinea pigs. Am. J. Physiol. Regul. Integr. Comp. Physiol 295, R1572–R1584 (2008).
Muroi, Y. et al. Selective inhibition of vagal afferent nerve pathways regulating cough using Nav 1.7 shRNA silencing in guinea pig nodose ganglia. Am. J. Physiol. Regul. Integr. Comp. Physiol 304, R1017–R1023 (2013).
Mazzone, S. B. et al. Selective expression of a sodium pump isozyme by cough receptors and evidence for its essential role in regulating cough. J. Neurosci. 29, 13662–13671 (2009).
Driessen, A. K., Farrell, M. J., Mazzone, S. B. & McGovern, A. E. The role of the paratrigeminal nucleus in vagal afferent evoked respiratory reflexes: a neuroanatomical and functional study in guinea pigs. Front. Physiol. 6, 378 (2015).
Brozmanova, M., Mazurova, L., Ru, F., Tatar, M. & Kollarik, M. Comparison of TRPA1-versus TRPV1-mediated cough in guinea pigs. Eur. J. Pharmacol. 689, 211–218 (2012).
Grace, M., Birrell, M. A., Dubuis, E., Maher, S. A. & Belvisi, M. G. Transient receptor potential channels mediate the tussive response to prostaglandin E2 and bradykinin. Thorax 67, 891–900 (2012).
Laude, E. A., Higgins, K. S. & Morice, A. H. A comparative study of the effects of citric acid, capsaicin and resiniferatoxin on the cough challenge in guinea-pig and man. Pulm. Pharmacol. 6, 171–175 (1993).
Birrell, M. A. et al. TRPA1 agonists evoke coughing in guinea pig and human volunteers. Am. J. Respir. Crit. Care Med. 180, 1042–1047 (2009).
Mukhopadhyay, I. et al. Transient receptor potential ankyrin 1 receptor activation in vitro and in vivo by pro-tussive agents: GRC 17536 as a promising anti-tussive therapeutic. PLoS ONE 9, e97005 (2014).
Chou, Y.-L., Mori, N. & Canning, B. J. Opposing effects of bronchopulmonary C-fiber subtypes on cough in guinea pigs. Am. J. Physiol. Regul. Integr. Comp. Physiol 314, R489–R498 (2018).
Weigand, L. A., Ford, A. P. & Undem, B. J. A role for ATP in bronchoconstriction-induced activation of guinea pig vagal intrapulmonary C-fibres. J. Physiol. 590, 4109–4120 (2012).
Deng, Z. et al. IFN-γ enhances the cough reflex sensitivity via calcium influx in vagal sensory neurons. Am. J. Respir. Crit. Care Med. 198, 868–879 (2018).
Driessen, A. K. et al. A role for neurokinin 1 receptor expressing neurons in the paratrigeminal nucleus in bradykinin-evoked cough in guinea-pigs. J. Physiol. 598, 2257–2275 (2020). This paper shows that bradykinin can induce cough through NK1R-expressing neurons in the Pa5, a circuit that is distinct from other cough-inducing pathways, such as ATP-evoked cough.
Drake, M. G., McGarvey, L. P. & Morice, A. H. From bench to bedside: the role of cough hypersensitivity in chronic cough. Clin. Transl. Med. 13, e1343 (2023).
Hsu, C.-C., Lin, Y. S., Lin, R.-L. & Lee, L.-Y. Immediate and delayed potentiating effects of tumor necrosis factor-α on TRPV1 sensitivity of rat vagal pulmonary sensory neurons. Am. J. Physiol. Lung Cell. Mol. Physiology 313, L293–L304 (2017).
Morice, A. H. et al. The effect of gefapixant, a P2X3 antagonist, on cough reflex sensitivity: a randomised placebo-controlled study. Eur. Respir. J. 54, 1900439 (2019).
Gavliakova, S. et al. Antitussive effects of nasal thymol challenges in healthy volunteers. Respir. Physiol. Neurobiol. 187, 104–107 (2013).
Xu, L. et al. Molecular mechanisms underlying menthol binding and activation of TRPM8 ion channel. Nat. Commun. 11, 3790 (2020).
Kuo, H. P., Rohde, J. A., Tokuyama, K., Barnes, P. J. & Rogers, D. F. Capsaicin and sensory neuropeptide stimulation of goblet cell secretion in guinea-pig trachea. J. Physiol. 431, 629–641 (1990).
Jang, D.-Y., Cho, J.-S., Lee, D.-Y., Hong, N.-P. & Ahn, H.-Y. The effect of substance P on nasal mucosal hypersensitivity through sensory nerve axonal reflex. J. Rhinol. 4, 38–42 (1997).
Choi, J. Y. et al. Substance P stimulates human airway submucosal gland secretion mainly via a CFTR-dependent process. J. Clin. Invest. 119, 1189–1200 (2009). This paper shows that SP released from TRPV1-positive nociceptors induces mucus secretion from submucosal glands through the CFTR, which is an important regulatory mechanism for airway mucus homeostasis and is relevant to the pathophysiology of cystic fibrosis.
Sponchiado, M. et al. Overexpression of substance P in pig airways increases MUC5AC through an NF-κB pathway. Physiol. Rep. 9, e14749 (2021).
Talbot, S. et al. Vagal sensory neurons drive mucous cell metaplasia. J. Allergy Clin. Immunol. 145, 1693–1696.e4 (2020).
Yang, D. et al. Nociceptor neurons direct goblet cells via a CGRP–RAMP1 axis to drive mucus production and gut barrier protection. Cell 185, 4190–4205.e25 (2022).
Bessac, B. F. et al. TRPA1 is a major oxidant sensor in murine airway sensory neurons. J. Clin. Invest. 118, 1899–1910 (2008).
Groneberg, D. A., Quarcoo, D., Frossard, N. & Fischer, A. Neurogenic mechanisms in bronchial inflammatory diseases. Allergy 59, 1139–1152 (2004).
Kwong, K. et al. Chronic smoking enhances tachykinin synthesis and airway responsiveness in guinea pigs. Am. J. Respir. Cell Mol. Biol. 25, 299–305 (2001).
Kayalar, Ö et al. Impact of particulate air pollution on airway injury and epithelial plasticity; underlying mechanisms. Front. Immunol. 15, 1324552 (2024).
Wang, J.-C. et al. Nociceptor neurons control pollution-mediated neutrophilic asthma. eLife 13, RP101988 (2026). This paper shows that the neurotrophic factor artemin, released by particulate matter (PM2.5)-activated alveolar macrophages, primes TRPA1-positive nociceptors to release exaggerated amounts of neuropeptides, thereby initiating the recruitment of neutrophils and γδ T cells.
Steib, A. et al. The TRPA1 cation channel is upregulated by cigarette smoke in mouse and human macrophages modulating lung inflammation. Sci. Rep. 15, 10661 (2025).
Luostarinen, S., Hämäläinen, M., Hatano, N., Muraki, K. & Moilanen, E. The inflammatory regulation of TRPA1 expression in human A549 lung epithelial cells. Pulm. Pharmacol. Ther. 70, 102059 (2021).
Taylor-Clark, T. E. & Undem, B. J. Ozone activates airway nerves via the selective stimulation of TRPA1 ion channels: ozone activates TRPA1. J. Physiol. 588, 423–433 (2010).
Balestrini, A. et al. A TRPA1 inhibitor suppresses neurogenic inflammation and airway contraction for asthma treatment. J. Exp. Med. 218, e20201637 (2021).
Mukhopadhyay, I., Kulkarni, A. & Khairatkar-Joshi, N. Blocking TRPA1 in respiratory disorders: does it hold a promise? Pharmaceuticals 9, 70 (2016).
Nova, Z., Skovierova, H. & Calkovska, A. Alveolar-capillary membrane-related pulmonary cells as a target in endotoxin-induced acute lung injury. IJMS 20, 831 (2019).
Lv, X. et al. αCGRP deficiency aggravates pulmonary fibrosis by activating the PPARγ signaling pathway. Genes. Immun. 24, 139–148 (2023). This paper shows that αCGRP deficiency promotes M2 macrophage polarization through the PPARγ–STAT6 pathway, thereby activating type 2 immune responses and accelerating the development of pulmonary fibrosis, thus highlighting a pivotal role for αCGRP in limiting pulmonary fibrosis.
Jiang, H. et al. CGRP is essential for protection against alveolar epithelial cell necroptosis by activating the AMPK/L-OPA1 signaling pathway during acute lung injury. J. Cell. Physiol. 239, e31169 (2024).
Lu, Y.-Z. et al. CGRP sensory neurons promote tissue healing via neutrophils and macrophages. Nature 628, 604–611 (2024).
Yamashita, A. et al. RAMP1 signaling mitigates acute lung injury by distinctively regulating alveolar and monocyte-derived macrophages. IJMS 25, 10107 (2024).
Zhang, R. et al. CGRP alleviates lipopolysaccharide-induced ARDS inflammation via the HIF-1α signaling pathway. Clin. Sci. 139, 373–387 (2025).
Chiu, I. M. et al. Bacteria activate sensory neurons that modulate pain and inflammation. Nature 501, 52–57 (2013).
Ruhl, C. R. et al. Mycobacterium tuberculosis sulfolipid-1 activates nociceptive neurons and induces cough. Cell 181, 293–305.e11 (2020).
Granton, E. et al. Biofilm exopolysaccharides alter sensory-neuron-mediated sickness during lung infection. Cell 187, 1874–1888.e14 (2024).
Pinho-Ribeiro, F. A. et al. Blocking neuronal signaling to immune cells treats streptococcal invasive infection. Cell 173, 1083–1097.e22 (2018).
Pinho-Ribeiro, F. A. et al. Bacteria hijack a meningeal neuroimmune axis to facilitate brain invasion. Nature 615, 472–481 (2023).
Baral, P. et al. Nociceptor sensory neurons suppress neutrophil and γδ T cell responses in bacterial lung infections and lethal pneumonia. Nat. Med. 24, 417–426 (2018). This paper shows that bacterial persistence is prolonged by CGRP released from activated TRPV1-positive nociceptors, which suppresses pro-inflammatory cytokine production and blunts the recruitment and function of neutrophils and γδ T cells.
Joshi, P. R. et al. Lung-innervating nociceptor sensory neurons promote pneumonic sepsis during carbapenem-resistant Klebsiella pneumoniae lung infection. Sci. Adv. 10, eadl6162 (2024).
Lorga, I. et al. CGRP suppresses protective SiglecFhi neutrophil development in neonatal group B Streptococcus pneumonia. Microorganisms 13, 2119 (2025).
Chen, W. et al. Neuronal-activated ILC2s promote IL-17A production in lung γδ T cells during sepsis. Front. Immunol. 12, 670676 (2021).
Aguilar, D. et al. Sensory neurons regulate stimulus-dependent humoral immunity in mouse models of bacterial infection and asthma. Nat. Commun. 15, 8914 (2024).
Verzele, N. A. J. et al. The impact of influenza pulmonary infection and inflammation on vagal bronchopulmonary sensory neurons. FASEB J. 35, e21320 (2021).
Mali, S. S. et al. SARS-CoV-2 papain-like protease activates nociceptors to drive sneeze and pain. Preprint at bioRxiv https://doi.org/10.1101/2024.01.10.575114 (2024).
Jaffal, S. M. & Abbas, M. A. TRP channels in COVID-19 disease: potential targets for prevention and treatment. Chem. Biol. Interact. 345, 109567 (2021).
Hou, Y. et al. Neuropeptide signalling orchestrates T cell differentiation. Nature 635, 444–452 (2024).
Defaye, M. et al. Induction of antiviral interferon-stimulated genes by neuronal STING promotes the resolution of pain in mice. J. Clin. Invest. 134, e176474 (2024).
Donnelly, C. R. et al. STING controls nociception via type I interferon signalling in sensory neurons. Nature 591, 275–280 (2021).
Bin, N.-R. et al. An airway-to-brain sensory pathway mediates influenza-induced sickness. Nature 615, 660–667 (2023). This paper shows that sickness behaviour during influenza infection is induced by the detection of locally produced PGE2 in the upper respiratory tract by PGE2-sensitive sensory neurons, revealing a primary airway-to-brain sensory pathway that mediates systemic sickness responses.
Mann-Nüttel, R., Mandal, S., Armbruster, M., Puttagunta, L. & Forsythe, P. Human pulmonary neuroendocrine cells respond to house dust mite extract with PAR-1 dependent release of CGRP. Allergy 80, 976–985 (2025). This paper shows that human PNECs detect house dust mite allergens with protease activity through PAR1, which induces CGRP release and amplifies SP secretion.
Perner, C. et al. Substance P release by sensory neurons triggers dendritic cell migration and initiates the type-2 immune response to allergens. Immunity 53, 1063–1077.e7 (2020).
Mathur, S. et al. Nociceptor neurons promote IgE class switch in B cells. JCI Insight 6, e148510 (2021).
Sui, P. et al. Pulmonary neuroendocrine cells amplify allergic asthma responses. Science 360, eaan8546 (2018).
Wallrapp, A. et al. Calcitonin gene-related peptide negatively regulates alarmin-driven type 2 innate lymphoid cell responses. Immunity 51, 709–723.e6 (2019). This paper shows that CGRP is a crucial negative regulator of alarmin-activated ILC2s, including those stimulated by IL-33 and TSLP, and may therefore attenuate allergic asthma.
Nagashima, H. et al. Neuropeptide CGRP limits group 2 innate lymphoid cell responses and constrains type 2 inflammation. Immunity 51, 682–695.e6 (2019).
Ye, Y. et al. Neuromedin U promotes human type 2 immune responses. Mucosal Immunol. 15, 990–999 (2022).
Wallrapp, A. et al. The neuropeptide NMU amplifies ILC2-driven allergic lung inflammation. Nature 549, 351–356 (2017).
Moriyama, M. et al. The neuropeptide neuromedin U activates eosinophils and is involved in allergen-induced eosinophilia. Am. J. Physiol. Lung Cell. Mol. Physiology 290, L971–L977 (2006).
Cardoso, V. et al. Neuronal regulation of type 2 innate lymphoid cells via neuromedin U. Nature 549, 277–281 (2017).
Talbot, S. et al. Silencing nociceptor neurons reduces allergic airway inflammation. Neuron 87, 341–354 (2015).
Nussbaum, J. C. et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature 502, 245–248 (2013).
Pascal, M. et al. The neuropeptide VIP potentiates intestinal innate type 2 and type 3 immunity in response to feeding. Mucosal Immunol. 15, 629–641 (2022).
Talbot, J. et al. Feeding-dependent VIP neuron–ILC3 circuit regulates the intestinal barrier. Nature 579, 575–580 (2020).
Pirzgalska, R. M. et al. Neuroepithelial VIP–VIPR1 interactions differentially control enteric type 1 and type 2 immunity. Nat. Immunol. 26, 2244–2255 (2025).
Jakob, M. O. et al. Enteric nervous system-derived VIP restrains differentiation of LGR5+ stem cells toward the secretory lineage impeding type 2 immune programs. Nat. Immunol. 26, 2227–2243 (2025).
Crosson, T. et al. FcεR1-expressing nociceptors trigger allergic airway inflammation. J. Allergy Clin. Immunol. 147, 2330–2342 (2021).
Crosson, T. et al. Cytokines reprogram airway sensory neurons in asthma. Cell Rep. 43, 115045 (2024). This paper shows that negative feedback in type 2 responses is mediated by IL-13Rα1–JAK–STAT6 signalling in vagal nociceptors, which increases expression of the NPY1R and thereby reduces neuronal excitability in response to NPY released by activated M2 macrophages.
Tamari, M. et al. Sensory neurons promote immune homeostasis in the lung. Cell 187, 44–61.e17 (2024).
Lu, Y. et al. Neuropeptide Y associated with asthma in young adults. Neuropeptides 59, 117–121 (2016).
Kormann, M. S. D. et al. G-protein-coupled receptor polymorphisms are associated with asthma in a large German population. Am. J. Respir. Crit. Care Med. 171, 1358–1362 (2005).
Shi, F., Zhang, Y. & Qiu, C. Gene polymorphisms in asthma: a narrative review. Ann. Transl. Med. 10, 711–711 (2022).
Tamayo, J. M., Osman, H. C., Schwartzer, J. J. & Ashwood, P. The influence of asthma on neuroinflammation and neurodevelopment: from epidemiology to basic models. Brain, Behavior, Immun. 116, 218–228 (2024).
Amit, M. et al. Neuro-immune cross-talk in cancer. Nat. Rev. Cancer 25, 573–589 (2025).
Savchuk, S. et al. Neuronal activity-dependent mechanisms of small cell lung cancer pathogenesis. Nature 646, 1232–1242 (2025).
Fnu, T. et al. Sympathetic neurons promote small cell lung cancer through the β2-adrenergic receptor. Cancer Dis. 15, 616–632 (2025).
Zheng, Y. et al. Mechanisms of neural infiltration-mediated tumor metabolic reprogramming impacting immunotherapy efficacy in non-small cell lung cancer. J. Exp. Clin. Cancer Res. 43, 284 (2024).
Zhou, Y. et al. Schwann cells augment cell spreading and metastasis of lung cancer. Cancer Res. 78, 5927–5939 (2018).
Zhou, Y., Li, J., Han, B., Zhong, R. & Zhong, H. Schwann cells promote lung cancer proliferation by promoting the M2 polarization of macrophages. Cell. Immunol. 357, 104211 (2020).
Balood, M. et al. Nociceptor neurons affect cancer immunosurveillance. Nature 611, 405–412 (2022).
McIlvried, L. A., Atherton, M. A., Horan, N. L., Goch, T. N. & Scheff, N. N. Sensory neurotransmitter calcitonin gene-related peptide modulates tumor growth and lymphocyte infiltration in oral squamous cell carcinoma. Adv. Biol. 6, e2200019 (2022).
Smith, J. et al. The neurokinin-1 receptor antagonist orvepitant is a novel antitussive therapy for chronic refractory cough. Chest 157, 111–118 (2020).
Boot, J. D. et al. Effect of an NK1/NK2 receptor antagonist on airway responses and inflammation to allergen in asthma. Am. J. Respir. Crit. Care Med. 175, 450–457 (2007).
Canning, B. J. Central regulation of the cough reflex: therapeutic implications. Pulm. Pharmacol. Ther. 22, 75–81 (2009).
Liu, T. et al. Local sympathetic innervations modulate the lung innate immune responses. Sci. Adv. 6, eaay1497 (2020).
Murray, K. et al. Inhibition of acute lung inflammation by a neuroimmune circuit induced by vagal nerve stimulation. Sci. Adv. 11, eadw7080 (2025).
Li, W. et al. A brain-to-lung signal from GABAergic neurons to ADRB2+ interstitial macrophages promotes pulmonary inflammatory responses. Immunity 58, 2069–2085.e9 (2025).
Moriyama, S. et al. β2-Adrenergic receptor-mediated negative regulation of group 2 innate lymphoid cell responses. Science 359, 1056–1061 (2018).
Borovikova, L. V. et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405, 458–462 (2000).
Su, X., Matthay, M. A. & Malik, A. B. Requisite role of the cholinergic α7 nicotinic acetylcholine receptor pathway in suppressing Gram-negative sepsis-induced acute lung inflammatory injury. J. Immunol. 184, 401–410 (2010).
Chen, H. et al. Postprandial parasympathetic signals promote lung type 2 immunity. Neuron 113, 670–683.e7 (2025).
Cao, Y. et al. Dopamine inhibits group 2 innate lymphoid cell-driven allergic lung inflammation by dampening mitochondrial activity. Immunity 56, 320–335.e9 (2023).