This prospective cohort study provides compelling evidence that pre-treatment vitamin D insufficiency is a strong, independent, and modifiable risk factor for severe paclitaxel-induced peripheral neuropathy in breast cancer patients. Our findings demonstrate that patients with vitamin D levels ≤ 20 ng/mL had a seven-fold increased risk of developing grade 3–4 sensory CIPN compared to those with sufficient levels, even after adjusting for other clinical and treatment-related factors.
Vitamin D insufficiency as a risk factor
The prevalence of vitamin D insufficiency in our cohort (39.3%) is consistent with rates reported in breast cancer populations worldwide, which typically range from 30–50%.19,20 More importantly, the association between low vitamin D levels and severe CIPN was both statistically significant and clinically meaningful. Patients who developed grade 3–4 sensory neuropathy had substantially lower mean vitamin D concentrations (17.5 vs. 24.6 ng/mL, p < 0.001). Additionally, 79.2% of those with severe CIPN had baseline vitamin D insufficiency compared to only 31.7% of those without severe neuropathy.
Our findings corroborate and extend previous retrospective studies examining this relationship. Jennaro et al12. reported that vitamin D deficiency was associated with increased severity of paclitaxel-induced neuropathy in a retrospective analysis of 290 breast cancer patients, though their study did not employ multivariate adjustment for confounding variables. Similarly, Stubblefield et al.21 found associations between low vitamin D levels and chemotherapy-induced neuropathy symptoms, but their analysis was limited by lack of standardized neuropathy assessment tools. Our study strengthens the evidence base by employing a prospective design, validated neuropathy assessment instruments (EORTC QLQ-CIPN20), standardized vitamin D measurement traceable to the VDSP reference method, gold-standard vitamin D measurement (LC-MS/MS), and comprehensive multivariate adjustment. Also, this association mirrors findings in large-scale studies such as Chen et al. (2023)22, where vitamin D insufficiency increased the odds of grade ≥ 3 CIPN (adjusted OR 1.65; p = 0.003).
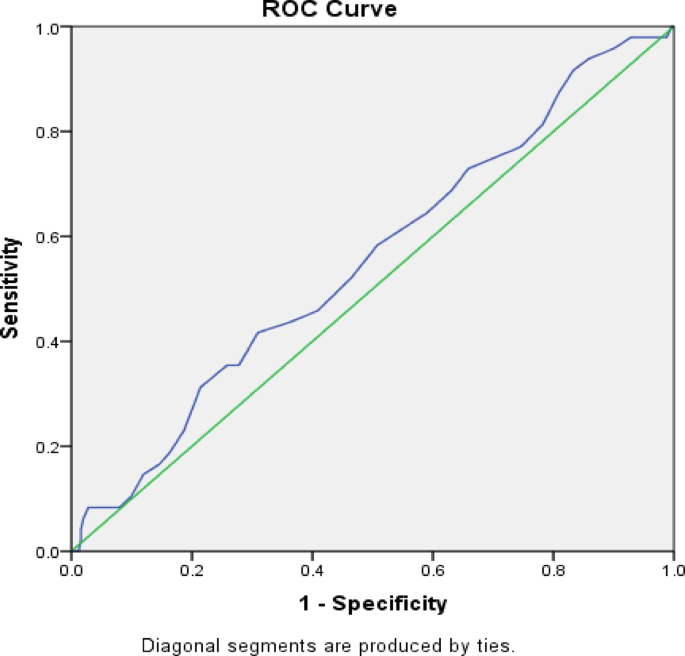
ROC analysis further supported the clinical relevance of vitamin D as a biomarker. Although the area under the curve (AUC) for predicting sensory CIPN was modest (AUC = 0.554), the AUC for motor CIPN was more compelling (AUC = 0.747), with a cutoff value of 26.5 ng/mL yielding 83% sensitivity and 68% specificity (p = 0.038). These findings parallel those of Abutaleb et al. (2024)23, who reported similar sensitivity and specificity metrics, suggesting that a higher vitamin D threshold may be protective.
Multivariate regression analysis in our study found vitamin D insufficiency to be a significant independent predictor of sensory CIPN (OR = 7.0, 95% CI: 3.27–14.97; p < 0.001). This mirrors the adjusted findings from Sun et al. (2024)24, who reported a sixfold increase in CIPN risk among vitamin D-deficient patients (OR = 6.214; 95% CI: 2.308–16.729). The Sun et al. study also identified additional risk factors such as age ≥ 60, cumulative paclitaxel dose > 900 mg, hypertension, and depression. While we did not find significant differences in age or BMI, menopausal status was significantly associated with CIPN in our cohort (p = 0.01), possibly due to hormonal influences on pain pathways or nutrient metabolism.
The lack of significant associations between disease characteristics (ER/PgR, HER2, nodal status) and CIPN in our study is consistent with Ghoreishi et al. (2018)25, who instead identified PR+ status, age, and body surface area (BSA) as influential. Though BSA data was not captured in our cohort, BMI did not show a significant effect, potentially due to the homogeneity in BMI distribution (mean = 29.4 ± 5.4).
The work by Zhang et al. (2024)26 supports the biological plausibility of our findings. They found a negative correlation between serum vitamin D levels and paclitaxel-induced neuropathic pain (Spearman r = − 0.324, p < 0.001). Furthermore, their study documented that vitamin D-deficient patients had more significant oxidative stress and glutathione depletion, mechanisms implicated in CIPN pathophysiology.
The biological plausibility of vitamin D’s neuroprotective role is supported by multiple mechanistic pathways. Vitamin D receptors are widely expressed in peripheral nervous system tissues, including dorsal root ganglia neurons, Schwann cells, and axons.27 Vitamin D has been shown to regulate nerve growth factor synthesis, modulate inflammatory cytokine production, reduce oxidative stress, and promote myelin repair—all processes relevant to chemotherapy-induced neurotoxicity.28,29 Additionally, vitamin D deficiency has been associated with increased susceptibility to peripheral neuropathy in other contexts, including diabetic neuropathy and chronic alcohol use,¹¹ further supporting its role in peripheral nerve health. Furthermore, our longitudinal data highlights that vitamin D insufficiency is not only associated with the acute onset of severe CIPN but also with its long-term persistence. The significantly higher rate of persistent grade ≥ 2 symptoms at 12 months in the insufficient group, along with the observation of the coasting phenomenon in nearly 30% of severe cases, underscores the durable impact of baseline micronutrient status on nerve recovery and chronic morbidity.
Interpretation of risk association versus discriminatory performance
It is important to contextualize the apparent discrepancy between the strong multivariable Odds Ratio (OR = 6.72) and the limited discriminatory performance (AUC = 0.554) for sensory CIPN. While an OR reflects the strength of an independent association after adjusting for covariates, the AUC measures the standalone ability of a continuous biomarker to perfectly discriminate cases from non-cases. In multifactorial conditions like CIPN, it is well-established that a biomarker can be a highly significant risk factor without possessing the necessary sensitivity and specificity to serve as an isolated screening tool30. Therefore, our findings indicate that while pre-treatment vitamin D insufficiency is a critical risk-modifying factor that increases susceptibility to neurotoxicity, it should be utilized in conjunction with other clinical variables (e.g., dosing schedule) for optimal risk stratification, rather than as a solitary diagnostic predictor.
Treatment schedule and neuropathy risk
Consistent with previous literature,31 we found that paclitaxel administration every 2 weeks was associated with increased risk of severe CIPN compared to weekly dosing (adjusted OR = 2.50, p = 0.009). This finding likely reflects differences in peak plasma concentrations and exposure duration, with higher peak levels potentially contributing to greater neurotoxicity. The interaction between dosing schedule and vitamin D status warrants further investigation, as it is possible that adequate vitamin D levels might mitigate some of the neurotoxic effects associated with less frequent, higher-dose administration.
Other risk factors
Our analysis identified postmenopausal status as a potential risk factor for severe CIPN, with a trend toward significance in multivariate analysis (adjusted OR = 1.88, p = 0.075). This association may reflect age-related changes in peripheral nerve function, hormonal influences on nerve health, or higher prevalence of comorbidities in postmenopausal women. Previous studies have yielded mixed results regarding menopausal status and CIPN risk,32 suggesting that this relationship may be modified by other factors such as hormone replacement therapy use or time since menopause.
Notably, we did not find significant associations between CIPN risk and age, BMI, tumor characteristics (nodal status, hormone receptor status, HER2 status), or baseline laboratory parameters. The lack of association with age contrasts with some previous studies33 but may reflect the relatively narrow age range in our cohort (mean 40.6 years) and the predominance of premenopausal and early postmenopausal women.
Clinical implications
The identification of vitamin D insufficiency as a modifiable risk factor for severe CIPN has important clinical implications. First, our findings suggest that routine pre-treatment vitamin D screening should be considered for all breast cancer patients scheduled to receive neurotoxic chemotherapy. Given the high prevalence of vitamin D insufficiency and the substantial impact on neuropathy risk, such screening could identify a large proportion of high-risk patients who might benefit from targeted interventions.
Second, vitamin D supplementation represents a potentially low-cost, safe, and widely available candidate preventive strategy pending randomized trial confirmation. While our observational study cannot directly demonstrate that vitamin D supplementation prevents CIPN, the strength of the association and supporting mechanistic evidence provide a strong rationale for randomized controlled trials evaluating vitamin D repletion as a neuroprotective intervention. Supplementation regimens should aim to achieve and maintain vitamin D levels > 30 ng/mL, which is considered optimal for non-skeletal health benefits.34.
Third, our findings may help refine risk stratification for CIPN. Patients identified with vitamin D insufficiency and other risk factors (such as biweekly dosing schedules or postmenopausal status) could be candidates for enhanced monitoring, dose modifications, or enrollment in clinical trials of neuroprotective agents. This personalized approach could optimize the balance between treatment efficacy and tolerability.
Furthermore, our additional analyses demonstrate that vitamin D insufficiency not only predicts severe progression but also significantly increases the risk of initial CIPN onset (Grade ≥ 1) and clinically meaningful worsening (Grade ≥ 2). Predicting the risk of worsening to Grade ≥ 2 holds particular clinical utility, as it represents the threshold at which symptoms begin to impair daily life, thereby guiding the intensity of proactive neurological monitoring.
Given the limited standalone discriminatory performance (AUC = 0.554), vitamin D should be incorporated into a composite risk stratification approach alongside other clinical variables rather than used as a standalone predictive biomarker.
Strengths and limitations
Our study has several notable strengths. The prospective design with systematic, protocol-defined assessments minimizes recall bias and ensures comprehensive data collection. A critical strength is the use of the Elecsys Vitamin D Total II assay, which is standardized against the ID-LC-MS/MS Reference Measurement Procedure in accordance with the Vitamin D Standardization Program (VDSP). This ensures that our measurements are traceable to the gold standard, addressing common concerns regarding immunoassay variability. Additionally, the EORTC QLQ-CIPN20 is a well-validated patient-reported outcome measure that captures patients’ experiences with neuropathy symptoms. Finally, our multivariate analysis adjusted for key confounding variables, strengthening causal inference.
However, several limitations warrant consideration. First, this was a single-center study conducted in Egypt, and the cohort was relatively young (mean age 40.6 years) compared to many real-world breast cancer populations where the median age at diagnosis is typically 55–65 years. This predominance of premenopausal and early postmenopausal women may have influenced both vitamin D metabolism and neuropathy susceptibility, and caution should be exercised when extrapolating these findings to older patient populations. Second, we did not implement vitamin D supplementation as part of the study protocol, preventing a direct assessment of whether correcting vitamin D insufficiency prevents CIPN. Third, while we excluded patients with known pre-existing neuropathy, subclinical peripheral nerve dysfunction may have been present in some participants and could have modified their response to paclitaxel. Fourth, we did not measure vitamin D binding protein or assess vitamin D receptor polymorphisms, which influence vitamin D bioavailability and activity. Fifth, the relatively short follow-up period (12 weeks during active treatment) may not fully capture the long-term natural history of CIPN, though our extended monitoring through 24 months will provide additional insights. Sixth, the analysis of motor CIPN was severely limited by the small number of events (n = 6, 2.0%), resulting in an underpowered ROC analysis (observed power ~ 42% at α = 0.05). Consequently, residual confounding from unmeasured variables—including overall nutritional status and physical activity levels—cannot be entirely excluded, and vitamin D insufficiency may represent either a direct causal factor or a surrogate marker for broader patient vulnerability.
Future research directions
Our findings highlight several important avenues for future research. First, randomized controlled trials are urgently needed to determine whether vitamin D supplementation can prevent or reduce the severity of paclitaxel-induced neuropathy. Such trials should employ adequate vitamin D dosing regimens (likely 2000–4000 IU daily or weekly equivalent doses) initiated several weeks before chemotherapy to allow for repletion of vitamin D stores. Target levels of 30–40 ng/mL should be achieved prior to chemotherapy initiation.
Second, mechanistic studies are needed to elucidate the specific pathways through which vitamin D exerts neuroprotective effects in the context of taxane exposure. This could include assessment of inflammatory biomarkers, oxidative stress markers, nerve growth factors, and evaluation of vitamin D’s effects on paclitaxel-induced mitochondrial dysfunction and microtubule disruption in peripheral neurons.
Third, investigation of gene-environment interactions between vitamin D receptor polymorphisms and vitamin D status may help identify patient subgroups most likely to benefit from supplementation. Similarly, studies examining whether vitamin D status modifies the relationship between paclitaxel pharmacokinetics and the risk of neuropathy could provide insights into dose optimization strategies.
Fourth, extension of our findings to other neurotoxic chemotherapy agents (platinum compounds, vinca alkaloids, proteasome inhibitors) would help establish whether vitamin D’s neuroprotective effects are specific to taxanes or represent a broader phenomenon applicable to multiple drug classes.
Fifth, the repeated CIPN assessments in our cohort provide a rich longitudinal dataset amenable to trajectory modeling approaches, such as latent class growth analysis or group-based trajectory modeling. Such analyses could identify distinct patterns of neuropathy evolution and patient subgroups with divergent trajectories, potentially refining risk stratification beyond dichotomized severity endpoints. We plan to pursue these analyses in a dedicated follow-up study.
Ultimately, economic analyses evaluating the cost-effectiveness of universal vitamin D screening and supplementation compared to targeted approaches or no intervention would inform implementation strategies and health policy decisions.