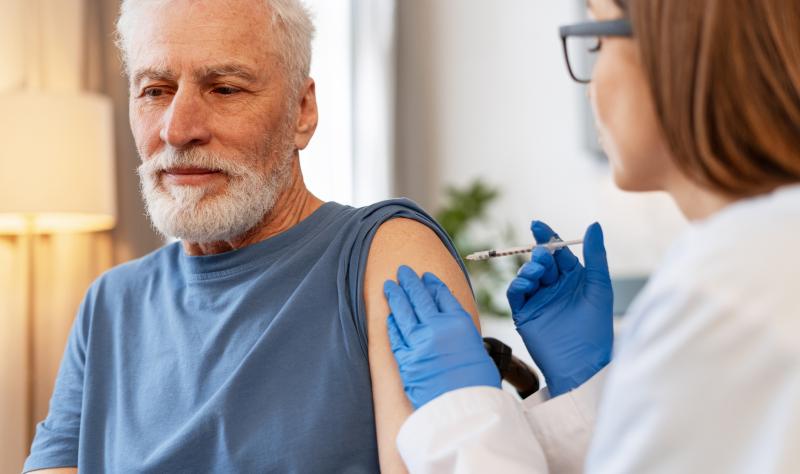
Giving a respiratory syncytial virus (RSV) vaccine at the same time as a shingles vaccine produces a similar immune response as spacing the shots a month apart, according to a phase 3 clinical trial published this week in Clinical Infectious Diseases. No safety concerns were identified with the co-administered vaccines.
The randomized, open-label study included 530 adults aged 50 years and older in the United States and Canada. Participants either received the adjuvanted RSV prefusion F protein-based vaccine (RSVPreF3) and the adjuvanted recombinant zoster vaccine (RZV) at the same visit or received them separately, one month apart.
Same-day vaccination does not blunt immune response
The primary goal of the trial was to determine whether co-administration would lessen the body’s immune response to the vaccines. The findings suggest it did not. When the shots were given at the same time, the antibody responses to RSV-A and RSV-B, as well to varicella zoster virus (which causes shingles), were non-inferior to vaccines given sequentially.
A month after getting the RSV vaccine, people who got the vaccines together had about the sane immune response as those who got them separately. For RSV-A, the co-administered group’s neutralizing antibody levels were about 10% higher (geometric mean titer [GMT], 1.1). For RSV-B, antibody levels were essentially identical to those when given separately (GMT, 1.0). After the full two-dose series, people who got the shingles vaccine had about a 20% higher antibody response (GMT, 1.2).
Both groups showed strong increases in antibody levels from baseline, with serologic response rates of roughly 75% to 80% for RSV and over 90% for shingles.
Mild, short-lived side effects in both groups
Adverse events were common but generally mild and short-lived. Within seven days of vaccination, 77.7% of participants in the co-administered group reported injection-site reactions, compared with 72.7% in the sequential group. Systemic symptoms like fatigue, muscle aches, and headache were reported by about 80% of participants in both groups. Most symptoms resolved within two days.
Roughly 23% of participants in the group that got both vaccines simultaneously reported at least one unexpected side effect within a month of getting the vaccine, compared with about 30% in the group that got them separately.
Serious side effects were uncommon, occurring in about 5% of the co-administered group and 2% of the sequential group, and none were fatal or linked to the vaccines. No participants developed serious neurologic conditions such as Guillain-Barre syndrome or other nerve disorders.
Potential for greater uptake
The study’s strengths include its relatively large sample size and the inclusion of adults aged 50 years and older, allowing the researchers to look at the effects in a broader population. Study limitations include possible reporting bias because of the open-label design and the study being conducted exclusively in North American populations, which limited the generalizability of the findings.
RSV and shingles both pose substantial risks to older adults. The findings suggest that administering the two vaccines at the same visit could improve uptake and reduce the number of healthcare visits without compromising effectiveness.