Abstract
The concept of “trained immunity,” which refers to a type of long-term immunological memory within innate cells, has significantly challenged the traditional division between the non-specific innate and antigen-specific adaptive immune systems. Simultaneously, it is now understood that the resolution of inflammation is not just a passive return to homeostasis but rather an active, strictly regulated biological process. This comprehensive review synthesizes these evolving concepts by hypothesizing that exercise, through its pleiotropic effects on cellular metabolism, may strongly stimulate both the active resolution of inflammation and the formation of innate immunological memory via epigenetic control (emerging evidence from non-exercise models like Bacillus Calmette-Guérin (BCG) vaccination and β-glucan exposure suggests parallels, but direct causation in exercise remains associative). The review describes the biological foundations of this relationship, including the metabolic changes that define trained immunity and the epigenetic reprogramming of myeloid progenitors. It explores the central role of myokines, particularly Interleukin-6 (IL-6), which act as critical mediators, steering the immune response toward a pro-resolving phenotype, while acknowledging contributions from other factors such as IL-15, catecholamines, and neural signals. Furthermore, the review outlines how exercise promotes the synthesis of specialized pro-resolving mediators (SPMs) and enhances efferocytosis, a key cellular mechanism for restoring tissue homeostasis. By providing a unifying framework, this analysis offers a mechanistic explanation for the profound health benefits of exercise, from enhanced immunosurveillance to the prevention of chronic inflammatory diseases. The review concludes by highlighting significant knowledge gaps and advocating for the use of multi-omics and in vivo models to fully elucidate this complex nexus and translate these discoveries into novel therapeutic strategies.
Introduction
The traditional understanding of the immune system is based on a clear division of labor: the adaptive branch creates a slower, antigen-specific memory response, whereas the innate branch offers a quick, non-specific first line of defense (1–4). After an initial stimulus, innate immune cells such as monocytes, macrophages, dendritic cells, neutrophils, natural killer (NK) cells, and tissue-resident macrophages undergo long-term functional reprogramming, allowing them to respond more strongly to a subsequent, unrelated assault (1, 5, 6). The physiological mechanisms that drive this enhanced responsiveness are multifaceted, involving a complex interplay between epigenetic and metabolic changes (1, 7, 8). Importantly, trained immunity is induced in the bone marrow, where long-lived progenitors like hematopoietic stem cells (HSCs) are reprogrammed to propagate the trained phenotype to progeny cells. At the same time, it is becoming more well recognized that inflammation resolution is a very active process as opposed to a passive one (9). Instead of simply dissipating, inflammation is actively curtailed and tissue homeostasis restored by a distinct class of molecules known as SPMs (9–11). These lipid mediators, derived from essential fatty acids, not only dampen the inflammatory cascade but also actively promote host defense and the clearance of pathogens and cellular debris (9, 12, 13). The dual nature of these processes—both anti-inflammatory and pro-host defense—distinguishes them from classical immunosuppression (12).
The relationship between exercise and immune function mirrors this complexity, defying a simple linear model (14). Early research focused on basic changes in immune cell counts (15, 16). By the 1980s, a more complex understanding had developed, represented by the “J-shaped” curve, which postulates that while prolonged, intense training can increase the incidence of minor infections, especially of the upper respiratory tract, moderate exercise lowers the risk of infection when compared to a sedentary lifestyle (15, 17, 18). However, there is still much disagreement on the “open window” theory, which postulates a brief period of immunosuppression following intense exercise. However, there is evidence that suggests acute exercise may actually improve immune surveillance (16, 19, 20). This review seeks to unify these seemingly disparate fields by proposing a hypothesis-driven mechanistic framework in which exercise is posited to serve as a potent physiological modulator of innate immunity (21, 22). The main argument is that exercise actively promotes the resolution of inflammation and the development of trained immunity (23, 24). Evidence suggests how exercise-induced signals, particularly myokines released from contracting skeletal muscle, orchestrate the epigenetic and metabolic reprogramming of innate immune cells (25–27). Concurrently, Exercise increases vital cellular functions including efferocytosis and encourages the production of pro-resolving lipid mediators (28, 29). This dual-pronged action provides a comprehensive explanation for the pleiotropic health benefits of exercise, from enhanced pathogen defense to the prevention of chronic inflammatory diseases (28, 30, 31). By dissecting these molecular and cellular pathways, this review aims to establish exercise as a powerful, non-pharmacological tool for immunomodulation.
The foundational function of innate immune memory
The long-held belief that memory is the sole realm of the adaptive immune system is being challenged by the idea of trained immunity, which represents a fundamental shift in immunological theory (32). This section provides a detailed exposition of innate immune memory, its molecular basis, and its critical distinction from adaptive immunity.
Defining trained immunity and its mechanisms
Long-term, non-specific improvement of innate immune cell function is a hallmark of trained immunity, allowing for a stronger reaction to later, frequently unrelated immunological stimuli (1, 33, 34). This stands in stark contrast to adaptive immunity, which is mediated by T and B cells and is extremely specific to a given antigen (1, 35, 36). A wide variety of stimuli, such as endogenous sterile stimuli like oxLDL and microbial ligands like β-glucan and BCG vaccine, can cause the induction of trained immunity (1, 37).
How this memory endures in innate immune cells with comparatively short lifespans, including neutrophils and monocytes, is a key conundrum of trained immunity (1, 38, 39). Reprogramming long-lived myeloid progenitor cells is the solution to this dilemma. The initial training stimulus leaves an immunological mark on bone marrow-derived HSCs in addition to influencing mature, circulating immune cells (1, 40, 41). These reprogrammed HSCs then generate functionally adapted progeny cells that maintain the “trained” phenotype for months to years after the initial exposure (1, 41, 42). This fundamental mechanism explains how the clinical effects of an intervention, such as BCG vaccination, can last for at least five years, as seen in studies on children in Uganda (1, 43, 44). This suggests that for exercise to induce lasting trained immunity, it must also influence the intricate regulatory network of HSCs, a significant area for future investigation [emerging evidence from animal models shows exercise affects HSC proliferation and lineage commitment, but direct links to trained immunity reprogramming remain associative and require further validation (45, 46)]. Furthermore, the establishment of durable trained immunity requires precise negative regulation to prevent pathological overactivation. TRIM29 (tripartite motif-containing protein 29), an E3 ubiquitin ligase, serves as a key checkpoint that simultaneously enables training of innate immunity and actively tames inflammation. TRIM29 is rapidly induced in alveolar macrophages and mucosal epithelial cells upon pathogen encounter and functions as a selective negative regulator of innate responses (47). It suppresses type-I interferon production, NF-κB activation, and proinflammatory cytokine release by directly ubiquitinating and targeting critical signaling adapters—such as STING (in DNA-sensing pathways) and MAVS (in RNA-sensing pathways)—for proteasomal degradation (48, 49). By dampening these pathways, TRIM29 prevents excessive inflammatory storms and tissue damage while still permitting the epigenetic and metabolic reprogramming that underlies long-term innate immune memory. This balanced “train-and-tame” action is particularly relevant in mucosal sites where repeated microbial exposure occurs, and it provides a molecular explanation for how physiological stressors (including exercise) can promote beneficial trained immunity without tipping into chronic inflammation. Animal models, particularly in mice, have demonstrated that exercise enhances HSC proliferation and function. For instance, endurance exercise mobilizes developmentally early stem cells into peripheral blood and increases their numbers in bone marrow, potentially improving tissue regeneration (50). Additionally, exercise training increases the percentage of HSCs in the vascular niche of bone marrow and promotes cell proliferation, which may contribute to sustained immune adaptations (51). In models of enhanced physical activity, such as transgenic mice with increased muscle activity, HSCs exhibit reduced aging-related decline, with improved pool size and functional capacity (52). These effects on HSC progeny could underpin exercise’s role in propagating trained immunity systemically, though direct epigenetic links in exercise contexts warrant further study.
Epigenetic and metabolic reprogramming as core drivers
The functional changes that define trained immunity are underpinned by a profound intracellular rewiring of gene transcription and metabolism (1, 53, 54). These two pathways must interact to induce and maintain innate immunological memory (1, 55).
Epigenetic mechanisms
Epigenetic reprogramming serves as the master switch for trained immunity. These changes can endure through cell divisions and modify gene expression without altering the underlying DNA sequence (56, 57). Histone alterations, which control DNA’s accessibility to transcriptional machinery, are essential to this process (58). Specifically, trained immunity is characterized by the acquisition of permissive marks such as H3K27ac marks at distal enhancers, H3K4me3 marks at gene promoters and H3K4me1 at enhancers (1, 59–61). However, repressive marks like H3K9me2 and H3K9me3 also contribute by silencing non-inflammatory genes, ensuring a balanced chromatin landscape. These changes result in euchromatin, a more open chromatin structure that makes it easier for transcription factors and other regulatory proteins to access and control gene expression (57, 62, 63). Following additional stimulation, this leads to increased transcription of inflammatory genes like TNFα and IL6 (58, 64). Exercise can directly influence this epigenetic landscape in immune cells. For example, early-life regular exercise in mice induces immunometabolic epigenetic modifications, enhancing anti-inflammatory immunity in middle age via changes in histone acetylation and DNA methylation patterns that overlap with trained immunity signatures (65). In humans, resistance exercise alters epigenetic marks in leukocytes, affecting pathways related to energy metabolism and inflammation, potentially distinct from or synergistic with trained immunity (66). These findings provide a direct link between exercise and long-term epigenetic reprogramming, though more studies are needed to delineate exercise-specific effects.
Metabolic reprogramming (immunometabolism)
The metabolic state of an immune cell is not a fixed characteristic; rather, it is a dynamic program that is modulated in response to immunological stimuli (67–69). While a shift from oxidative phosphorylation (OXPHOS) to aerobic glycolysis (often referred to as a Warburg-like effect) is associated with some aspects of trained immunity, this is not a universal feature. Instead, trained immunity involves increased activity in multiple metabolic pathways, including glycolysis, OXPHOS, the tricarboxylic acid (TCA) cycle, amino acid metabolism (e.g., glutaminolysis), and lipid metabolism (54). These diverse changes provide the energy and precursors needed for robust immune responses and epigenetic remodeling (67, 70). This metabolic shift is regulated by key metabolic sensors, especially HIF-1α (67, 71, 72). The activation of HIF-1α upregulates the expression of numerous glucose transporters and glycolytic enzymes, thereby increasing the overall rate of glycolysis (67, 73). This metabolic reprogramming is not merely a consequence of trained immunity but is a prerequisite for it (74). The central connection is that certain metabolites can directly affect the enzymes involved in epigenetic remodeling (75, 76). For example, S-adenosylmethionine (SAM) serves as a methyl donor for DNA methyltransferases, such as DNMT3A (77). This direct, metabolic-fueled link to epigenetic changes suggests a powerful causal pathway: any intervention, such as exercise, that alters cellular metabolism could potentially initiate or modulate trained immunity by providing the necessary metabolic substrates for these long-term epigenetic changes (78–80) (Figure 1).
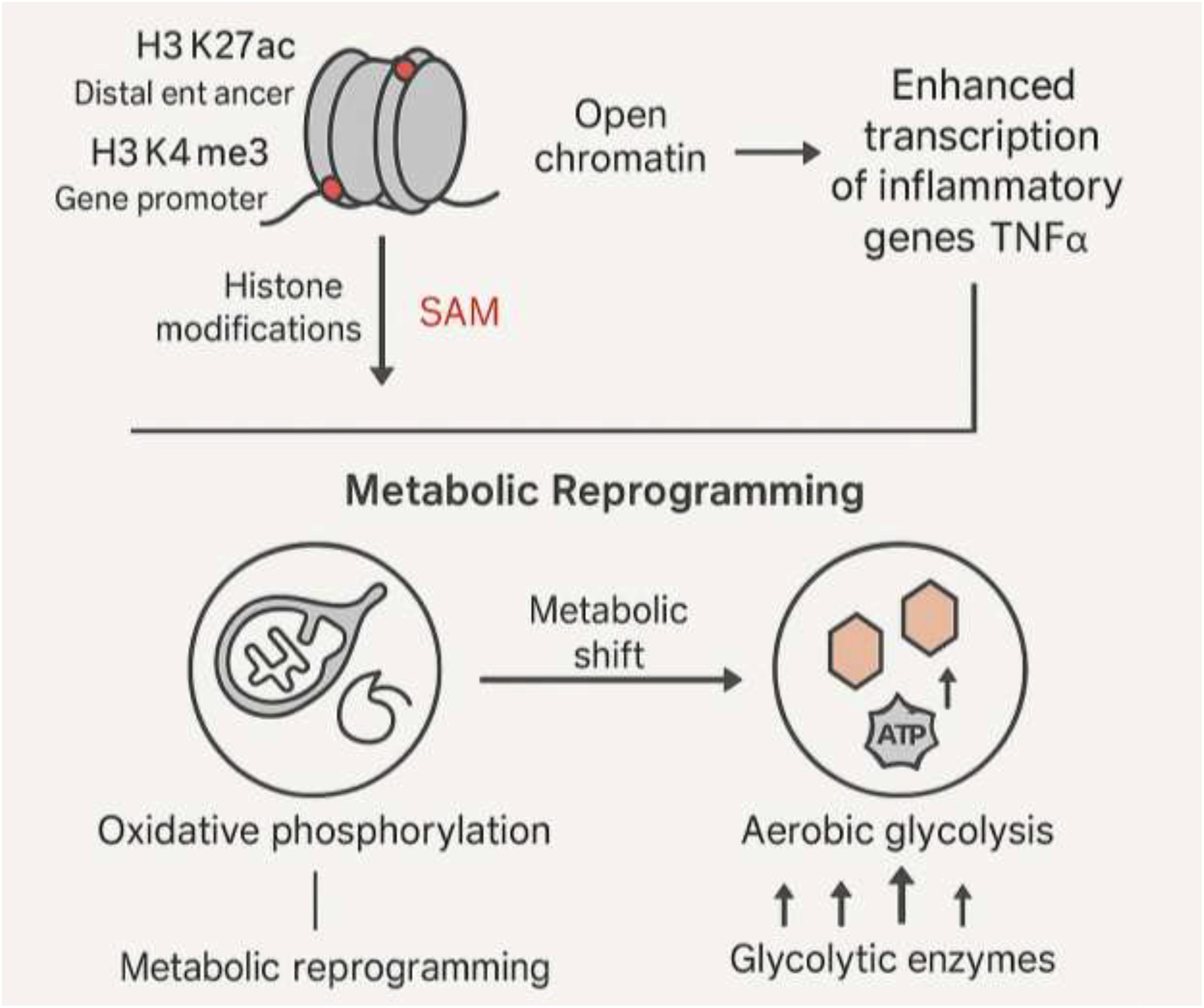
Epigenetic and metabolic reprogramming driving trained immunity.
Epigenetic modifications, including H3K27ac at enhancers and H3K4me3 at promoters, open chromatin and enhance transcription of inflammatory genes like TNFα. In parallel, metabolic reprogramming shifts immune cells from OXPHOS to aerobic glycolysis, regulated by metabolites like SAM and sensors like HIF-1α. These metabolic intermediates fuel epigenetic enzymes, linking metabolism to chromatin remodeling. Together, epigenetic and metabolic rewiring establish long-term innate immune memory, enabling heightened responses upon secondary stimulation.
Skeletal muscle as a mediator: the myokine-immunometabolism axis
Skeletal muscle is linked with locomotion and metabolism for a long time but a more recent paradigm shift has established it as a key endocrine organ that communicates with various tissues throughout the body (25, 81, 82). The release of bioactive compounds called myokines mediates this communication (83). This section will explore how exercise-induced myokines act as critical intermediaries between physical activity and systemic immune modulation.
Myokines: the endocrine arm of exercise
Small proteins and peptides known as myokines are produced and secreted in response to skeletal muscle contraction, especially during exercise (25, 84). Numerous physiological effects are mediated by these signaling molecules, including immunological response, metabolic process regulation, and whole-body homeostasis maintenance (25, 85). Prominent examples of myokines include Interleukin-6 (IL-6), Decorin, Myostatin, and Irisin (25, 86). These molecules facilitate crucial inter-organ crosstalk, influencing distant tissues like adipose tissue, the liver, and the brain (25).
Interleukin-6: a pleiotropic hub
Interleukin-6 is arguably the most extensively studied myokine, and its role in immune regulation is highly complex and nuanced (87, 88). Despite being a well-known pro-inflammatory cytokine, exercise-induced IL-6 is frequently said to have strong anti-inflammatory properties (15). This apparent contradiction highlights the context-dependent nature of its function. Exercise can cause a slight, acute increase in IL-6 in the body, which has direct anti-inflammatory benefits (15, 89). In contrast, more intense or prolonged bouts of exercise can lead to a greater production of inflammatory cytokines (90).
A deeper understanding of IL-6’s pleiotropic role reveals that its function is not simply to suppress inflammation but to actively steer the immune response toward resolution (91). For instance, IL-6 has been shown to enhance the capacity of human macrophages to phagocytose apoptotic cells, a critical step in inflammation resolution (92). Furthermore, by boosting the release of IL-4 and IL-10, IL-6 encourages an anti-inflammatory cytokine profile (92). It also favors the alternative immunoregulatory/M2-like macrophage activation state, which is associated with tissue repair and anti-inflammatory functions, while attenuating pro-inflammatory/M1-like phenotype (87). This positions exercise-induced IL-6 as a crucial link between physical activity and the active resolution of inflammation.
Beyond IL-6: additional pathways and mediators
While IL-6 is a prominent mediator, exercise-induced immune modulation involves a broader network of cytokines, neuroendocrine factors, and neural signals that interact synergistically (45). For instance, other myokines such as IL-15 and IL-7 are released during muscle contraction and contribute to immune regulation. IL-15 promotes the survival and proliferation of natural killer (NK) cells and T lymphocytes, enhancing immunosurveillance, while IL-7 supports lymphocyte homeostasis and may aid in anti-inflammatory responses (46). Neuroendocrine mediators, including catecholamines (e.g., adrenaline and noradrenaline) released via sympathetic activation, modulate immune cells through β-adrenergic receptors, influencing cytokine production and shifting metabolism toward anti-inflammatory states (45, 93). Glucocorticoids like cortisol, elevated during acute exercise, exert anti-inflammatory effects by suppressing pro-inflammatory cytokines but can become immunosuppressive if chronically elevated (45). Additionally, neural inputs, such as vagus nerve signaling in the cholinergic anti-inflammatory pathway, provide rapid modulation of inflammation by inhibiting cytokine release from macrophages (94). This multi-factorial system highlights the limitations of an IL-6-centered framework: overemphasizing IL-6 may overlook synergistic or compensatory roles of these mediators, potentially leading to an incomplete understanding of exercise’s immunomodulatory effects. Future studies should integrate these pathways to better elucidate their interactions (45).
Myokines and immunometabolism
Immune cell metabolic reprogramming is inextricably tied to the signaling cascade that myokines start (95, 96). Trained immunity, as previously discussed, requires a metabolic shift to meet the high energy demands of cellular activation (1). Exercise, by its very nature, is a powerful modulator of systemic metabolism (25). The causal link between these two phenomena is the myokine-immunometabolism axis (97). Exercise-induced myokines act as systemic signals that directly influence the metabolic programs of innate immune cells (78). This effect may help to explain how some metabolic changes (such as a switch from OXPHOS to glycolysis) that are characteristic of trained immunity can be induced by a systemic stimulus like exercise (67). These changes align with the broader metabolic adaptations in trained immunity, including TCA cycle flux and lipid metabolism, as modulated by exercise (54). Myokines like IL-6, which can stimulate fatty acid oxidation and lipolysis, provide the necessary energetic resources for this metabolic transition (87). This establishes a tangible, mechanistic pathway by which exercise “trains” the innate immune system (Figure 2, Table 1).
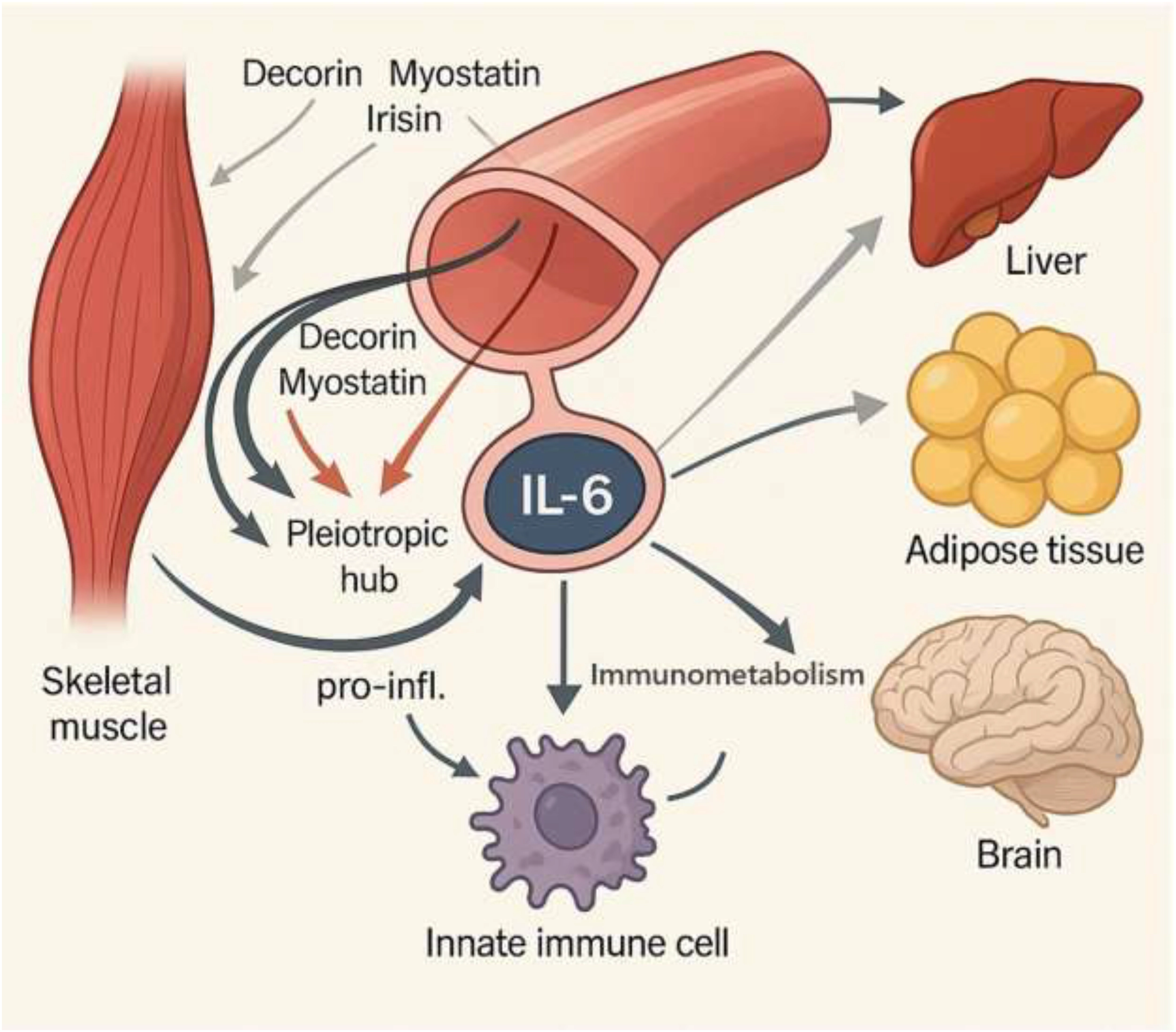
Myokines: The endocrine arm of exercise. Myokines, which are systemic messengers that target organs like the liver, adipose tissue, and brain, are released when skeletal muscle contraction occurs. These include IL-6, Decorin, Myostatin, and Irisin. IL-6 serves as a pleiotropic hub, exerting dual effects: acute, exercise-induced IL-6 promotes anti-inflammatory resolution by enhancing macrophage phagocytosis, inducing IL-4/IL-10 secretion, and driving M2 macrophage polarization; prolonged or excessive IL-6 contributes to pro-inflammatory signaling. Myokines also regulate immunometabolism, shifting immune cells from oxidative phosphorylation toward glycolysis and supporting fatty acid oxidation and lipolysis. This mechanistic axis links exercise to immune regulation, inflammation resolution, and metabolic adaptation.
MyokinePrimary stimulusTarget cell/organEffect on immunityInterleukin-6 (IL-6)Muscle contraction (aerobic/resistance)Macrophages, Adipose Tissue, LiverEnhances macrophage phagocytosis, promotes M2 anti-inflammatory phenotype, signals for inflammation resolutionDecorinMuscle contractionAdipose Tissue, LiverAnti-inflammatory and anti-fibrotic effects, reduces hepatic inflammationMyostatinMuscle contractionAdipose Tissue, LiverLower plasma levels reduce pro-inflammatory factors like TNFαIrisinMuscle contractionAdipose Tissue, LiverMay induce anti-inflammatory response, although high levels are also linked to inflammationBrain-Derived Neurotrophic Factor (BDNF)Muscle contractionNeurons, Immune cellsPromotes neuronal survival, modulates immune cell functionLeukemia Inhibitory Factor (LIF)Muscle contractionImmune cellsPromotes immune cell proliferation and differentiationInterleukin-15 (IL-15)Muscle contractionNK cells, T lymphocytesPromotes survival and proliferation of NK and T cells, enhances immunosurveillanceInterleukin-7 (IL-7)Muscle contractionLymphocytesSupports lymphocyte homeostasis and anti-inflammatory responses
Important myokines induced by exercise and their immunomodulatory impacts.
Resolving inflammation: the final phase of the immune response
In order to restore tissue homeostasis and avoid chronic inflammatory illness, inflammation resolution is an active, coordinated process (9, 98). This section will detail the key molecular and cellular players in this process and demonstrate how exercise directly contributes to its efficiency.
The pillars of inflammation resolution
Following the initial acute inflammatory response, a programmed resolution phase, known as catabasis, is initiated (9, 99). This process is characterized by a set of “five pillars,” which include the active elimination of bacteria, debris, and dead cells; vascular integrity restoration; tissue regeneration; fever remission; and inflammatory pain decrease (9). Chronic inflammatory disorders are largely caused by the failure of this active mechanism (100).
Specialized pro-resolving mediators
A cornerstone of inflammation resolution is a class of signaling molecules known as SPMs (9). Lipid mediator class-switching is the process by which these are enzymatically produced from necessary fatty acids (9). Key families of SPMs include lipoxins, resolvins (e.g., Resolvin D1), protectins, and maresins (9). SPMs differ from immunosuppressive compounds in that they actively support host defense, the removal of pathogens and cellular detritus, and inflammation reduction (9). Regular physical activity has been shown to potentially accelerate and strengthen the pro-resolving lipid mediator response following acute exercise stress (101). Furthermore, Exercise increases RvD1 levels and stimulates macrophage phagocytosis, a crucial pro-resolving mechanism, according to study (28) (Table 2).
SPM familyPrecursor fatty acidKey pro-resolving actionsLink to exerciseResolvins (D/E-series)Omega-3 PUFAs (DHA/EPA)Promotes clearance of pathogens and apoptotic cells, inhibits neutrophil infiltration, stimulates anti-inflammatory cytokine productionExercise enhances Resolvin D1 levels and strengthens the response to acute stress (28)LipoxinsOmega-6 PUFAs (Arachidonic Acid)Inhibits neutrophil recruitment and promotes their non-inflammatory apoptosis, suppresses pro-inflammatory signalingExercise may influence the class-switching from pro-inflammatory to pro-resolving lipid mediators (9)MaresinsOmega-3 PUFAs (DHA)Augment macrophage phagocytosis and microbial killing, promote tissue repairPotential link, but specific research on maresins and exercise is an area for future studyProtectinsOmega-3 PUFAs (DHA)Neuroprotective, anti-inflammatory, and promotes efferocytosisPotential link, but specific research on protectins and exercise is an area for future study
SPMs and their mechanisms.
Efferocytosis: a critical cellular mechanism
Efferocytosis, a carefully controlled biological process, occurs when macrophages quickly eliminate apoptotic cells (77, 102, 103). This procedure is essential for stopping necrotic cells from releasing pro-inflammatory DAMPs, which can cause sterile inflammation and worsen chronic illness (77, 104). The timely clearance of apoptotic cells is, in itself, a powerful pro-resolving signal (77). The degradation of these cells provides macrophages with key metabolites, like fatty acids, amino acids, and cholesterol, which causes the macrophage to change into a pro-resolving and anti-inflammatory phenotype (77).
An intricate nexus exists between exercise, myokine signaling, and efferocytosis. Exercise-induced IL-6 increases human macrophages’ capacity to phagocytose apoptotic cells (92, 105). This creates a clear, multi-layered causal pathway: exercise triggers the systemic release of myokines and SPMs, which in turn enhance efferocytosis and fuel the pro-resolving macrophage phenotype (106). This mechanism elegantly demonstrates how physical activity actively promotes the restoration of tissue homeostasis, providing a robust defense against chronic inflammatory conditions (92).
Unifying the concepts: a model of exercise-induced immune modulation
The complex relationship between exercise, trained immunity, and inflammation resolution can be synthesized into a unified model that connects molecular, cellular, and systemic effects (107). This model resolves the paradox of exercise’s dual effects on immunity and provides a comprehensive framework for its physiological benefits (107, 108).
Physical activity, particularly moderate, regular exercise, acts as a controlled physiological stressor (15, 109). Skeletal muscle’s endocrine activity is triggered by this stimulus, which causes the systemic release of several myokines, the main one being IL-6 (25). This systemic myokine signaling, in turn, influences the metabolism of innate immune cells (78). This metabolic shift provides the necessary energetic and material resources to fuel the epigenetic changes at key inflammatory gene promoters and enhancers that define trained immunity (1). This long-term reprogramming leads to a persistent state of enhanced immunosurveillance and heightened responsiveness to pathogens (15).
Simultaneously, myokines like IL-6 directly increase efferocytosis, and exercise stimulates the synthesis of pro-resolving lipid mediators like SPMs (28, 110). In addition to preventing the development of sterile inflammation, this active removal of apoptotic cells also strengthens the pro-resolving macrophage character, ensuring the efficient and timely resolution of inflammation (77).
This model provides a compelling explanation for the J-shaped curve of exercise and illness risk. Moderate, regular exercise is associated with inducing a beneficial, transient inflammatory state that is followed by a robust, myokine- and SPM-driven resolution phase (15). This repeated, controlled “training” of the immune system leads to chronic adaptations (107). In contrast, heavy, chronic training may induce an inflammatory state so profound or persistent that the resolution mechanisms are overwhelmed, leading to the observed transient immunosuppression and increased risk of minor infections (111) (Table 3). The key is the delicate balance between the initial pro-inflammatory stimulus and the subsequent pro-resolving response.
Cell typeAcute exercise (<60 min, moderate)Acute prolonged strenuous exercise (>90 min)Chronic regular exerciseNeutrophilsIncreased recirculation, Enhanced phagocytosisTransient decrease, Depressed phagocytosisEnhanced immune regulationNK CellsIncreased recirculation and cytotoxicitySignificant decrease in circulating numbersMay be elevated at rest in some athletes, improved functionMonocytesIncreased recirculationSignificant decrease in circulating numbers, reduced functionDecreased systemic inflammation, enhanced functionMacrophagesEnhanced anti-pathogen activityReduced antigen presentationEnhanced efferocytosis, anti-inflammatory phenotype
Exercise’s acute and chronic effects on innate immune cells.
Knowledge gaps, conflicting evidence, and future directions
Exercise immunology has made great strides, yet there are still debates and unresolved issues in the field. A critical appraisal of the existing literature is essential for advancing our understanding and translating these findings into clinical practice.
The “open window” debate and its methodological limitations
The “open window” hypothesis, which posits a temporary period of immunosuppression after strenuous exercise, is a cornerstone of exercise immunology (19, 112). However, some contemporary evidence and critical reviews argue that there is limited reliable data to support this claim (16). A major contributing factor to this debate and the often-conflicting results in the literature is the reliance on traditional ex vivo immune markers. Clinical outcomes including the likelihood of upper respiratory tract infections are not necessarily correlated with these markers, such as variations in circulating cell counts or cytokine levels in blood tests (113). A temporary change in a single immune variable in the blood does not necessarily equate to a compromised host defense, due to the inherent redundancy and compensatory mechanisms of the immune system (113, 114).
Future research must move beyond these correlational studies and adopt more rigorous methodologies (115). The use of controlled in vivo infection challenge models is necessary to establish a direct causal link between exercise-induced changes and host defense against pathogens (113). Additionally, Research should take a multifaceted approach, taking into consideration additional variables that affect immunological health, such sleep, as exposure to pathogens, nutrition, and psychological stress (112).
Specific gaps in our understanding
A comprehensive understanding of the exercise-immunity nexus requires addressing several key knowledge gaps:
It is still unclear the precise signaling pathways connect exercise-induced myokines to the metabolic and epigenetic remodeling of HSCs (1, 116). Direct evidence linking exercise to epigenetic shaping of the immune cell landscape is emerging but remains limited; while overlaps with trained immunity exist, many studies are associative, extrapolated from non-exercise models.
The full spectrum of myokines involved in immune regulation is yet to be fully characterized, with most research focusing on a limited number of molecules like IL-6 (46).
There is clear absence enough research of on the infection risk and immune adaptations in predominantly strength-based athletes, despite the similar immunological changes observed in aerobic and resistance exercise (113, 117, 118).
The precise upstream signals that trigger the release of exercise-induced molecules like cell-free DNA (cfDNA) are not the major inflammatory cytokines, suggesting other, yet-to-be-discovered mechanisms (119).
Much of the evidence linking exercise to trained immunity and HSC reprogramming is indirect or associative, often extrapolated from non-exercise models like BCG or β-glucan; direct causal studies in exercise contexts are needed (45).
Future directions
To fully elucidate the complex interplay between exercise, trained immunity, and inflammation resolution, we advocate for a multi-omics approach that integrates data from the genome, epigenome, transcriptome, proteome, metabolome, and microbiome (1, 120). This systems biology approach will allow for a holistic understanding of the intricate networks that regulate exercise-induced immune modulation (1).
Furthermore, clinical research should employ rigorous methodology, including controlled infection challenge models, to move beyond correlational ex vivo data and establish direct causal links between exercise and host defense (113). Future research should also look into how various exercise styles (such as resistance versus aerobic) affect the myokine profile and how that affects trained immunity and the resolution of inflammation (121). Ultimately, gaining a comprehensive understanding of these pathways is essential for creating innovative, focused treatment strategies to fight chronic inflammatory disorders and infectious diseases (58).
Conclusion
Exercise emerges as a powerful physiological modulator of immune function by bridging two rapidly evolving concepts in immunology: trained immunity and inflammation resolution. Through coordinated epigenetic reprogramming and metabolic rewiring, exercise imprints long-lasting functional adaptations on innate immune cells, enhancing their capacity for immunosurveillance while simultaneously fostering a pro-resolving phenotype. As molecular conduits, central mediators like myokines (particularly IL-6) and SPMs connect tissue homeostasis and systemic immune control to skeletal muscle function. Together, these mechanisms explain how regular physical activity not only strengthens resistance against pathogens but also mitigates chronic inflammation, a common denominator in aging and numerous non-communicable diseases. While significant progress has been made, critical knowledge gaps remain, particularly in defining the temporal dynamics of exercise-induced innate memory, various exercise techniques’ dosage and response, and the integration of multi-omics approaches to unravel this complexity in vivo. Future research should aim to translate these mechanistic insights into targeted exercise-mimetic therapies or personalized training prescriptions, harnessing the dual potential of exercise to train immunity and resolve inflammation. Ultimately, this concept presents exercise as a biologically based therapeutic approach for fostering resilience, avoiding illness, and prolonging health span rather than just as a lifestyle intervention.
StatementsAuthor contributions
YL: Data curation, Investigation, Methodology, Writing – original draft, Writing – review & editing. DW: Conceptualization, Data curation, Investigation, Methodology, Supervision, Writing – original draft, Writing – review & editing.
Funding
The author(s) declared that financial support was not received for this work and/or its publication.
Conflict of interest
The author(s) declared that this work was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declared that generative AI was used in the creation of this manuscript. Figures were produced from adapted illustrations supplied by Servier Medical Art (Servier; https://smart.servier.com/), licensed under a Creative Commons Attribution 4.0 Unported License. We also acknowledge Canva Pty Ltd (Sydney, Australia) for providing the design software that enabled the creation of the figures; all graphical content remains the original work of the authors. During the development of this work, the authors employed Grok and ChatGPT to refine the writing process and to improve the manuscript’s clarity and linguistic quality. Following the utilization of this tool/service, the authors conducted a review and made necessary revisions, assuming full responsibility for the content of the published article.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
KoekenVvan CrevelRNeteaMGLiY.
Resolving trained immunity with systems biology. Eur J Immunol. (2021) 51:773–84. doi: 10.1002/eji.202048882
WarringtonRWatsonWKimHLAntonettiFR.
An introduction to immunology and immunopathology. Allergy Asthma Clin Immunol. (2011) 7 Suppl 1:S1. doi: 10.1186/1710-1492-7-S1-S1
MarshallJSWarringtonRWatsonWKimHL.
An introduction to immunology and immunopathology. Allergy Asthma Clin Immunol. (2018) 14:49. doi: 10.1186/s13223-018-0278-1
WangRLanCBenlaghaKCamaraNOSMillerH.
The interaction of innate immune and adaptive immune system. MedComm (2020). (2024) 5:e714. doi: 10.1002/mco2.714
Dominguez-AndresJNeteaMG.
Long-term reprogramming of the innate immune system. J Leukoc Biol. (2019) 105:329–38. doi: 10.1002/JLB.MR0318-104R
BekkeringSDomínguez-AndrésJJoostenLABRiksenNPNeteaMG.
Trained immunity: reprogramming innate immunity in health and disease. Annu Rev Immunol. (2021) 39:667–93. doi: 10.1146/annurev-immunol-102119-073855
DaskalakiMGLapiIHurstAEAl-QahtaniAVergadiETsatsanisCet al.
Epigenetic and metabolic regulation of macrophage responsiveness and memory. J Immunol. (2025). doi: 10.1093/jimmun/vkaf135
EtchegarayJPMostoslavskyR.
Interplay between metabolism and epigenetics: A nuclear adaptation to environmental changes. Mol Cell. (2016) 62:695–711. doi: 10.1016/j.molcel.2016.05.029
BasilMCLevyBD.
Specialized pro-resolving mediators: endogenous regulators of infection and inflammation. Nat Rev Immunol. (2016) 16:51–67. doi: 10.1038/nri.2015.4
SerhanCNKrishnamoorthySRecchiutiAChiangN.
Novel anti-inflammatory–pro-resolving mediators and their receptors. Curr Top Med Chem. (2011) 11:629–47.
BannenbergGSerhanCN.
Specialized pro-resolving lipid mediators in the inflammatory response: An update. Biochim Biophys Acta. (2010) 1801:1260–73. doi: 10.1016/j.bbalip.2010.08.002
SerhanCNChiangNDalliJLevyBD.
Lipid mediators in the resolution of inflammation. Cold Spring Harbor Perspect Biol. (2015) 7:a016311. doi: 10.1101/cshperspect.a016311
DalliJSerhanCN.
Pro-resolving mediators in regulating and conferring macrophage function. Front Immunol. (2017) 8:1400. doi: 10.3389/fimmu.2017.01400
GleesonM.
Immune function in sport and exercise. J Appl Physiol (1985). (2007) 103:693–9. doi: 10.1152/japplphysiol.00008.2007
NiemanDCWentzLM.
The compelling link between physical activity and the body’s defense system. J Sport Health Sci. (2019) 8:201–17. doi: 10.1016/j.jshs.2018.09.009
CampbellJPTurnerJE.
Debunking the myth of exercise-induced immune suppression: redefining the impact of exercise on immunological health across the lifespan. Front Immunol. (2018) 9:648. doi: 10.3389/fimmu.2018.00648
MalmC.
Susceptibility to infections in elite athletes: the S-curve. Scand J Med Sci Sports. (2006) 16:4–6. doi: 10.1111/j.1600-0838.2005.00499.x
Chamorro-ViñaCFernandez-del-ValleMTacónAM.
Excessive exercise and immunity: the J-shaped curve. In: The active female: health issues throughout the lifespan. New York:
Springer (2013). p. 357–72.
CampbellJPTurnerJE.
There is limited existing evidence to support the common assumption that strenuous endurance exercise bouts impair immune competency. Expert Rev Clin Immunol. (2019) 15:105–9. doi: 10.1080/1744666X.2019.1548933
KakanisMWPeakeJBrenuEWSimmondsMGrayBHooperSLet al.
The open window of susceptibility to infection after acute exercise in healthy young male elite athletes. Exerc Immunol Rev. (2010) 16:119–37. doi: 10.1016/j.jsams.2010.10.642
WestNPPyneDBRenshawGCrippsAW.
Antimicrobial peptides and proteins, exercise and innate mucosal immunity. FEMS Immunol Med Microbiol. (2006) 48:293–304. doi: 10.1111/j.1574-695X.2006.00132.x
KoelwynGJWennerbergEDemariaSJonesLW.
Exercise in regulation of inflammation-immune axis function in cancer initiation and progression. Oncol (Williston Park). (2015) 29:908–20, 922.
ArtsRJWJoostenLABNeteaMG.
The potential role of trained immunity in autoimmune and autoinflammatory disorders. Front Immunol. (2018) 9:298. doi: 10.3389/fimmu.2018.00298
NeteaMGDomínguez-AndrésJBarreiroLBChavakisTDivangahiMFuchsEet al.
Defining trained immunity and its role in health and disease. Nat Rev Immunol. (2020) 20:375–88. doi: 10.1038/s41577-020-0285-6
LuZWangZZhangXANingK.
Myokines may be the answer to the beneficial immunomodulation of tailored exercise-A narrative review. Biomolecules. (2024) 14. doi: 10.3390/biom14101205
BrittECJohnSVLocasaleJWFanJ.
Metabolic regulation of epigenetic remodeling in immune cells. Curr Opin Biotechnol. (2020) 63:111–7. doi: 10.1016/j.copbio.2019.12.008
McGeeSLWalderKR.
Exercise and the skeletal muscle epigenome. Cold Spring Harb Perspect Med. (2017) 7. doi: 10.1101/cshperspect.a029876
ZhengJJPena CalderinEHillBGBhatnagarAHellmannJ.
Exercise promotes resolution of acute inflammation by catecholamine-mediated stimulation of resolvin D1 biosynthesis. J Immunol. (2019) 203:3013–22. doi: 10.4049/jimmunol.1900144
MarkworthJFMaddipatiKRCameron-SmithD.
Emerging roles of pro-resolving lipid mediators in immunological and adaptive responses to exercise-induced muscle injury. Exerc Immunol Rev. (2016) 22:110–34.
WoodsJAVieiraVJKeylockKT.
Exercise, inflammation, and innate immunity. Neurol Clin. (2006) 24:585–99. doi: 10.1016/j.ncl.2006.03.008
SerhanCN.
Pro-resolving lipid mediators are leads for resolution physiology. Nature. (2014) 510:92–101. doi: 10.1038/nature13479
NeteaMG.
Training innate immunity: the changing concept of immunological memory in innate host defence. Eur J Clin Invest. (2013) 43:881–4. doi: 10.1111/eci.12132
MishraSArshAMRathoreJS.
Trained innate immunity and diseases: Bane with the boon. Clin Immunol Commun. (2022) 2:118–29. doi: 10.1016/j.clicom.2022.08.004
BhargaviGSubbianS.
The causes and consequences of trained immunity in myeloid cells. Front Immunol. (2024) 15:1365127. doi: 10.3389/fimmu.2024.1365127
NeteaMGet al.
Trained immunity: A program of innate immune memory in health and disease. Science. (2016) 352:aaf1098. doi: 10.1126/science.aaf1098
TercanHet al.
Trained immunity: long-term adaptation in innate immune responses. Arterioscler Thromb Vasc Biol. (2021) 41:55–61. doi: 10.1161/ATVBAHA.120.314212
BekkeringSet al.
In vitro experimental model of trained innate immunity in human primary monocytes. Clin Vaccine Immunol. (2016) 23:926–33. doi: 10.1128/CVI.00349-16
HamadaAet al.
Trained immunity carried by non-immune cells. Front Microbiol. (2018) 9:3225. doi: 10.3389/fmicb.2018.03225
KalafatiLet al.
The role of neutrophils in trained immunity. Immunol Rev. (2023) 314:142–57. doi: 10.1111/imr.13142
RuffinattoLet al.
Hematopoietic stem cell a reservoir of innate immune memory. Front Immunol. (2024) 15:1491729. doi: 10.3389/fimmu.2024.1491729
LiaoWet al.
Hematopoietic stem cell state and fate in trained immunity. Cell Commun Signal. (2025) 23:182. doi: 10.1186/s12964-025-02192-1
De ZuaniMFričJ.
Train the trainer: hematopoietic stem cell control of trained immunity. Front Immunol. (2022) 13:827250. doi: 10.3389/fimmu.2022.827250
MoorlagSet al.
BCG vaccination induces long-term functional reprogramming of human neutrophils. Cell Rep. (2020) 33:108387. doi: 10.1016/j.celrep.2020.108387
CirovicBet al.
BCG vaccination in humans elicits trained immunity via the hematopoietic progenitor compartment. Cell Host Microbe. (2020) 28:322–334.e5. doi: 10.1016/j.chom.2020.05.014
SchefferDDLLatiniA.
Exercise-induced immune system response: Anti-inflammatory status on peripheral and central organs. Biochim Biophys Acta Mol Basis Dis. (2020) 1866:165823. doi: 10.1016/j.bbadis.2020.165823
Dumond BourieAet al.
Myokines: crosstalk and consequences on liver physiopathology. Nutrients. (2023) 15. doi: 10.3390/nu15071729
XingJet al.
Identification of a role for TRIM29 in the control of innate immunity in the respiratory tract. Nat Immunol. (2016) 17:1373–80. doi: 10.1038/ni.3580
LiQet al.
TRIM29 negatively controls antiviral immune response through targeting STING for degradation. Cell Discov. (2018) 4:13. doi: 10.1038/s41421-018-0010-9
XingJet al.
TRIM29 promotes DNA virus infections by inhibiting innate immune response. Nat Commun. (2017) 8:945. doi: 10.1038/s41467-017-00101-w
MaryczKet al.
Endurance exercise mobilizes developmentally early stem cells into peripheral blood and increases their number in bone marrow: implications for tissue regeneration. Stem Cells Int. (2016) 2016:5756901. doi: 10.1155/2016/5756901
De LisioMPariseG.
Characterization of the effects of exercise training on hematopoietic stem cell quantity and function. J Appl Physiol. (2012) 113:1576–84. doi: 10.1152/japplphysiol.00717.2012
QuWWet al. Effects of Enhanced Physical Activity on Hematopoietic Stem Cell Aging in PEPCK-Cmus Transgenic Mice. New York:
American Society of Hematology (2011).
ArtsRJJoostenLANeteaMG.
Immunometabolic circuits in trained immunity. Semin Immunol. (2016) 28:425–30. doi: 10.1016/j.smim.2016.09.002
FerreiraAVet al.
Metabolic regulation in the induction of trained immunity. Semin Immunopathol. (2024) 46:7. doi: 10.1007/s00281-024-01015-8
FerreiraAVDomiguéz-AndrésJNeteaMG.
The role of cell metabolism in innate immune memory. J Innate Immun. (2022) 14:42–50. doi: 10.1159/000512280
SunSAguirre-GamboaRBarreiroLB.
Transmission of stimulus-induced epigenetic changes through cell division is coupled to continuous transcription factor activity. Front Immunol. (2023) 14:1129577. doi: 10.3389/fimmu.2023.1129577
MutchlerALet al.
Epigenetic regulation of innate and adaptive immune cells in salt-sensitive hypertension. Circ Res. (2025) 136:232–54. doi: 10.1161/CIRCRESAHA.124.325439
van der HeijdenCet al.
Epigenetics and trained immunity. Antioxid Redox Signal. (2018) 29:1023–40. doi: 10.1089/ars.2017.7310
Domínguez-AndrésJet al.
Advances in understanding molecular regulation of innate immune memory. Curr Opin Cell Biol. (2020) 63:68–75. doi: 10.1016/j.ceb.2019.12.006
FanucchiSMhlangaMM.
Lnc-ing trained immunity to chromatin architecture. Front Cell Dev Biol. (2019) 7:2. doi: 10.3389/fcell.2019.00002
FanucchiSet al.
Immune genes are primed for robust transcription by proximal long noncoding RNAs located in nuclear compartments. Nat Genet. (2019) 51:138–50. doi: 10.1038/s41588-018-0298-2
DongXWengZ.
The correlation between histone modifications and gene expression. Epigenomics. (2013) 5:113–6. doi: 10.2217/epi.13.13
AlAfaleqNOet al.
Generation of site-specific ubiquitinated histones through chemical ligation to probe the specificities of histone deubiquitinases. Front Epigenet Epigenom. (2023) 1. doi: 10.3389/freae.2023.1238154
PflugerJWagnerD.
Histone modifications and dynamic regulation of genome accessibility in plants. Curr Opin Plant Biol. (2007) 10:645–52. doi: 10.1016/j.pbi.2007.07.013
ZhangNet al.
Early-life exercise induces immunometabolic epigenetic modification enhancing anti-inflammatory immunity in middle-aged male mice. Nat Commun. (2024) 15:3103. doi: 10.1038/s41467-024-47458-3
VoisinSet al.
Exercise training and DNA methylation in humans. Acta Physiol. (2015) 213:39–59. doi: 10.1111/apha.12414
MohammadnezhadLet al.
Metabolic reprogramming of innate immune cells as a possible source of new therapeutic approaches in autoimmunity. Cells. (2022) 11. doi: 10.3390/cells11101663
LoftusRMFinlayDK.
Immunometabolism: cellular metabolism turns immune regulator. J Biol Chem. (2016) 291:1–10. doi: 10.1074/jbc.R115.693903
AndrejevaGRathmellJC.
Similarities and distinctions of cancer and immune metabolism in inflammation and tumors. Cell Metab. (2017) 26:49–70. doi: 10.1016/j.cmet.2017.06.004
Domínguez-AndrésJJoostenLANeteaMG.
Induction of innate immune memory: the role of cellular metabolism. Curr Opin Immunol. (2019) 56:10–6. doi: 10.1016/j.coi.2018.09.001
KieransSJTaylorCT.
Regulation of glycolysis by the hypoxia-inducible factor (HIF): implications for cellular physiology. J Physiol. (2021) 599:23–37. doi: 10.1113/JP280572
BasheeruddinMQausainS.
Hypoxia-inducible factor 1-alpha (HIF-1α): an essential regulator in cellular metabolic control. Cureus. (2024) 16:e63852. doi: 10.7759/cureus.63852
GaoJLChenYG.
Natural compounds regulate glycolysis in hypoxic tumor microenvironment. BioMed Res Int. (2015) 2015:354143. doi: 10.1155/2015/354143
van TuijlJet al.
Immunometabolism orchestrates training of innate immunity in atherosclerosis. Cardiovasc Res. (2019) 115:1416–24. doi: 10.1093/cvr/cvz107
FanucchiSet al.
The intersection of epigenetics and metabolism in trained immunity. Immunity. (2021) 54:32–43. doi: 10.1016/j.immuni.2020.10.011
WangPet al.
Metabolite regulation of epigenetics in cancer. Cell Rep. (2024) 43:114815. doi: 10.1016/j.celrep.2024.114815
SchilperoortMet al.
The role of efferocytosis-fueled macrophage metabolism in the resolution of inflammation. Immunol Rev. (2023) 319:65–80. doi: 10.1111/imr.13214
NiemanDCPenceBD.
Exercise immunology: Future directions. J Sport Health Sci. (2020) 9:432–45. doi: 10.1016/j.jshs.2019.12.003
RiksenNPNeteaMG.
Immunometabolic control of trained immunity. Mol Aspects Med. (2021) 77:100897. doi: 10.1016/j.mam.2020.100897
ArtsRJet al.
Glutaminolysis and fumarate accumulation integrate immunometabolic and epigenetic programs in trained immunity. Cell Metab. (2016) 24:807–19. doi: 10.1016/j.cmet.2016.10.008
IglesiasP.
Muscle in endocrinology: from skeletal muscle hormone regulation to myokine secretion and its implications in endocrine-metabolic diseases. J Clin Med. (2025) 14. doi: 10.3390/jcm14134490
IizukaKMachidaTHirafujiM.
Skeletal muscle is an endocrine organ. J Pharmacol Sci. (2014) 125:125–31. doi: 10.1254/jphs.14R02CP
MoonesanM.
Muscle-kidney crosstalk; the role of myokines. J Ren Endocrinol. (2023) 9:e25129. doi: 10.34172/jre.2023.25129
WeigertCet al.
The secretome of the working human skeletal muscle–a promising opportunity to combat the metabolic disaster? Proteomics Clin Appl. (2014) 8:5–18.
JiaWet al.
Bile acid signaling in the regulation of whole body metabolic and immunological homeostasis. Sci China Life Sci. (2024) 67:865–78. doi: 10.1007/s11427-023-2353-0
BarbalhoSMet al.
Myokines: a descriptive review. J Sports Med Phys Fitness. (2020) 60:1583–90. doi: 10.23736/S0022-4707.20.10884-3
CovarrubiasAJHorngT.
IL-6 strikes a balance in metabolic inflammation. Cell Metab. (2014) 19:898–9. doi: 10.1016/j.cmet.2014.05.009
PalMFebbraioMAWhithamM.
From cytokine to myokine: the emerging role of interleukin-6 in metabolic regulation. Immunol Cell Biol. (2014) 92:331–9. doi: 10.1038/icb.2014.16
IslamHet al.
Interpreting ‘anti-inflammatory’ cytokine responses to exercise: focus on interleukin-10. J Physiol. (2021) 599:5163–77. doi: 10.1113/JP281356
RenkeGet al.
β-glucan “Trained immunity” Immunomodulatory properties potentiate tissue wound management and accelerate fitness recover. Immunotarget Ther. (2022) 11:67–73. doi: 10.2147/ITT.S381145
MauerJDensonJLBrüningJC.
Versatile functions for IL-6 in metabolism and cancer. Trends Immunol. (2015) 36:92–101. doi: 10.1016/j.it.2014.12.008
FrisdalEet al.
Interleukin-6 protects human macrophages from cellular cholesterol accumulation and attenuates the proinflammatory response. J Biol Chem. (2011) 286:30926–36. doi: 10.1074/jbc.M111.264325
AthanasiouNBogdanisGCMastorakosG.
Endocrine responses of the stress system to different types of exercise. Rev Endocr Metab Disord. (2023) 24:251–66. doi: 10.1007/s11154-022-09758-1
TraceyKJ.
The inflammatory reflex. Nature. (2002) 420:853–9. doi: 10.1038/nature01321
RatterJMet al.
Environmental signals influencing myeloid cell metabolism and function in diabetes. Trends Endocrinol Metab. (2018) 29:468–80. doi: 10.1016/j.tem.2018.04.008
GaraudeJ.
Reprogramming of mitochondrial metabolism by innate immunity. Curr Opin Immunol. (2019) 56:17–23. doi: 10.1016/j.coi.2018.09.010
BayMLPedersenBK.
Muscle-organ crosstalk: focus on immunometabolism. Front Physiol. (2020) 11:567881. doi: 10.3389/fphys.2020.567881
SolimanAMet al.
Molecular dynamics of inflammation resolution: therapeutic implications. Front Cell Dev Biol. (2025) 13:1600149. doi: 10.3389/fcell.2025.1600149
SerhanCNSavillJ.
Resolution of inflammation: the beginning programs the end. Nat Immunol. (2005) 6:1191–7. doi: 10.1038/ni1276
HajishengallisGNeteaMChavakisT.
Trained immunity in chronic inflammatory diseases and cancer. Nat Rev Immunol. (2025) 25. doi: 10.1038/s41577-025-01132-x
MalanLet al.
Regular moderate physical activity potentially accelerates and strengthens both the pro-inflammatory and pro-resolving lipid mediator response after acute exercise stress. Prostaglandins Leukot Essent Fatty Acids. (2024) 202:102642. doi: 10.1016/j.plefa.2024.102642
Gheibi HayatSMet al.
Efferocytosis: molecular mechanisms and pathophysiological perspectives. Immunol Cell Biol. (2019) 97:124–33. doi: 10.1111/imcb.12206
TrzeciakAWangYTPerryJSA.
First we eat, then we do everything else: The dynamic metabolic regulation of efferocytosis. Cell Metab. (2021) 33:2126–41. doi: 10.1016/j.cmet.2021.08.001
PoonIKHRavichandranKS.
Targeting efferocytosis in inflammaging. Annu Rev Pharmacol Toxicol. (2024) 64:339–57. doi: 10.1146/annurev-pharmtox-032723-110507
Muñoz-CánovesPet al.
Interleukin-6 myokine signaling in skeletal muscle: a double-edged sword? FEBS J. (2013) 280:4131–48.
Pena CalderinEHHillBGHellmannJ.
Exercise and specialized proresolving lipid mediators stimulate mitochondrial metabolism in macrophages. Circulation. (2021) 144:A14116–6. doi: 10.1161/circ.144.suppl_1.14116
AllenJSunYWoodsJA.
Exercise and the regulation of inflammatory responses. Prog Mol Biol Transl Sci. (2015) 135:337–54. doi: 10.1016/bs.pmbts.2015.07.003
LangstonPKMathisD.
Immunological regulation of skeletal muscle adaptation to exercise. Cell Metab. (2024) 36:1175–83. doi: 10.1016/j.cmet.2024.04.001
RadakZet al.
Exercise, oxidative stress and hormesis. Ageing Res Rev. (2008) 7:34–42. doi: 10.1016/j.arr.2007.04.004
CalderinEPet al.
Exercise-induced specialized proresolving mediators stimulate AMPK phosphorylation to promote mitochondrial respiration in macrophages. . Mol Metab. (2022) 66:101637. doi: 10.1016/j.molmet.2022.101637
JonesAWDavisonG.
Exercise, immunity, and illness. In: Muscle and exercise physiology. New York:
Elsevier (2019). p. 317–44.
Effects of Exercise on Immune Function – Gatorade Sports Science Institute. Available online at: https://www.gssiweb.org/sports-science-exchange/article/sse-151-effects-of-exercise-on-immune-function/1000.
DavisonGChidleyCJonesAW.
Rigorous methodological approaches to address knowledge gaps in exercise, nutrition, immunity, and infection risk research. Physiol Rep. (2025) 13:e70479. doi: 10.14814/phy2.70479
CasanovaJLAbelL.
Human genetics of infectious diseases: Unique insights into immunological redundancy. Semin Immunol. (2018) 36:1–12. doi: 10.1016/j.smim.2017.12.008
NishSMedzhitovR.
Host defense pathways: role of redundancy and compensation in infectious disease phenotypes. Immunity. (2011) 34:629–36. doi: 10.1016/j.immuni.2011.05.009
SinghAKAlthoffMJCancelasJA.
Signaling pathways regulating hematopoietic stem cell and progenitor aging. Curr Stem Cell Rep. (2018) 4:166–81. doi: 10.1007/s40778-018-0128-6
Immunology and Exercise – Brian Tabor | Health & Wellness Articles. Available online at: https://www.performancemenu.com/article/96/Immunology-and-Exercise/.
KurowskiMet al.
Physical exercise, immune response, and susceptibility to infections-current knowledge and growing research areas. Allergy. (2022) 77:2653–64. doi: 10.1111/all.15328
RodriguesKBet al.
Exercise intensity and training alter the innate immune cell type and chromosomal origins of circulating cell-free DNA in humans. Proc Natl Acad Sci U.S.A. (2025) 122:e2406954122. doi: 10.1073/pnas.2406954122
ChuXet al.
Multi-omics approaches in immunological research. Front Immunol. (2021) 12:668045. doi: 10.3389/fimmu.2021.668045
BettarigaFet al.
Exercise training mode effects on myokine expression in healthy adults: A systematic review with meta-analysis. J Sport Health Sci. (2024) 13:764–79. doi: 10.1016/j.jshs.2024.04.005
Summary
Keywords
epigenetic reprogramming, exercise immunology, inflammation resolution, sustainable development, trained immunity
Citation
Li Y and Wang D (2026) Forging resilient warriors within: exercise’s epic role in training innate immunity and taming inflammation’s storm. Front. Immunol. 17:1777470. doi: 10.3389/fimmu.2026.1777470
Received
29 December 2025
Revised
28 January 2026
Accepted
29 January 2026
Published
29 April 2026
Volume
17 – 2026
Edited by
Junji Xing, Houston Methodist Research Institute, United States
Reviewed by
Lisa Ulrike Teufel, Radboud University Medical Centre, Netherlands
Omkar Shinde, Sinhgad Dental College and Hospital, India
Updates

Check for updates
Copyright
© 2026 Li and Wang.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dongdong Wang, 2025070013@yibinu.edu.cn
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.