Human milk fortifiers are increasingly seen as essential products in medical nutrition in Japan, as they are added to mother’s or donor milk fed to premature babies to boost nutrients. While government subsidies are dismantling barriers to accessing these products, breakthrough clinical research underscores their essential role in brain support. Early growth is no longer a matter of weight gain, but is a critical window for neurodevelopmental health.
Nutrition Insight speaks to Melinda Eliott, MD, neonatologist and chief medical officer at Prolacta Bioscience, about the latest regulatory shifts propelling the sector in Japan, including the country’s decision to classify these products as subsidized prescription medicines. She also highlights emerging European guidelines that treat such products as a vital Substance of Human Origin.
“The use of human milk-based fortifiers is gaining momentum both clinically and commercially, as evidenced by Japan’s recent prescription drug designation of Prolacta Bioscience’s PreemieFort human milk-based fortifiers. These are the same products as Prolact+ in the US, and Humavant outside of the US, Canada, and Japan,” Eliott tells us.
Clinical and commercial momentum
Human milk fortifiers are added to breast milk to provide the extra calories, protein, and other nutrients premature infants need to make up for the growth they missed in utero. Until now, these were primarily categorized as niche nutritional supplements — a supportive but secondary element of neonatal care.
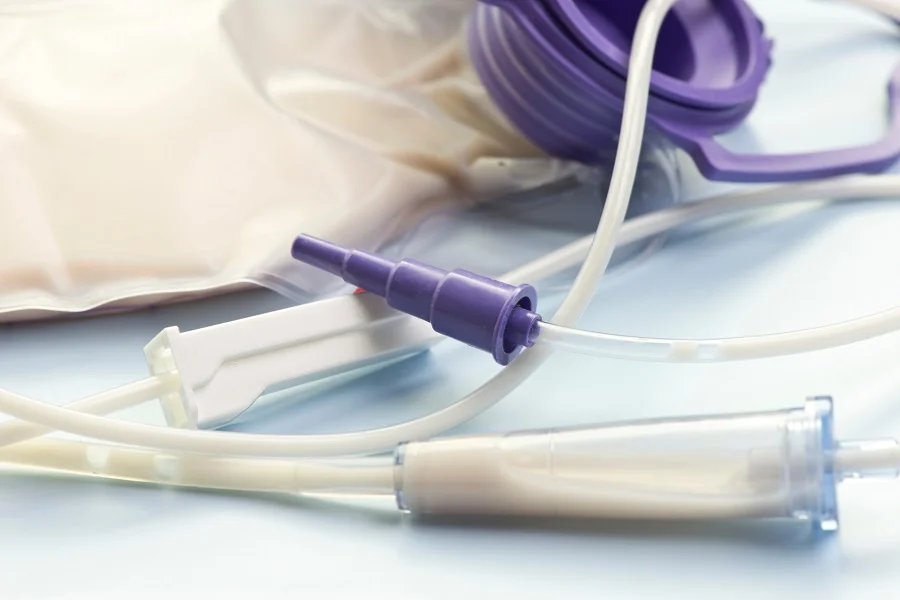
 Prolacta Bioscience’s PreemieFort Enteral Solution fortifier is made from human donor milk, although fortifiers can also be made using cow milk (Image credit: Prolacta Bioscience).Prolacta Bioscience’s PreemieFort Enteral Solution fortifier is made from human donor milk, although fortifiers can also be made using cow milk. Japan’s world-first drug approval of the product took it beyond the status of a nutritional supplement to a necessary medicine with proven outcomes.
Prolacta Bioscience’s PreemieFort Enteral Solution fortifier is made from human donor milk, although fortifiers can also be made using cow milk (Image credit: Prolacta Bioscience).Prolacta Bioscience’s PreemieFort Enteral Solution fortifier is made from human donor milk, although fortifiers can also be made using cow milk. Japan’s world-first drug approval of the product took it beyond the status of a nutritional supplement to a necessary medicine with proven outcomes.
“Drug designation also enables national reimbursement, meaning access becomes equitable and consistent, not dependent on hospitals or insurance providers,” Eliott highlights. “Japanese families of very preterm infants are lucky because all vulnerable babies have access to PreemieFort. That means their baby won’t just survive, but will have the best chance to thrive, thanks to better early growth.”
In Europe, she adds that Croatia already has national reimbursement coverage for Prolacta’s human milk-based fortifiers, and Poland is close behind.
“Europe is also changing how it classifies human milk products. With its SoHO guidelines (Substances of Human Origin), which are expected in 2027, the European Commission has stated that food law is not fit to manage the clinical intent, donor protection, ethical constraints, or biological risks associated with human milk products.”
“To date, more than 125,000 premature infants globally have benefited from Prolacta’s human milk-based fortifiers and formulas. It’s encouraging to see regulatory progress bringing broader, more equitable access to Neonatal Intensive Care Units (NICUs) globally.”
Thriving beyond survival
Published in Nature, a recent landmark clinical trial, “Joint Assessment of Systematic Metabolism and Intestinal Nutrition and Epithelium” (JASMINE), explored the growth outcomes in preterm infants fed Prolacta Bioscience’s human-milk-based fortifier. The findings explain how early growth is a critical marker for long-term developmental health, particularly for the brain.
“Early growth is critical for preterm infants because the brain doesn’t wait,” Eliott stresses. “What we feed premature infants in the first days and weeks of life directly shapes their long-term neurodevelopment because that is when the brain is growing at its most rapid pace.”
She says that a number of independent studies have shown that when premature infants are prescribed full enteral (using the gastrointestinal tract) feeding earlier, their brains grow better and have healthier neurodevelopmental outcomes.
“Japan has some of the world’s best premature infant survival rates, but the country accomplished this in part by avoiding or delaying cow milk-based products, due to the known risk of necrotizing enterocolitis (NEC) and other complications. This approach increased survival, but often at the cost of early growth, which may compromise neurodevelopment,” she highlights.
“JASMINE demonstrated that with early fortification using Prolacta’s human milk-based fortifiers, clinicians don’t have to trade off safety for growth. Very low birth weight infants in the human milk arm showed better weight and length gain velocity, as well as improved head circumference growth, without compromising Japan’s exceptionally low NEC rate (<2%).”
Evolving category of medical nutrition
Japan approved these fortifiers as prescription drugs rather than nutrition products, which could signal a wider regulatory shift around medical nutrition.
Japan approved these fortifiers as prescription drugs rather than nutrition products, which could signal a wider regulatory shift around medical nutrition.
“Japan’s decision reflects a growing global recognition that human milk is a biological solution, like blood or plasma, rather than a food product,” says Eliott.
“What Japan accomplished is meaningful on multiple fronts. The approval required a New Drug Application filing with Japan’s Pharmaceuticals and Medical Devices Agency, a rigorous Good Clinical Practice audit, and full Good Manufacturing Practice manufacturing inspections in California.”
She adds that human milk fortifiers’ drug designation also enables national reimbursement under Japan’s universal health care system. “This means every baby who needs PreemieFort can now receive it. That’s a template other countries can follow, and I expect many of them are paying close attention.”
Barriers to wider adoption
Eliott also highlights the biggest barriers preventing wider adoption of human milk-based fortifiers in neonatal care today. “Cost and reimbursement remain the primary barrier for wider adoption of human milk-based nutrition. Many neonatologists have repeatedly said to me, ‘if these products were free, we would definitely use them.’”
“However, in my unit, we showed cost savings when we adopted Prolacta’s fortifiers because preemies just did better, had fewer complications, and went home sooner. But that is hard for some hospital administrators to fully understand,” she adds.
 Human milk fortifiers address the significant variability in donor human milk across milk banks.“By granting human milk-based fortifiers drug designation, it puts them squarely in the pharmacy cost calculation, which could influence changing DRG (Diagnosis-Related Group) payments in the US. With hospital reimbursement, it removes the number one barrier to access, just as it does for other life-supporting prescription products.”
Human milk fortifiers address the significant variability in donor human milk across milk banks.“By granting human milk-based fortifiers drug designation, it puts them squarely in the pharmacy cost calculation, which could influence changing DRG (Diagnosis-Related Group) payments in the US. With hospital reimbursement, it removes the number one barrier to access, just as it does for other life-supporting prescription products.”
Propelling precision nutrition
Eliott believes human milk-based fortification for extremely preterm infants may be the most compelling case for why precision nutrition matters.
“These infants have highly specific nutritional needs,” she notes. “The composition of what we feed them in the first weeks of life will influence their brain architecture, immune development, gut microbiome, and long-term outcomes.”
Additionally, she highlights the significant variability in mothers’ own milk and donor human milk across milk banks. “This variability needs to be addressed when feeding these very small and critically ill infants.”
“Adjusting the feedings for that particular baby with human milk-based fortifiers allows for more precise nutrition for that infant. But it is not just the macronutrients (protein, fat, and carbohydrates); it is where these macronutrients come from. Human milk is meant for human babies. This is why Japan has changed its standard of care and adopted human milk-based nutrition as essential medicine.”
A growing body of evidence links early human milk-based fortification to improved neurodevelopment. Eliott points to a 2022 study that showed that infants fed Prolacta’s exclusive human milk (EHMD) diet had improved cognitive outcomes at 18–22 months, compared to those fed cow milk-based products.
“And the 2025 Chou study of 13 Kaiser Permanente NICUs — with more than 1,000 infants — found improved motor outcomes at age three for premature infants who received an EHMD compared to those who did not.”
“That’s precision nutrition delivering on its promise for the most vulnerable patients in medicine,” she concludes.