 Kemin’s live homologous genotype 7 vaccine demonstrated superior immunogenicity, early onset of immunity, long-lasting protection, and significant reduction in virus shedding against a velogenic NDV GVII strain. Photo: Kemin
Kemin’s live homologous genotype 7 vaccine demonstrated superior immunogenicity, early onset of immunity, long-lasting protection, and significant reduction in virus shedding against a velogenic NDV GVII strain. Photo: Kemin
Newcastle Disease virus (NDV) is a major pathogen that compromises flock health and profitability in modern poultry production. Among the different genotypes, Genotype VII (GVII) is widely recognised as the predominant and most virulent form, responsible for major outbreaks in Asia, South Africa and several European countries.
Many existing vaccines, particularly those based on classical strains such as LaSota, have been reported to provide insufficient protection against GVII, which may be linked to genotype mismatch in vaccination. Therefore, developing genotype-matched vaccines is crucial for effective disease control. This study aimed to evaluate the immunogenicity, onset of immunity and protective efficacy of the Kemin live homologous vaccine against challenge with a velogenic NDV GVII strain.
Materials and method
A recombinant live NDV vaccine using a GVII and VG/GAstrains [rNDV:VG/GA-F7] was used, and for the challenge, a velogenic NDV GVII strain (EG-567F) was used. A total of 100 day-old Specific Pathogen Free (SPF) White Leghorn chickens were obtained and randomly assigned into 3 groups. Birds were housed in isolators under controlled conditions and fed ad libitum.
Subgroups were challenged intramuscularly at different time points (Table1) with a standardised dose (5.0 log10 ELD50/100µL) of the velogenic NDV GVII strain. Oropharyngeal swabs were collected at 3, 5, 7, and 10 days post-challenge to monitor virus shedding. Birds were observed daily for clinical signs, survival, and recovery.
Blood samples were collected weekly up to 6 weeks to measure antibody titers using the Hemagglutination Inhibition (HI) test against GVII antigen, with a titer above 2 log2 considered positive. Oropharyngeal swabs were taken at 3, 5, 7, and 10 days post-challenge to monitor virus shedding. Additionally, daily clinical observations and mortality were recorded throughout the study.
Vaccine immunogenicity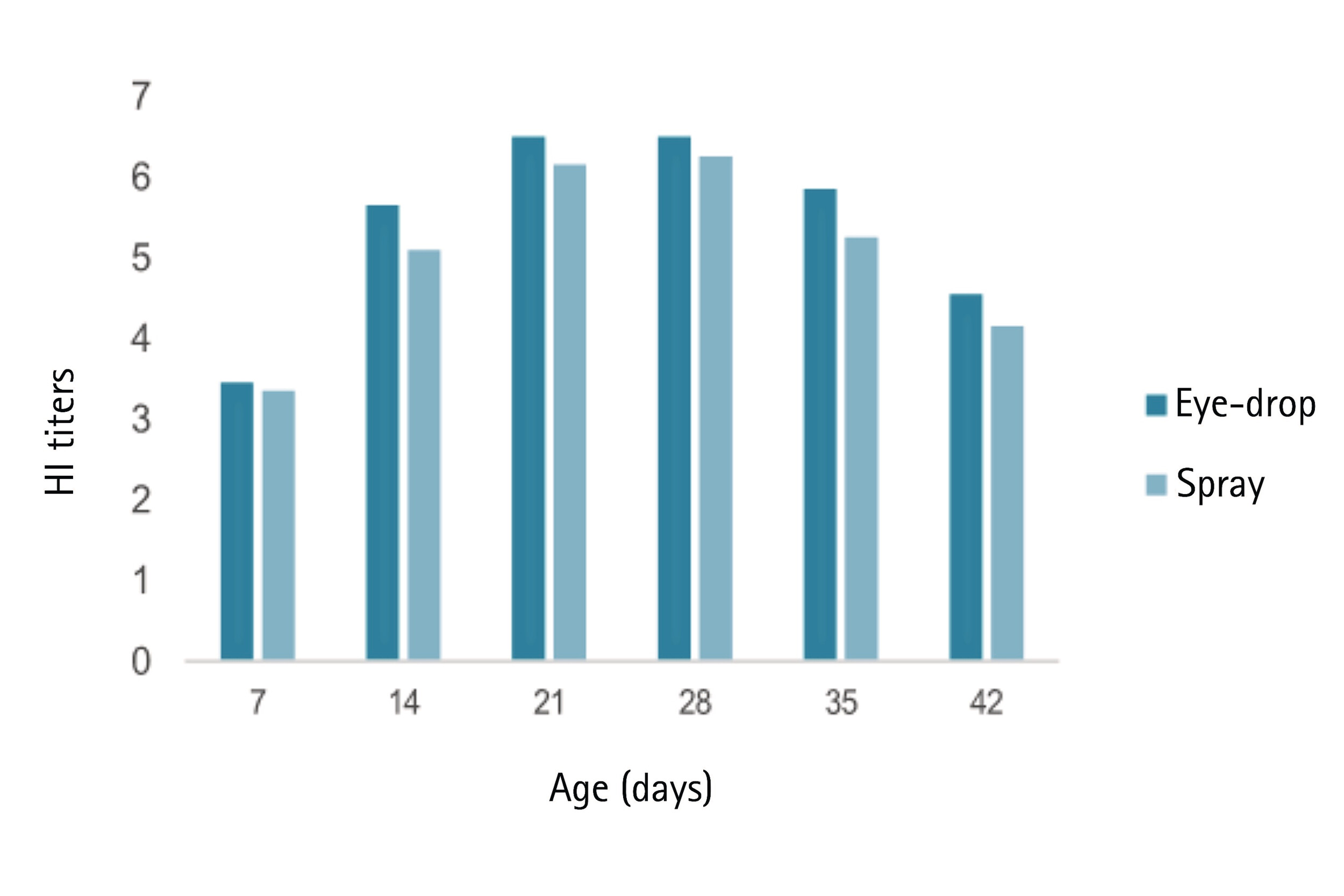 Figure 1 – Antibody response (HI titer) against NDV GVII over time
Figure 1 – Antibody response (HI titer) against NDV GVII over time
Detectable HI antibody titers appeared early at 7 days post-vaccination. By day14, mean titersagainst NDV GVII were 5.65±0.88 for the eye-drop group and 5.10±0.64 for the spray group. Peaking around 21 days, reaching6.50±0.69 and 6.15±0.75, respectively.
Antibody titers remained above the protective threshold (>4.0 log2) throughout the 6-week study period. No significant difference was observed between administration methods.
Protective efficacy post challenge Figure 2 – Survival observations post challenge at 42 days
Figure 2 – Survival observations post challenge at 42 days
Following the 3-week challenge, 100% of vaccinated birds survived with minimal clinicalsigns such as mild ruffled feathers and slight respiratory symptoms, which resolved within a few days. In the 6-week challenge, vaccinated birds exhibited a minimum of 80% survival rate, demonstrating strong, lasting immunity. In contrast, all unvaccinated birds died within 6 days post-challenge, confirming the lethality of the challenge virus and validating the model.
Vaccinated birds showed a reduction in virus shedding compared to unvaccinated controls. In the 3-week challenge group, no virus was detected from vaccinated birds beyond 7 days post-challenge. In the 6-week challenge group, virus shedding ceased completely by 10 days post-challenge. On the other hand, unvaccinated control birds exhibited 100% virus shedding until death.
Early and strong immunity, lasting protection and reduced shedding
The rapid seroconversion within 7 days and strong antibody titers by 14–21 days demonstrated the vaccine’s ability to trigger early protection, a very important point in commercial poultry systems where birds are exposed early to pathogens.
Even at 6 weeks post-vaccination (observation period of the study), vaccinated birds maintained high antibody titers and protection rates, confirming the tested vaccine´s capacity for extended immunity beyond the onset of exposure. Importantly, vaccinated birds cleared virus shedding much earlier than unvaccinated controls. This reduction limits virus transmission and environmental contamination.
Both administration routes eye-drop and spray provided comparable protection, offering flexibility for field use depending on operational preferences and bird population size.
Conclusion
Kemin’s live homologous genotype 7 vaccine demonstrated superior immunogenicity, early onset of immunity, long-lasting protection, and significant reduction in virus shedding against a velogenic NDV GVII strain. The tested vaccine effectively prevented mortality due to challenge with NDV GVII strain therefore represents a reliable solution for controlling Newcastle Disease, particularly in regions where GVII challenges predominate.
References are available on request.

Kemin Company profile
Kemin provides “inspired molecular solutions” specifically developed to provide nutrition and health benefits for humans and animals. Committed to feed and food safety, Kemin maintains top-of-the-line manufacturing facilities where approximately 500 specialty ingredients are made for the global feed and food industries as well as the health, nutrition and beauty markets. A privately held, family-owned and operated company, Kemin has nearly 2,000 employees and operates in more than 90 countries with manufacturing facilities in Belgium, Brazil, China, India, Italy, Singapore, South Africa and the United States. Visit Kemin.com






