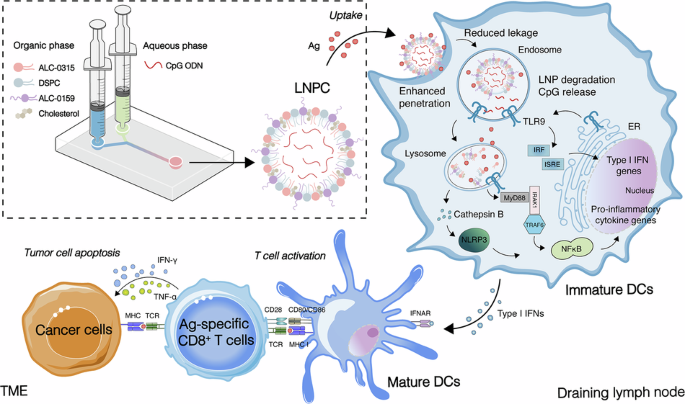
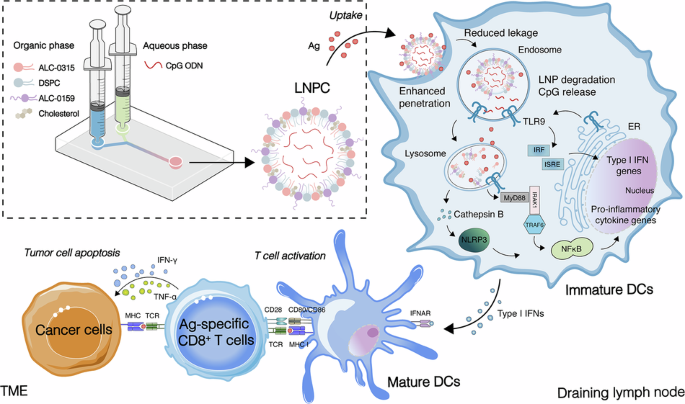
The success of therapeutic cancer vaccines hinges on overcoming the immunosuppressive tumor microenvironment (TME) and activating robust, durable cellular immunity. Here, we engineered a highly translatable vaccine adjuvant delivery system (LNPC) by integrating the Toll-like receptor 9 (TLR9) agonist CpG with the FDA-approved ionizable lipid ALC-0315. This lipid nanoparticle (LNP) architecture establishes a safe, localized immunological depot that completely shields CpG from systemic nuclease degradation. Crucially, it enables the spatiotemporally coordinated co-delivery of physically admixed antigens and features a “smart” pH-responsive mechanism for targeted endosomal payload release. Consequently, LNPC efficiently activates the TLR9/MyD88 signaling pathway, promotes robust dendritic cell maturation, and drives a pronounced Th1-biased polarization. It uniquely induces the massive expansion of polyfunctional CD8+ T cells across diverse immunogens. In aggressive B16F10-OVA melanoma and HPV-associated TC-1 tumor models, LNPC-adjuvanted vaccines successfully remodeled the highly suppressive TME, achieving near-complete tumor clearance, significantly prolonged survival, and durable immune memory without inducing off-target systemic toxicity. Ultimately, the LNPC system represents a versatile, highly effective adjuvant platform with substantial clinical translational potential, offering a compelling new strategy for personalized tumor immunotherapy.
 The alternative text for this image may have been generated using AI.
The alternative text for this image may have been generated using AI.