(Photo Credit: United States Consumer Product Safety Commission)
Vitaquest recall overview:
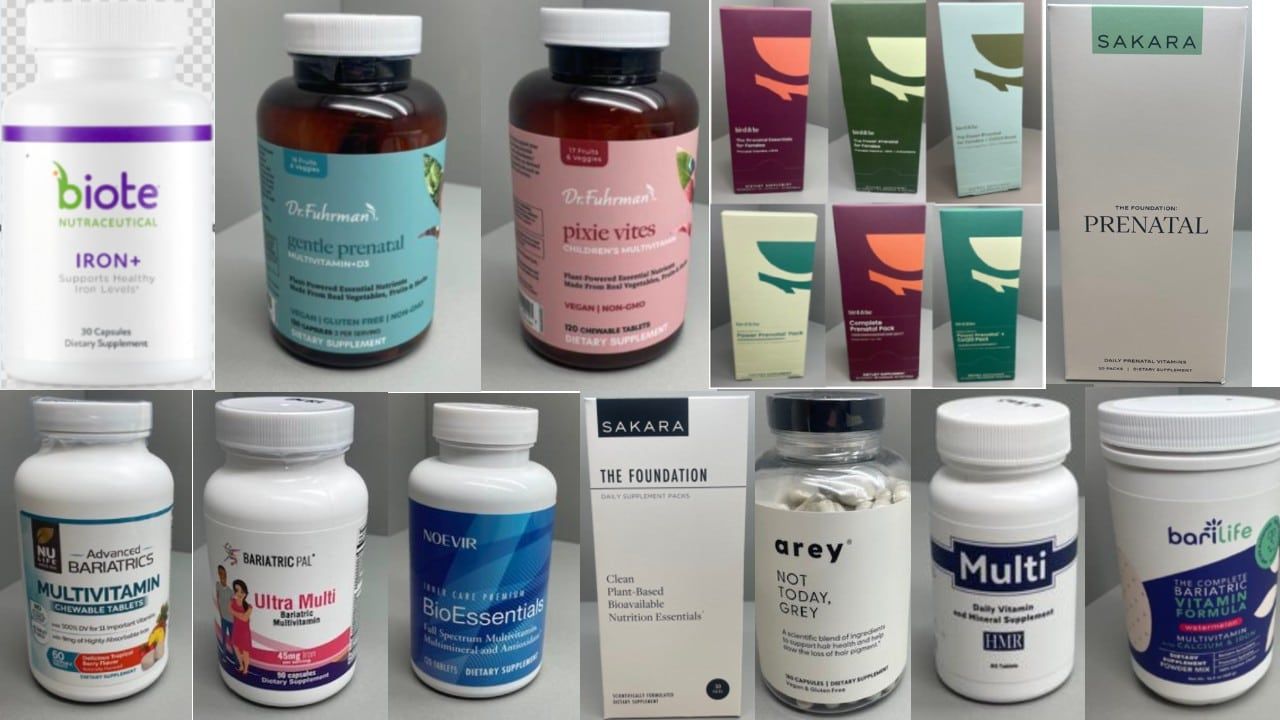
Who: Vitaquest International is recalling approximately 356,140 units of various iron-containing dietary supplements.
Why: The recall is due to the supplements’ failure to meet child-resistant packaging standards, posing a risk of poisoning to young children.
Where: The recall is effective in the United States.
Vitaquest International is recalling more than 356,000 units of iron-containing dietary supplements due to packaging that fails to meet child-resistant standards. This oversight presents a serious risk of poisoning if young children accidentally ingest the Vitaquest supplements.
The recall, announced on April 9, affects a range of products from brands, including Arey, Bari Life, Bird&Be, Biote, Dr. Fuhrman, NuLife, HMR, Bariatric Pal, Noevir, Zenbean and Sakara.
These products, which include multivitamins and prenatal supplements, are packaged in containers that do not comply with the Poison Prevention Packaging Act’s requirements for child-resistant packaging.
“The dietary supplements contain iron, which must be in child-resistant packaging as required by the Poison Prevention Packaging Act,” the recall notice states.
Recall urges consumers to keep Vitaquest supplements out of reach of children
Consumers are advised to store these supplements out of children’s reach and contact Vitaquest International for a free child-resistant replacement cap or storage pouch.
The recall affects products sold nationwide at various retailers, including Credo Beauty, Erewhon and The Vitamin Shoppe, as well as online platforms like Amazon.com, from April 2023 through February 2026.
Vitaquest International is taking steps to mitigate the risk by offering free replacement caps or storage pouches that meet child-resistant standards.
For further information, consumers can contact Vitaquest International toll-free at 844-298-4545, email [email protected] or visit its website.
Vitaquest International reports that no injuries or illnesses have been linked to the recalled supplements so far. The company is not currently facing legal action over the recall, but Top Class Actions monitors such recalls closely as they sometimes lead to class action lawsuits.
Are you affected by the Vitaquest recall? Let us know in the comments.
Read About More Class Action Lawsuits & Class Action Settlements:
We tell you about cash you can claim EVERY WEEK! Sign up for our free newsletter.
“*” indicates required fields