 Researchers have identified a small RNA molecule that appears to play a central role in disrupting how the liver manages fat, offering new insight into a widespread metabolic disease. Credit: StockA hidden molecular switch linked to liver disease may be reversible, using an unexpected, familiar compound.
Researchers have identified a small RNA molecule that appears to play a central role in disrupting how the liver manages fat, offering new insight into a widespread metabolic disease. Credit: StockA hidden molecular switch linked to liver disease may be reversible, using an unexpected, familiar compound.
About 30% of people worldwide are affected by metabolic-associated fatty liver disease (MASLD), a condition closely tied to obesity and type 2 diabetes that can quietly progress to liver damage. Despite its growing impact, treatment options remain limited. New research now points to an unexpected target, and a familiar vitamin, that could help change that.
A team led by Jang Hyun Choi at UNIST, in collaboration with Hwayoung Yun at Pusan National University and Neung Hwa Park at Ulsan University Hospital, has identified a small RNA molecule called microRNA-93 (miR-93) as a central driver of the disease. Their findings show that this molecule acts like a molecular switch that disrupts how the liver processes fat.
MicroRNAs are short strands of RNA that fine-tune gene activity. In this case, miR-93 was found at unusually high levels in liver tissue from both patients with MASLD and mice fed diets rich in fat and fructose, a combination known to mimic modern dietary patterns. Elevated miR-93 interferes with the liver’s ability to break down fat by blocking SIRT1, a gene that helps regulate energy use and fat metabolism.
Disrupted Metabolism and Energy Balance
This disruption has wide-ranging effects. When SIRT1 is suppressed, the liver shifts toward storing fat instead of burning it. The researchers found that miR-93 also alters broader metabolic pathways, reducing fatty acid oxidation while increasing processes linked to cholesterol production.
It also dampens the LKB1 AMPK signaling pathway, a key system that normally helps cells respond to energy stress and maintain metabolic balance.
 Mitochondria play a central role in energy metabolism, which is impaired in MASLD. Credit: Stock
Mitochondria play a central role in energy metabolism, which is impaired in MASLD. Credit: Stock
To test whether reversing this process could improve liver health, the team created mice that could not produce miR-93. Even when fed a high-fat, high-fructose diet, these mice developed far less fat buildup in the liver. They also showed better blood sugar control and improved mitochondrial function, suggesting healthier energy metabolism at the cellular level.
A Familiar Vitamin Emerges as a Candidate
The researchers then screened 150 FDA-approved drugs to find compounds that could lower miR-93. Niacin, also known as vitamin B3, stood out as the most effective. In treated mice, niacin reduced miR-93 levels, restored SIRT1 activity, and reactivated the liver’s fat-burning pathways. As a result, fat accumulation in the liver dropped and overall metabolic function improved.
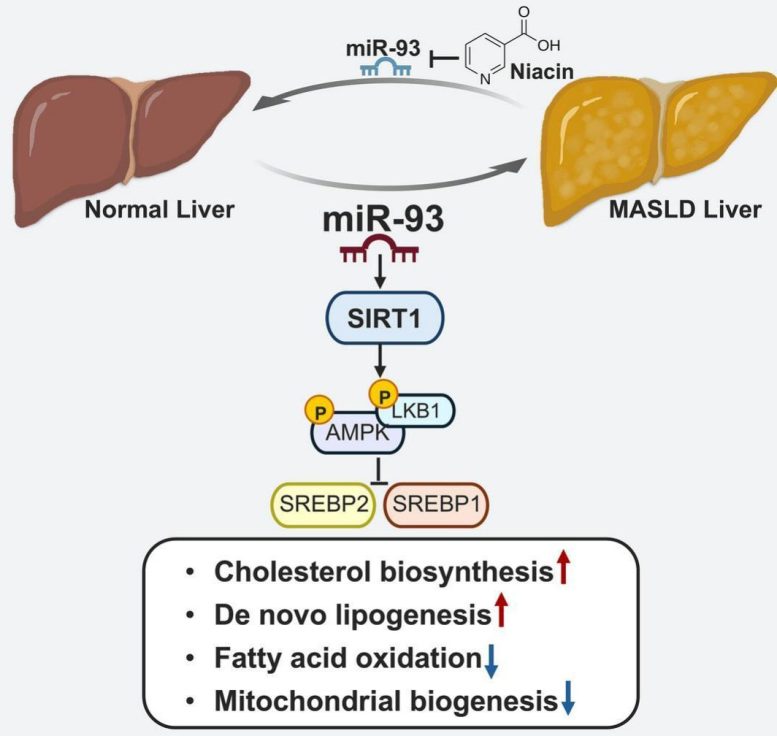 Graphical abstract illustrating the proposed mechanism by which miR-93 drives metabolic dysfunction-associated steatotic liver disease (MASLD). Credit: 10.1016/j.metabol.2025.156266
Graphical abstract illustrating the proposed mechanism by which miR-93 drives metabolic dysfunction-associated steatotic liver disease (MASLD). Credit: 10.1016/j.metabol.2025.156266
Niacin is already widely used to manage high cholesterol, which could make it an attractive candidate for repurposing. However, the researchers note that its effects in MASLD patients will need to be confirmed in clinical studies, especially since high doses of niacin can have side effects and must be carefully managed.
The team explained, “This study precisely elucidates the molecular origin of MASLD and demonstrates the potential for repurposing an already approved vitamin compound to modulate this pathway, which has high translational clinical relevance.”
They added, “Given that niacin is a well-established and safe medication used to treat hyperlipidemia, it holds promise as a candidate for combination therapies targeting miRNA pathways in MASLD.”
A related clinical study is already underway (ClinicalTrials.gov ID: NCT06843148), led by the Université de Sherbrooke. This randomized crossover trial is examining how niacin influences fat distribution and metabolic responses in individuals with MASLD, with a focus on whether it can shift fatty acids away from the liver. The results may help determine whether this approach translates into measurable benefits in patients.
Reference: “Hepatic miR-93 promotes the pathogenesis of metabolic dysfunction-associated steatotic liver disease by suppressing SIRT1” by Yo Han Lee, Jinyoung Lee, Joonho Jeong, Kieun Park, Bukyung Baik, Yuseong Kwon, Kimyeong Kim, Keon Woo Khim, Haneul Ji, Ji Young Lee, Kwangho Kim, Ji Won Kim, Tam Dao, Misung Kim, Tae Young Lee, Yong Ryoul Yang, Haejin Yoon, Dongryeol Ryu, Seonghwan Hwang, Haeseung Lee and Jang Hyun Choi, 12 April 2025, Metabolism.
DOI: 10.1016/j.metabol.2025.156266
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.