Anyone who has weathered a bad stomach bug knows the feeling: a loss of appetite that sets in and lingers, even after the initial illness. The same thing happens for millions of people worldwide with long-term parasitic worm infections. But scientists have long puzzled over exactly why.
Now, researchers at UC San Francisco have traced the molecular pathway that connects the gut immune system to the brain during a parasitic infection, explaining how it causes a loss of appetite.
“The question we wanted to answer was not just how the immune system fights parasites, but how it recruits the nervous system to change behavior,” said co-senior author David Julius , PhD, professor and chair of Physiology at UCSF and recipient of the 2021 Nobel Prize in Physiology or Medicine. “It turns out there’s a very elegant molecular logic to how that happens.”
The findings, published in Nature on March 25, reveal an unexpected communication system between two cell types and could shed light on a range of conditions involving gut discomfort – from food intolerances to irritable bowel syndrome.
Two cells communicating
The new study focused on two rare cell types in the gut. Tuft cells detect parasites and trigger an immune response. Enterochromaffin (EC) cells send signals that activate nerve fibers leading to the brain. EC cells are known to cause nausea, pain, and gut discomfort, but whether they communicate with tuft cells was unknown.
“My lab has long been interested in how tuft cells, after they initially respond to a parasitic infection, release signals to other cell types,” said co-senior author Richard Locksley , MD, a UCSF immunologist.
Image
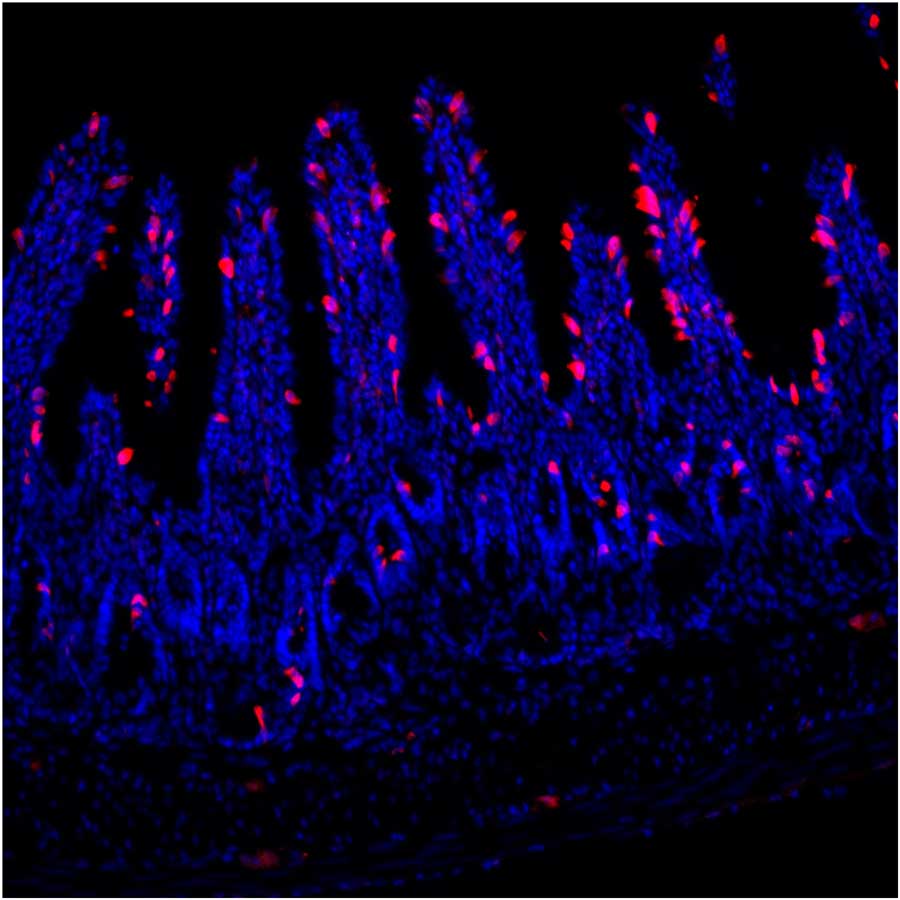
Specialized immune-sensing cells called tuft cells (red) in inflamed intestinal tissue. During parasitic infection, the body produces many more of these cells to help fight back. The blue staining shows intestinal cell nuclei.
Image by Koki Tohara, UCSF
First author Koki Tohara , PhD, a postdoctoral researcher at UCSF, found the answer by placing genetically engineered sensor cells next to tuft cells under a microscope. When the tuft cells were exposed to succinate, a molecule produced by parasitic worms, the sensor cells lit up. This revealed that tuft cells were releasing acetylcholine, a common chemical messenger for nerve cells.
When acetylcholine was added to lab-grown gut tissue containing EC cells, they released serotonin. This activated vagal nerve fibers that carry signals from the gut to the brain.
“What we found is that tuft cells are doing something neurons do, but by a completely different mechanism,” Tohara said. “They’re using acetylcholine to communicate, but without any of the usual cellular machinery that neurons rely on to release it.”
The team also discovered that tuft cells release acetylcholine in two distinct phases, which explains why people often don’t lose their appetite until days into an infection. In the first phase, a brief burst of acetylcholine is released. Later, after the immune system has mounted a full response, tuft cells multiply and produce a slow, sustained release of acetylcholine that activates EC cells.
“This explains why you feel fine at first, but then start to feel sick as the infection becomes established,” Julius said. “The gut is essentially waiting to confirm that the threat is real and persistent before it tells the brain to change your behavior.”
Image
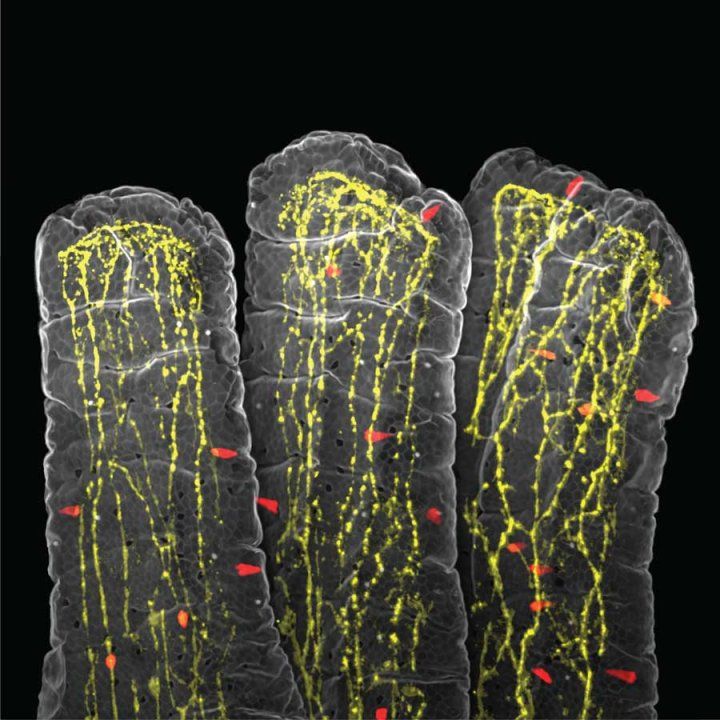
A 3D view inside the small intestine showing its finger-like projections called villi. Red cells release serotonin, a chemical messenger that triggers sensations like nausea. Yellow fibers are nerve endings that detect these signals and send them to the brain. Image by Koki Tohara, UCSFImplications beyond parasites
To test whether the pathway matters beyond the lab, the researchers tracked how much food mice ate during a parasitic worm infection. As expected, mice with normal tuft cell function ate less as the infection took hold. But mice that could not produce acetylcholine in their tuft cells kept eating normally, confirming that the molecular chain drives the behavioral response.
The new findings could be relevant for treating the symptoms of a parasite infection.
“Controlling the outputs of tuft cells could be a way to control some of the physiologic responses associated with these infections,” Locksley said, adding that the study also could have broader implications.
Tuft cells are found throughout the body – not just in the gut, but also the airways, gallbladder, and reproductive tract. Disruptions in this pathway could contribute to conditions like irritable bowel syndrome, food intolerances, and chronic visceral pain.
The work was carried out in collaboration with Stuart Brierly, PhD, and his lab at the University of Adelaide in Australia.
Authors: Other UCSF authors are: Jinhao Xu, PhD, Hong-Erh Liang, PhD, and Nathan D. Rossen. For a full list of authors, see the paper.
Funding: The study was supported in part by the National Institutes of Health (NS105038, NS113869 and DK135714), the BRAIN Initiative (1U01NS113358, 1U01NS120824, AI026918, HL107202), the National Natural Science Foundation of China (31925017), the New Cornerstone Science Foundation, the XPLORER PRIZE, the Howard Hughes Medical Institute, the National Health and Medical Research Council of Australia (APP2008727), a NHMRC Ideas Grant (APP2029332), the Damon Runyon Cancer Research Foundation (DRG-(2387-30)), and a Larry L. Hillblom Fellowship Grant (2024-A-044-FEL).
/Public Release. This material from the originating organization/author(s) might be of the point-in-time nature, and edited for clarity, style and length. Mirage.News does not take institutional positions or sides, and all views, positions, and conclusions expressed herein are solely those of the author(s).View in full here.




